Trophic ecology and gill raker morphology of seven catostomid species in Iowa rivers
|
|
|
- Claude George
- 6 years ago
- Views:
Transcription
1 Journal of Applied Ichthyology J. Appl. Ichthyol. 27 (2011), Published This article is a US Government work and is in the public domain in the USA ISSN Received: June 14, 2010 Accepted: January 10, 2011 doi: /j x Trophic ecology and gill raker morphology of seven catostomid species in Iowa rivers By J. R. Spiegel 1, M. C. Quist 2 and J. E. Morris 1 1 Department of Natural Resource Ecology and Management, Iowa State University, Ames, IA, USA; 2 U.S. Geological Survey, Idaho Cooperative Fish and Wildlife Research Unit, Department of Fish and Wildlife Resources, University of Idaho, Moscow, ID, USA Summary Understanding the trophic ecology of closely-related species is important for providing insight on inter-specific competition and resource partitioning. Although catostomids often dominate fish assemblages in lotic systems, little research has been conducted on their ecology. This study was developed to provide information on the trophic ecology of catostomids in several Iowa rivers. Food habits, diet overlap, and gill raker morphology were examined for highfin carpsucker Carpiodes velifer, quillback C. cyprinus, river carpsucker C. carpio, golden redhorse Moxostoma erythrurum, shorthead redhorse M. macrolepidotum, silver redhorse M. anisurum, and northern hogsucker Hypentelium nigricans sampled from four Iowa rivers (2009). Diet overlap among all species was calculated with MoristaÕs index (C). Food habit niche width was quantified with LevinÕs index (B) and similarity in gill raker morphology was compared with analysis of covariance. Values from MoristaÕs index suggested significant overlap in the diets of highfin carpsucker and river carpsucker (C = 0.81), quillback and river carpsucker (C = 0.66), and shorthead redhorse and silver redhorse (C = 0.67). LevinÕs index indicated that golden redhorse (B = 0.32), quillback (B = 0.53), and river carpsucker (B = 0.41) had the most generalized feeding strategies as their food niche widths were substantially wider than the other species. Gill raker length and spacing were positively correlated with the standard length of the fish for all species (gill raker length: r 2 = , P 0.01; gill raker spacing: r 2 = , P 0.01). Slopes of regression of gill raker length and spacing to standard lengths were significantly (P 0.05) different among species, indicating that rates of change in gill raker morphology with body length varied among species. Differences in gill raker morphology likely allow catostomids to partition resources and reduce competitive interactions. Introduction The family Catostomidae consists of 76 species of fish that are widely distributed across North America (Cooke et al., 2005). Like many groups of fishes, catostomids are of high conservation concern in many regions due to their life history characteristics (e.g. movement patterns) and the widespread degradation of aquatic habitats (e.g. Cooke et al., 2005; Jelks et al., 2008). Of the 17 species native to Iowa, two species have been extirpated (i.e. lake chubsucker Erimyzon sucetta, greater redhorse Moxostoma valenciennesi) and five species (i.e. river redhorse M. carinatum, black redhorse M. duquesnei, silver redhorse M. anisurum, highfin carpsucker Carpiodes velifer, black buffalo Ictiobus niger) are either declining or their status is unknown (Zohrer, 2006). Catostomids in Iowa are threatened by the same habitat alterations that have caused 68 of the 144 native fish species in Iowa to be identified as species of greatest conservation need (Zohrer, 2006). Since European settlement, the landscape of Iowa has undergone significant change whereby agriculture has altered over 80% of the land area (Natural Resources Conservation Service, 2000). Changes in land use from native prairie and forest to row crops and pasture have had substantial deleterious effects on lotic systems, including altered substrate composition (i.e. sedimentation), water quality (e.g. temperature, nutrient dynamics), and flow regimes (Morgan et al., 1983; Cooper, 1987; Holopainen and Huttunen, 1992; Litvan et al., 2007). Consequently, changes in watershed conditions and instream habitat have been associated with declines in fish assemblage diversity, reduced abundance and biomass, and shifts to assemblages dominated by trophic generalists (Karr et al., 1985; Walser and Bart, 1999). Many of the changes to the landscape occurred prior to the documentation of fish assemblages in Iowa, thus there is little historical data about native catostomid populations. Traditionally, catostomids have received much less attention than prominent riverine sport fish species, such as smallmouth bass Micropterus dolomieui and channel catfish Ictalurus punctatus. However, catostomids and other non-game fishes (e.g. cyprinids, small-bodied percids and ictalurids) are an important component of aquatic food webs (Bertrand and Gido, 2007). The diversity of catostomids in Iowa is high with 17 species from seven genera (i.e. Cycleptus, Erimyzon, Ictiobus, Carpiodes, Moxostoma, Minytrema,andHypentelium) native to the stateõs lakes, streams, and rivers (Zohrer, 2006). Many of these species have similar external morphology and often occur in sympatry. Thus, understanding how catostomids partition food resources may provide insight on inter-specific interactions, niche overlap, and food resource partitioning that allow for co-occurrence of species and may also provide insight with regard to conservation and management (McNeely, 1987; Gray et al., 1997). In addition to describing and comparing diets, knowledge of gill raker morphology may provide additional insight on the trophic ecology of fishes (e.g. Barton, 2007; Miller et al., 2007; Ingram and Shurin, 2009). Gill rakers are highly diverse with respect to their length, shape, and spacing, and their morphology typically reflects diet (Barton, 2007). For example, species with long rakers and small spacing typically feed on small invertebrate prey items whereas U.S. Copyright Clearance Centre Code Statement: /2011/ $15.00/0
2 1160 J. R. Spiegel, M. C. Quist and J. E. Morris piscivores usually have short, stout, widely-spaced gill rakers (e.g. Ehlinger and Wilson, 1988; Barton, 2007; Wood, 2007). In this study, we examined food habits and gill raker morphology of catostomids in Iowa rivers. The objectives of this study were to: (i) describe the food habits and gill raker morphology, (ii) evaluate trophic overlap, and (iii) evaluate how gill raker morphology might relate to partitioning of food resources of seven species of catostomids in Iowa rivers. Materials and methods River carpsucker C. carpio, highfin carpsucker, quillback C. cyprinus, shorthead redhorse M. macrolepidotum, golden redhorse M. erythrurum, silver redhorse, and northern hogsucker Hypentelium nigricans were sampled from four nonwadeable rivers in central and eastern Iowa during Sampled were the Boone, North Raccoon, Shellrock, and Wapsipinicon rivers. All four river systems are in the Mississippi River basin. The Boone and North Raccoon rivers are located in central Iowa and are tributaries to the Des Moines River. The Shellrock and Wapsipinicon rivers are located in eastern Iowa and are direct tributaries to the Mississippi River. A 3-km reach was sampled from each river using boatmounted, pulsed-dc electrofishing (Smith Root VVP-15B, Vancouver, WA). Sampling sites were selected based on fish assemblage data collected in 2007 and 2008 by Iowa State University personnel (Neebling and Quist, 2010). Output was standardized to 3000 W based on water conductivity and dropper exposure (Burkhardt and Gutreuter, 1995). Each 3- km reach was separated into six 500-m sections to provide an estimate of variation for catch rates and to minimize stress to sampled fishes. Effort focused on channel habitats and was performed with a single pass in a downstream direction. All fish were collected by two netters. Although the species composition varied slightly among reaches, the seven catostomid species were relatively abundant across all reaches. The only exception was that river carpsucker was absent from the Wapsipinicon River. Fish were processed at the end of each 500-m section, identified, measured to the nearest mm for both total (TL) and standard length (SL), and weighed to the nearest gram. Fish were euthanized with a lethal dose of FinquelÔ (200 mg L )1 ; Argent Chemical Laboratories, Redmond, WA) and immediately processed in the field to minimize post-capture digestion. The digestive tract was removed from the esophagus by cutting at the junction of the esophagus and at the vent (Brezner, 1958). The entire digestive tract was fixed in 4% buffered formalin (Bowen, 1996). The gills including the pharyngeal pad were removed from the right side of the fish and similarly preserved. In the laboratory, gut contents were removed from the ÔgizzardÕ to the first turn of the intestine (approximately the anterior 20% of the digestive tract) and placed in a petri dish for processing (Welker and Scarnecchia, 2003). A compound dissecting microscope was used to identify and count prey items which were identified to the lowest practical taxonomic level. Because many of the prey items were severely damaged from ingestion and digestion, the lowest taxonomic level was typically Order. Several indices were used to quantify food habits. Frequency of occurrence (%F) of each food category and invertebrate taxonomic order was estimated. In addition, the number of invertebrates consumed by each fish species was used to calculate prey specific abundance (%SA). Diet overlap between fish species was calculated using MoristaÕs index. MoristaÕs index is a robust diet overlap index when prey numbers are available because it has the least bias for different resource distributions among species and varying sample size (Smith and Zaret, 1982). MoristaÕs index was calculated as: C jk ¼ P h n i P ij 2 Pn i n ij 1 N j 1 i P ij P ik þ P n i P ik h nik 1 N k 1 where C j,k is MoristaÕs index of diet overlap between species j and k, P ij and P ik are the proportion of resource i of the total resources used by species j and k respectively, n ij and n ik are the number of individuals of species j and k that use resource i, and N j and N k are the total number of each species in the sample. Index values >0.60 are generally considered to indicate significant overlap between two species (Zaret and Rand, 1971; Angradi and Griffith, 1990). In addition to MoristaÕs index, prey importance, feeding strategy, and niche width were examined using a modification of the graphical Costello method (Amundsen et al., 1996). The Costello method plots prey-specific abundance against frequency of occurrence for assessment of fish diets at the population level (Chipps and Garvey, 2007). The location of prey items on the axis of the plot provides insight into feeding strategy (i.e. specialist or generalist), prey importance (i.e. high or low), and the contribution of prey items to niche width (Amundsen et al., 1996). The diversity of food items consumed by catostomid species was further quantified by calculating LevinsÕ index: B i ¼ P 1 j P 2 ij 1 1A ðn 1Þ Where B i is LevinsÕ index for species i, and P ij is the proportion of prey j in predator iõs diet, and n is the number of prey categories (Marshall and Elliott, 1997). Index values vary from 0 to 1 with 0 representing species with specialized diets and 1 representing species with generalized diets. The first branchial arch from the right side of the body was removed to examine the length and spacing of gill rakers. The first branchial arch was selected because it typically contains the most well developed gill rakers (Nelson, 1967). Additionally, gill rakers located on other gill arches are generally smaller and contribute minimally to prey retention (OÕBrien and Luecke, 1983). Length and width of spacing between the inner edges of gill rakers was measured for five gill rakers centered on the angular gill arch (Tanaka et al., 2006). Gill raker length and spacing was measured to the nearest mm using a microscope connected to a computer using image analysis software (IMAGE LUS 5.1; Media Cybernetics, Bethesda, MD). Differences PROPLUS in length and spacing of gill rakers among species were tested using analysis of covariance (ANCOVA), with standard length of each fish used as the covariate (Tanaka et al., 2006). Analysis of covariance was used to test if the slope of the regression of fish standard length to gill raker length or spacing was statistically different between two species. Prior to statistical analysis, measurements of gill raker length and spacing were averaged for each individual fish. Results from all statistical tests were considered significant at a = All analyses were conducted using SAS (SAS Institute, 2008). i
3 Trophic ecology and gill raker morphology 1161 Results Stomachs were collected from 765 individuals, including 174 quillback, 123 highfin carpsucker, 135 river carpsucker, 107 golden redhorse, 124 shorthead redhorse, 60 northern hogsuckers, and 42 silver redhorse. Length varied within and among species (Table 1); mean lengths were typically lowest for northern hogsucker and highest for silver redhorse. Minimum and maximum total lengths were similar among species. Diets of the study species varied among species, but were generally dominated by invertebrates associated with benthic habitats (Table 2). Interestingly, the presence of aquatic insects that are typically found in the water column or on the surface (e.g. chironomid larvae and subimagos, Ephemeroptera subimagos) in the stomach contents suggests that catostomids consume prey found throughout the water column. Although the study species could be considered generalists by the diversity of prey items in their diet, several species exhibited notable specializations. For instance, highfin carpsuckers consumed more mollusks than the other species (%F = 23.7; %SA = 55.4; Table 2). Golden redhorse and northern hogsucker diets contained more Ephemeroptera (golden redhorse %F = 77.5; northern hogsucker %F = 66.0) and in higher numbers (golden redhorse %SA = 20.2; northern hogsucker %SA = 40.8) than the other catostomid species (Table 2). Chironomid larvae were the most numerous prey item for golden redhorse, shorthead redhorse, silver redhorse, and northern hogsucker and the third most important prey item for highfin carpsucker, quillback, and river carpsucker (Table 2). Oligochaetes and mollusks were the most numerous prey items for highfin carpsucker, quillback, and river carpsucker. Algae were more common in the diet of Carpiodes spp. than the other species (Table 2). Golden redhorse, quillback, and river carpsucker had the most generalized feeding strategies (Table 3) while highfin carpsucker, shorthead redhorse, silver redhorse, and northern hogsuckers had more specialized diets focused on a few prey items (Fig. 1). Results from the diet overlap analysis indicated that only three species pairs overlapped significantly with regard to diet composition. Those pairs included highfin carpsucker and river carpsucker (C = 0.81), quillback and river carpsucker (C = 0.66), and shorthead redhorse and silver redhorse (C = 0.67; Table 4). However, several other species pairs had moderate overlapping values between 0.40 and 0.60, including golden redhorse and shorthead redhorse (C = 0.42), golden redhorse and silver redhorse (C = 0.44), highfin carpsucker and quillback (C = 0.52), northern hogsucker and shorthead redhorse (C = 0.45), northern hogsucker and silver redhorse (C = 0.48), and river carpsucker and shorthead redhorse (C = 0.42; Table 4). Gill raker length and spacing was positively correlated with standard length for all species (length: r 2 = , P 0.01; spacing: r 2 = , P 0.01; Figs 2 and 3). Comparisons of the slopes of gill raker length to standard length were significantly different among species (P = ), indicating that rates of change in gill raker length with standard length varied by species. Gill raker length increased fastest with changes in length for Carpiodes spp. Quillback, river carpsucker, silver redhorse, and highfin carpsucker typically had the longest gill rakers for a given standard length. Similar to gill raker length, the slopes of gill raker spacing to standard length differed (P = ) among species. Gill raker spacing increased with increasing standard length at a Table 1 Mean, standard deviation (SD), minimum, and maximum length (mm) for seven catostomids sampled from four Iowa rivers, 2009 Common name Scientific name N Mean SD Min Max Highfin carpsucker Carpiodes velifer River carpsucker Carpiodes carpio Quillback Carpiodes cyprinus Northern hogsucker Hypentelium nigricans Golden redhorse Moxostoma erythrurum Shorthead redhorse Moxostoma macrolepidotum Silver redhorse Moxostoma anisurum Table 2 Frequency of occurrence (%F) and prey specific abundance (%SA) of prey items in diets of catostomids sampled from four Iowa rivers, Species include highfin carpsucker (HFCS), quillback (QBCK), river carpsucker (RVCS), golden redhorse (GORH), shorthead redhorse (SHRH), silver redhorse (SVRH), and northern hogsucker (NHGS). Prey items include unidentified Diptera (D), Chironomidae larvae (Cl), Chironomidae pupae (Cp), Simulidae (S), Oligochaete (O), Bivalva (M), Pleocoptera (P), Ephmeroptera (E), Odonata (Od), Ceratopogonidae (Ce), Hemiptera (He), Hirudinea (Hi), Tricoptera (T), Coleoptera (Co), Planorbidae (Pl), Cambaridae (Cr), fish eggs, algae, and gravel Species Index D Cl Cp S O M P E Od Ce He Hi T Co Pl Cr Eggs Algae Gravel HFCS F SA QBCK F SA RVCS F SA GORH F SA SHRH F SA SVRH F SA NHGS F SA
4 1162 J. R. Spiegel, M. C. Quist and J. E. Morris Table 4 MoristaÕs food overlap (C) between species combinations for catostomids sampled from four Iowa rivers, 2009 Species 1 Species 2 MoristaÕs C Highfin carpsucker Quillback 0.52 Golden redhorse 0.07 Northern hogsucker 0.06 River carpsucker 0.81 Shorthead redhorse 0.10 Silver redhorse 0.09 Shorthead redhorse Northern hogsucker 0.45 Golden redhorse 0.42 Silver redhorse 0.67 River carpsucker 0.42 Quillback 0.38 Golden redhorse Northern hogsucker 0.39 River carpsucker 0.27 Silver redhorse 0.44 Quillback 0.25 Quillback Northern hogsucker 0.22 River carpsucker 0.66 Silver redhorse 0.38 River carpsucker Silver redhorse 0.39 Northern hogsucker 0.27 Northern hogsucker Silver redhorse 0.48 Fig. 1. Graphs of feeding strategy, prey importance, and niche width contribution of invertebrate categories for catostomids sampled from four Iowa rivers, Prey items include Diptera (D), Chironomidae larvae (Cl), Chironomidae pupae (Cp), Simulidae (S), Oligochaete (O), Bivalva (M), Pleocoptera (P), Ephmeroptera (E), Odonata (Od), Ceratopogonidae (Ce), Hemiptera (He), Hirudinea (Hi), Tricoptera (T), Coleoptera (Co), Planorbidae (Pl), Cambaridae (Cr), fish eggs(eg), algae, and gravel consumed a substantial number and diversity of invertebrates, but they also consumed large amounts of algae, detritus, sand, and gravel. This was reflected in the food niche widths of Carpiodes spp., which were wider than the other catostomid species except golden redhorse. Previous studies on the food habits of Carpiodes spp. have varied in their conclusions. In Table 3 Niche width (LevinsÕ standardized index = B) for seven catostomids sampled from four Iowa rivers, 2009 Species B Highfin carpsucker 0.21 Quillback 0.53 River carpsucker 0.41 Golden redhorse 0.32 Shorthead redhorse 0.09 Northern hogsucker 0.19 higher rate for northern hogsucker, golden redhorse, and silver redhorse than for the other species. Although the relationship between gill raker spacing and standard length was dependent on species, northern hogsucker, golden redhorse, and silver redhorse typically had the widest spacing between gill rakers. Discussion This study describes similarities and differences in the trophic ecology of catostomids in IowaÕs rivers. In general, Carpiodes spp. had highly diverse diets compared to the other catostomids species. Individuals from all three species not only Fig. 2. Gill raker length (GRL) vs standard length (SL) for seven catostomids sampled from four Iowa rivers, 2009
5 Trophic ecology and gill raker morphology 1163 Fig. 3. Gill raker spacing (GRS) vs standard length (SL) for seven catostomids sampled from four Iowa rivers, 2009 the upper Missouri River, river carpsuckers fed primarily on zooplankton (Welker and Scarnecchia, 2003). In contrast, river carpsuckers from the lower Missouri River fed on a variety of prey items, including aquatic insects, oligochaetes, aquatic macrophytes, and mollusks (Brezner, 1958). In the current study, Carpiodes spp. typically had the longest and most closely-spaced gill rakers of the study species. Thus, the diversity of prey items consumed by Carpiodes spp. is likely related to the length and spacing of their gill rakers which allows them to efficiently retain a variety of animal and plant materials (Wright et al., 1983; Gillespie and Fox, 2003). In conjunction with the long, narrowly-spaced gill rakers, the gut morphology of Carpiodes spp. suggests that they are capable of consuming a diversity of prey items. Specifically, the intestine of Carpiodes spp. is different from many other catostomids in that they have a small muscular organ, termed the ÔgizzardÕ in Brezner (1958). The gizzard appears to function as a secondary grinding mechanism. The presence of a gizzard may also explain the large amount of sand and gravel that was often observed in Carpiodes spp. gut contents. In contrast to Carpiodes spp., golden redhorse, shorthead redhorse, silver redhorse, and northern hogsuckers had gill raker morphology that is likely better adapted to selectively prey on macroinvertebrates. Their gill rakers were more widely spaced and shorter than those of Carpiodes spp. Analyses of gut contents from Moxostoma spp. and northern hogsucker revealed that they rarely contained algae, detritus, silt, sand, or gravel. The increased space between the gill rakers of these species likely allows fine items to be flushed from the buccal cavity, while retaining invertebrates for consumption. For instance, Harlan et al. (1987) reported an observation where a northern hogsucker while feeding on invertebrates also ingested sand and gravel, which was then ejected prior to ingesting prey items. Meyer (1962) found diets of silver redhorse, shorthead redhorse, and golden redhorse to be dominated by insects in the Des Moines River, Iowa. The three most abundant prey taxa were Chironomidae, Ephemeroptera, and Trichoptera. Similarly, shorthead redhorse were reported to primarily consume dipterans, trichopterans, and ephemeropterans in the Mississippi River (Bur, 1976). Northern hogsuckers diets were dominated by ephemeropterans and trichopterans in a Missouri stream (Matheney and Rabeni, 1995). Our study showed similar results where chironomids, ephemeropterans, and trichopterans were generally the most frequently encountered and abundant taxa in the diets of Moxostoma spp. and northern hogsuckers. Although some differences in diet among catostomids in Iowa rivers were observed, diets could be considered relatively similar among taxa. A likely explanation for the high degree of overlap is that food resources are not limited in IowaÕs river systems. When food resources are abundant, coexisting fish species often exhibit high diet overlap (Bettoli et al., 1991). In contrast, when food resources become limiting, species shift to less favored food items and overlap decreases (Werner and Hall, 1976; Schoener, 1982; Bettoli et al., 1991). Werner and Hall (1976) showed that morphological and behavioral differences among bluegill Lepomis macrochirus, pumpkinseed L. gibbosus, and green sunfish L. cyanellus allowed for coexistence. If only one of the three species inhabited a system, a majority of their prey items was strongly associated with aquatic vegetation. However, when all three species were sympatric, each species consumed prey associated with vegetation, but their unique morphological characteristics (e.g. gill rakers) enabled each species to exploit unique resources. This study provides important information on the trophic ecology of catostomids in rivers of a highly-disturbed landscape. Despite having similar external morphology, differences in gill raker morphology and an ability to use a variety of food resources (e.g. aquatic insects, mollusks, algae, zooplankton) appear to allow a variety of catostomids to coexist in larger river systems. Additional research is needed on the trophic ecology and morphology of catostomids that are declining (e.g. black redhorse) to better understand their habitat requirements and the potential influence of species interactions. In addition, future work should examine growth and diets of catostomids in different assemblages to help elucidate the influence of resource partitioning on population dynamics and fish assemblage structure. Acknowledgements We thank L. Brown, N. Hogberg and M. Mork for their assistance in the field and laboratory. This project was supported, in part, by Iowa State University Department of Natural Resource Ecology and Management, the Iowa Department of Natural Resources (IDNR) through Grant No HA-18, and the Idaho Cooperative Fish and Wildlife Research Unit. The Unit is jointly sponsored by the University of Idaho, U.S. Geological Survey, Idaho Department of Fish and Game, and Wildlife Management Institute. The use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government or IDNR.
6 1164 J. R. Spiegel, M. C. Quist and J. E. Morris References Amundsen, P. A.; Gabler, H. M.; Staldvik, F. J., 1996: A new approach to graphical analysis of feeding strategy from stomach contents data)modification of the Costello (1990) method. J. Fish Biol. 48, Angradi, T. R.; Griffith, J. S., 1990: Diel feeding chronology and the diet selection of rainbow trout (Oncorhynchus mykiss) in the HenryÕs Fork of the Snake River, Idaho. Can. J. Fish. Aquat. Sci. 47, Barton, M., 2007: BondÕs biology of fishes, 3rd edn. Thompson Brooks Cole, Belmont, CA. Bertrand, K. N.; Gido, K. B., 2007: Effects of the herbivorous minnow, southern redbelly dace (Phoxinus erythrogaster), on stream productivity and ecosystem structure. Oecologia 151, Bettoli, P. W.; Morris, J. E.; Noble, R. L., 1991: Changes in the abundance of two atherinid species after aquatic vegetation removal. Trans. Am. Fish. Soc. 120, Bowen, S. H., 1996: Quantitative description of diet. In: Fisheries techniques, 2nd edn. B. R. Murphy and D. W. Willis (Eds). American Fisheries Society, Bethesda, MD, pp Brezner, J., 1958: Food habitats of the northern river carpsucker in Missouri. Progr. Fish-Cult. 20, Bur, M. T., 1976: Age, growth and food habits of Catostomidae in Pool 8 of the upper Mississippi River. M. Sc. Thesis, University of Wisconsin, La Crosse, WI. Burkhardt, R. W.; Gutreuter, S., 1995: Improving electrofishing consistency by standardizing power. N. Am. J. Fish. Manage. 15, Chipps, S. R.; Garvey, J. E., 2007: Assessment of diets and feeding pattern. In: Analysis and interpretation of freshwater fisheries data. C. S. Guy and M. L. Brown (Eds). American Fisheries Society, Bethesda, MD, pp Cooke, S. J.; Bunt, C. M.; Hamilton, S. J.; Jennings, C. A.; Pearson, M. P.; Cooperman, M. S.; Markle, D. F., 2005: Threats, conservation strategies, and prognosis for suckers (Catostomidae) in North America: insights from regional case studies of a diverse family of non-game fishes. Biol. Conserv. 121, Cooper, C. M., 1987: Benthos in Bear Creek, Mississippi: effects of habitat variation and agricultural sediments. J. Freshw. Ecol. 4, Ehlinger, T. J.; Wilson, D. S., 1988: A complex foraging polymorphism in bluegill sunfish. Proc. Nat. Acad. Sci. 85, Gillespie, G. J.; Fox, M. G., 2003: Morphological and life-history differentiation between littoral and pelagic forms of pumpkinseed. J. Fish Biol. 62, Gray, E. V.; Boltz, J. M.; Kellogg, K. A.; Stauffer, J. R., Jr, 1997: Food resource partitioning by nine sympatric darter species. Trans. Am. Fish. Soc. 126, Harlan, J. R.; Speaker, E. B.; Mayhew, J., 1987: Iowa fish and fishing. Iowa Conservation Commission, Des Moines. Holopainen, A. L.; Huttunen, P., 1992: Effects of forest clearcutting and soil disturbance on the biology of small forest brooks. Hydrobiologia 244, Ingram, T.; Shurin, J. B., 2009: Trait-based assembly and phylogenetic structure in northeast Pacific rockfish assemblages. Ecology 90, Jelks, H. L.; Walsh, S. J.; Contreras-Balderas, S.; Diaz-Pardo, E.; Hendrickson, D. A.; Lyons, J.; Mandrak, N. E.; McCormick, F.; Nelson, J. S.; Platania, S. P.; Porter, B. A.; Renaud, C. B.; Schmitter-Soto, J. J.; Taylor, E. B.; Warren, M. L., Jr, 2008: Conservation status of imperiled North American freshwater and diadromous fishes. Fisheries 33(8), Karr, J. R.; Toth, L. A.; Dudley, D. R., 1985: Fish communities of midwestern rivers: a history of degradation. BioSci. 35, Litvan, M. E.; Stewart, T. W.; Pierce, C. L.; Larson, C. J., 2007: Effects of grade control structures on the macroinvertebrate assemblage of an agriculturally impacted stream. River Res. Appl. 24, Marshall, S.; Elliott, M., 1997: A comparison of univariate and multivariate numerical and graphical techniques for determining inter- and intraspecific feeding relationships in estuarine fish. J. Fish Biol. 51, Matheney, P. M.; Rabeni, C. F., 1995: Patterns of movement and habitat use by northern hogsuckers in an Ozark stream. Trans. Am. Fish. Soc. 124, McNeely, D. L., 1987: Niche relations within an Ozark stream cyprinid assemblage. Environ. Biol. Fishes 18, Meyer, W. H., 1962: Life history of three species of redhorse (Moxostoma) in the Des Moines River, Iowa. Trans. Am. Fish. Soc. 91, Miller, J. L.; Hamon, T.; Jones, T.; West, F., 2007: Kokanee exploit large prey in the absence of limnetic predators. In: Sockeye salmon evolution, ecology, and management. C. A. Woody (Ed.). American Fisheries Society, Bethesda, MD, pp Morgan, R. P., II; Rasin, J., Jr; Noe, L. A., 1983: Sediment effects on eggs and larvae of striped bass and white perch. Trans. Am. Fish. Soc. 112, Natural Resources Conservation Service (NRCS), 2000: Natural resources inventory Summary Report. U.S Department of Agriculture, Ames, Iowa. Neebling, T. E.; Quist, M. C., 2010: Fish assemblages in IowaÕs nonwadeable rivers: relationships with habitat and sampling methods. Fish. Manage. Ecol. 17, Nelson, G. J., 1967: Epibranchial organs in lower teleostean fishes. J. Zool. 153, OÕBrien, W. J.; Luecke, C., 1983: A new estimate of zooplankton retention by gill-rakers and its ecological significance. Trans. Am. Fish. Soc. 112, SAS Institute, 2008: The SAS system for windows, version 9.2, SAS Institute, Cary, NC. Schoener, T. W., 1982: The controversy over interspecific competition. Am. Sci. 70, Smith, E. P.; Zaret, T. M., 1982: Bias in estimating niche overlap. Ecology 63, Tanaka, H.; Aoki, I.; Ohshimo, S., 2006: Feeding habits and gill raker morphology of three planktivorous pelagic fish species off the coast of northern and western Kyushu in summer. J. Fish Biol. 68, Walser, C. A.; Bart, H. L., Jr, 1999: Influence of agriculture on in-stream habitat and fish community structure in Piedmont watersheds of the Chattahoochee River System. Ecol. Freshw. Fish. 8, Welker, T. L.; Scarnecchia, D. L., 2003: Differences in species composition and feeding ecology of catostomid fishes in two distinct segments of the Missouri River, North Dakota, U.S.A. Environ. Biol. Fishes 68, Werner, E. E.; Hall, D. J., 1976: Niche shifts in sunfishes: experimental evidence and significance. Science 191, Wood, C. C., 2007: Sockeye salmon ecotypes: origin, vulnerability to human impacts, and conservation value. In: Sockeye salmon evolution, ecology, and management. C. A. Woody (Ed.). American Fisheries Society, Bethesda, MD, pp Wright, D. I.; OÕBrien, W. J.; Luecke, C., 1983: A new estimate of zooplankton retention by gill rakers and its ecological significance. Trans. Am. Fish. Soc. 112, Zaret, T. M.; Rand, A. S., 1971: Competition in tropical stream fishes: support for the competitive exclusion principle. Ecology 52, Zohrer, J. J., 2006: Iowa comprehensive wildlife conservation plan. Iowa Department of Natural Resources, Des Moines. AuthorÕs address: Michael C. Quist, U.S. Geological Survey, Idaho Cooperative Fish and Wildlife Research Unit, Department of Fish and Wildlife Resources, Box , University of Idaho, Moscow, ID 83844, USA. mcquist@uidaho.edu
Summer food habits and gill raker morphology of seven Catostomid species in Iowa rivers
 Graduate Theses and Dissertations Graduate College 200 Summer food habits and gill raker morphology of seven Catostomid species in Iowa rivers Jason Spiegel Iowa State University Follow this and additional
Graduate Theses and Dissertations Graduate College 200 Summer food habits and gill raker morphology of seven Catostomid species in Iowa rivers Jason Spiegel Iowa State University Follow this and additional
POPULATION DEMOGRAPHICS OF CATOSTOMIDS IN LARGE RIVER ECOSYSTEMS: EFFECTS OF DISCHARGE AND TEMPERATURE ON RECRUITMENT DYNAMICS AND GROWTH
 RIVER RESEARCH AND APPLICATIONS River Res. Applic. 28: 1567 1586 (12) Published online 5 June 11 in Wiley Online Library (wileyonlinelibrary.com).1545 POPULATION DEMOGRAPHICS OF CATOSTOMIDS IN LARGE RIVER
RIVER RESEARCH AND APPLICATIONS River Res. Applic. 28: 1567 1586 (12) Published online 5 June 11 in Wiley Online Library (wileyonlinelibrary.com).1545 POPULATION DEMOGRAPHICS OF CATOSTOMIDS IN LARGE RIVER
Food Habits of Rainbow Trout Stocked in Argyle Lake, Illinois
 Transactions of the Illinois State Academy of Science (1993), Volume 86, 1 and 2, pp. 71-77 Food Habits of Rainbow Trout Stocked in Argyle Lake, Illinois Larry A. Jahn and Daniel J. Lendman Department
Transactions of the Illinois State Academy of Science (1993), Volume 86, 1 and 2, pp. 71-77 Food Habits of Rainbow Trout Stocked in Argyle Lake, Illinois Larry A. Jahn and Daniel J. Lendman Department
Investigating reproduction and abundance of bighead carp (Hypophthalmichthys nobilis) and silver carp (H. molitrix) in the Greenup pool, Ohio River
 Investigating reproduction and abundance of bighead carp (Hypophthalmichthys nobilis) and silver carp (H. molitrix) in the Greenup pool, Ohio River Nathan Fleshman Dr. Thomas Jones Photo: USGS Photo: USGS
Investigating reproduction and abundance of bighead carp (Hypophthalmichthys nobilis) and silver carp (H. molitrix) in the Greenup pool, Ohio River Nathan Fleshman Dr. Thomas Jones Photo: USGS Photo: USGS
I L L I N 0 PRODUCTION NOTE. University of Illinois at Urbana-Champaign Library Large-scale Digitization Project, 2007.
 I L L I N S UNIVERSITY OF ILLINOIS AT URBA-CHAMPAIGN PRODUCTION NOTE University of Illinois at Urbana-Champaign Library Large-scale Digitization Project, 7. Uf'HS cft, Natural History Survey Library Illinois
I L L I N S UNIVERSITY OF ILLINOIS AT URBA-CHAMPAIGN PRODUCTION NOTE University of Illinois at Urbana-Champaign Library Large-scale Digitization Project, 7. Uf'HS cft, Natural History Survey Library Illinois
Rat Cove and Brookwood Point littoral fish survey, 2002
 Rat Cove and Brookwood Point littoral fish survey, 2 Katie Wayman 1 INTRODUCTION During the summer of 2, efforts to monitor the Otsego Lake littoral fish community continued with trap netting at both Rat
Rat Cove and Brookwood Point littoral fish survey, 2 Katie Wayman 1 INTRODUCTION During the summer of 2, efforts to monitor the Otsego Lake littoral fish community continued with trap netting at both Rat
MISSISSIPPI MAKEOVER A Plan for Restoration, Just Around the Bend
 MISSISSIPPI MAKEOVER A Plan for Restoration, Just Around the Bend Interim Report Card 2013 Project coordinated by the Dakota County Soil and Water Conservation District and sponsored by the Minnesota Pollution
MISSISSIPPI MAKEOVER A Plan for Restoration, Just Around the Bend Interim Report Card 2013 Project coordinated by the Dakota County Soil and Water Conservation District and sponsored by the Minnesota Pollution
The Prevalence of Different Species of Fish in Four Different Habitats of Douglas Lake
 Lukas Bell-Dereske EEB 320 Professor Amy Schrank August 16, 2007 Abstract The Prevalence of Different Species of Fish in Four Different Habitats of Douglas Lake How fish abundance and diversity varies
Lukas Bell-Dereske EEB 320 Professor Amy Schrank August 16, 2007 Abstract The Prevalence of Different Species of Fish in Four Different Habitats of Douglas Lake How fish abundance and diversity varies
MARTINDALE POND Wayne County 2004 Fish Management Report. Christopher C. Long Assistant Fisheries Biologist
 MARTINDALE POND Wayne County 2004 Fish Management Report Christopher C. Long Assistant Fisheries Biologist Fisheries Section Indiana Department of Natural Resources Division of Fish and Wildlife I.G.C.
MARTINDALE POND Wayne County 2004 Fish Management Report Christopher C. Long Assistant Fisheries Biologist Fisheries Section Indiana Department of Natural Resources Division of Fish and Wildlife I.G.C.
Minnesota Department of Natural Resources Division of Fish and Wildlife Section of Fisheries. Stream Survey Report. Three Mile Creek 2011
 Minnesota Department of Natural Resources Division of Fish and Wildlife Section of Fisheries Stream Survey Report Three Mile Creek 2011 By Joseph D. Stewig Montrose Area Fisheries Office TABLE OF CONTENTS
Minnesota Department of Natural Resources Division of Fish and Wildlife Section of Fisheries Stream Survey Report Three Mile Creek 2011 By Joseph D. Stewig Montrose Area Fisheries Office TABLE OF CONTENTS
Protect Our Reefs Grant Interim Report (October 1, 2008 March 31, 2009) Principal investigators: Donald C. Behringer and Mark J.
 Investigating the role of the spotted spiny lobster (Panulirus guttatus) in the recovery of the long spined sea urchin (Diadema antillarum) on the coral reefs of the Florida Keys Protect Our Reefs Grant
Investigating the role of the spotted spiny lobster (Panulirus guttatus) in the recovery of the long spined sea urchin (Diadema antillarum) on the coral reefs of the Florida Keys Protect Our Reefs Grant
Evaluation of Newbury Weirs (Rock Riffles) for Improving Habitat Quality and Biotic Diversity in Illinois Streams.
 Evaluation of Newbury Weirs (Rock Riffles) for Improving Habitat Quality and Biotic Diversity in Illinois Streams. Report submitted to The Wildlife Preservation Fund Illinois Department of Natural Resources
Evaluation of Newbury Weirs (Rock Riffles) for Improving Habitat Quality and Biotic Diversity in Illinois Streams. Report submitted to The Wildlife Preservation Fund Illinois Department of Natural Resources
Arizona Game and Fish Department Region VI Fisheries Program
 Arizona Game and Fish Department Region VI Fisheries Program Saguaro Lake Fish Survey Report April 18 th 21 st, 2016 Author: Bryant Dickens Report Date: July 7, 2016 The Arizona Game and Fish Department
Arizona Game and Fish Department Region VI Fisheries Program Saguaro Lake Fish Survey Report April 18 th 21 st, 2016 Author: Bryant Dickens Report Date: July 7, 2016 The Arizona Game and Fish Department
The Uptake of Mercury and Relationship to Food Habits of Target Fish Species in the South River and South Fork Shenandoah River
 The Uptake of Mercury and Relationship to Food Habits of Target Fish Species in the South River and South Fork Shenandoah River G. Murphy, T. Newcomb, and D. Orth Department of Fisheries and Wildlife Sciences
The Uptake of Mercury and Relationship to Food Habits of Target Fish Species in the South River and South Fork Shenandoah River G. Murphy, T. Newcomb, and D. Orth Department of Fisheries and Wildlife Sciences
Onondaga Lake Fishery: 2011 Fact Sheet
 October, 2011 Onondaga County Department of Water Environment Protection SUNY College of Environmental Science and Forestry Joanne M. Mahoney, County Executive Tom Rhoads, P.E., Commissioner Onondaga Lake
October, 2011 Onondaga County Department of Water Environment Protection SUNY College of Environmental Science and Forestry Joanne M. Mahoney, County Executive Tom Rhoads, P.E., Commissioner Onondaga Lake
The relationship between the spatial distribution of common carp and their environmental DNA in a small lake
 The relationship between the spatial distribution of common carp and their environmental DNA in a small lake Jessica Eichmiller*, Przemyslaw Bajer, and Peter Sorensen Department of Fisheries, Wildlife,
The relationship between the spatial distribution of common carp and their environmental DNA in a small lake Jessica Eichmiller*, Przemyslaw Bajer, and Peter Sorensen Department of Fisheries, Wildlife,
Seasonal change in the gastric evacuation rate of rainbow trout feeding on natural prey
 Journal of Fish Biology (2007) 71, 1873 1878 doi:10.1111/j.1095-8649.2007.01647.x, available online at http://www.blackwell-synergy.com Seasonal change in the gastric evacuation rate of rainbow trout feeding
Journal of Fish Biology (2007) 71, 1873 1878 doi:10.1111/j.1095-8649.2007.01647.x, available online at http://www.blackwell-synergy.com Seasonal change in the gastric evacuation rate of rainbow trout feeding
MIDDLE FORK RESERVOIR Wayne County 2004 Fish Management Report. Christopher C. Long Assistant Fisheries Biologist
 MIDDLE FORK RESERVOIR Wayne County 2004 Fish Management Report Christopher C. Long Assistant Fisheries Biologist FISHERIES SECTION INDIANA DEPARTMENT OF NATURAL RESOURCES DIVISION OF FISH AND WILDLIFE
MIDDLE FORK RESERVOIR Wayne County 2004 Fish Management Report Christopher C. Long Assistant Fisheries Biologist FISHERIES SECTION INDIANA DEPARTMENT OF NATURAL RESOURCES DIVISION OF FISH AND WILDLIFE
FISHERIES BLUE MOUNTAINS ADAPTATION PARTNERSHIP
 FISHERIES A warming climate, by itself, substantially affects the hydrology of watersheds in the Blue Mountains. Among the key hydrologic changes projected under all scenarios for the 2040s and beyond
FISHERIES A warming climate, by itself, substantially affects the hydrology of watersheds in the Blue Mountains. Among the key hydrologic changes projected under all scenarios for the 2040s and beyond
Fish Community and Aquatic Ecosystem Responses to the Cessation of Eurasian Watermilfoil Chemical Treatment on Lake Ellwood, Wisconsin
 Fish Community and Aquatic Ecosystem Responses to the Cessation of Eurasian Watermilfoil Chemical Treatment on Lake Ellwood, Wisconsin Greg G. Sass Wisconsin Department of Natural Resources Fisheries and
Fish Community and Aquatic Ecosystem Responses to the Cessation of Eurasian Watermilfoil Chemical Treatment on Lake Ellwood, Wisconsin Greg G. Sass Wisconsin Department of Natural Resources Fisheries and
An Assessment of the Fish Community in Lake Acworth
 An Assessment of the Fish Community in Lake Acworth By John Damer Fisheries Biologist Georgia Department of Natural Resources Wildlife Resources Division Fisheries Section Calhoun, GA 30701 July 2008 Introduction
An Assessment of the Fish Community in Lake Acworth By John Damer Fisheries Biologist Georgia Department of Natural Resources Wildlife Resources Division Fisheries Section Calhoun, GA 30701 July 2008 Introduction
Trip Report: Eagle Creek, Arizona
 Trip Report: Eagle Creek, Arizona 15-17 June 2009 Participants: Abraham Karam, Brian Kesner, and Mike Childs Native Fish Lab Marsh & Associates, LLC 5016 South Ash Avenue Suite 108 Tempe, Arizona 85282
Trip Report: Eagle Creek, Arizona 15-17 June 2009 Participants: Abraham Karam, Brian Kesner, and Mike Childs Native Fish Lab Marsh & Associates, LLC 5016 South Ash Avenue Suite 108 Tempe, Arizona 85282
Managing Development and Chesapeake Bay s Estuarine Fish
 Managing Development and Chesapeake Bay s Estuarine Fish Habitat Image and or Graphic Fisheries James H. Uphoff Jr. and Margaret M. McGinty Maryland Fisheries Service Funded through USFWS Federal Aid Maryland
Managing Development and Chesapeake Bay s Estuarine Fish Habitat Image and or Graphic Fisheries James H. Uphoff Jr. and Margaret M. McGinty Maryland Fisheries Service Funded through USFWS Federal Aid Maryland
Resource Partitioning and Life History Patterns Among Salmonids in the Estuarine Habitat Mosaic
 Resource Partitioning and Life History Patterns Among Salmonids in the Estuarine Habitat Mosaic Bethany Craig, Chris Eaton, and Si Simenstad University of Washington May 26, 2010 c/o WA DOE Salmon in the
Resource Partitioning and Life History Patterns Among Salmonids in the Estuarine Habitat Mosaic Bethany Craig, Chris Eaton, and Si Simenstad University of Washington May 26, 2010 c/o WA DOE Salmon in the
Digital ESF. SUNY College of Environmental Science and Forestry. Devon Shaw. Joe Rubenstein. Shane Currey. Andrew Koch
 SUNY College of Environmental Science and Forestry Digital Commons @ ESF Cranberry Lake Biological Station Environmental and Forest Biology 2017 Session A, 2017 First Place: Changes in activity levels
SUNY College of Environmental Science and Forestry Digital Commons @ ESF Cranberry Lake Biological Station Environmental and Forest Biology 2017 Session A, 2017 First Place: Changes in activity levels
Cedar Lake Comprehensive Survey Report Steve Hogler and Steve Surendonk WDNR-Mishicot
 Cedar Lake- 2006 Comprehensive Survey Report Steve Hogler and Steve Surendonk WDNR-Mishicot ABSTRACT Cedar Lake is a 142 acre lake located in the southwest corner of Manitowoc County. It is a seepage lake
Cedar Lake- 2006 Comprehensive Survey Report Steve Hogler and Steve Surendonk WDNR-Mishicot ABSTRACT Cedar Lake is a 142 acre lake located in the southwest corner of Manitowoc County. It is a seepage lake
The Fish Fauna of Cranberry Bog, Town of Burlington, Otsego County, N.Y.
 80 The Fish Fauna of Cranberry Bog, Town of Burlington, Otsego County, N.Y. Steven M. Foster* ABSTRACT In June 1995 a baseline fish survey of Cranberry Bog was conducted in order to identify the species
80 The Fish Fauna of Cranberry Bog, Town of Burlington, Otsego County, N.Y. Steven M. Foster* ABSTRACT In June 1995 a baseline fish survey of Cranberry Bog was conducted in order to identify the species
Alberta Conservation Association 2017/18 Project Summary Report
 Alberta Conservation Association 2017/18 Project Summary Report Project Name: Abundance, Distribution, Spawning, and Thermal Habitat of Westslope Cutthroat Trout and Bull Trout in the Stimson Creek Watershed
Alberta Conservation Association 2017/18 Project Summary Report Project Name: Abundance, Distribution, Spawning, and Thermal Habitat of Westslope Cutthroat Trout and Bull Trout in the Stimson Creek Watershed
Managing Chesapeake Bay s Land Use, Fish Habitat, and Fisheries: Studies. Jim Uphoff & Margaret McGinty, Fisheries Service
 Managing Chesapeake Bay s Land Use, Fish Habitat, and Fisheries: Positive Image and or Negative Graphic Case Studies Jim Uphoff & Margaret McGinty, Fisheries Service Maryland Fisheries Service has been
Managing Chesapeake Bay s Land Use, Fish Habitat, and Fisheries: Positive Image and or Negative Graphic Case Studies Jim Uphoff & Margaret McGinty, Fisheries Service Maryland Fisheries Service has been
EEB 122b PRACTICE SECOND MIDTERM
 EEB 122b PRACTICE SECOND MIDTERM Page 1 1. You are interested in conducting an experiment with two competing species of plants. Below are the zero-growth isoclines for the two species. C D a) Draw the
EEB 122b PRACTICE SECOND MIDTERM Page 1 1. You are interested in conducting an experiment with two competing species of plants. Below are the zero-growth isoclines for the two species. C D a) Draw the
Arizona Game and Fish Department Region I Fisheries Program. Chevelon Canyon Lake Fish Survey Report Trip Report April 2015
 Arizona Game and Fish Department Region I Fisheries Program Chevelon Canyon Lake Fish Survey Report Trip Report April 2015 Authors: Sally Petre and Mike Lopez Report Date: June 25, 2015 The Arizona Game
Arizona Game and Fish Department Region I Fisheries Program Chevelon Canyon Lake Fish Survey Report Trip Report April 2015 Authors: Sally Petre and Mike Lopez Report Date: June 25, 2015 The Arizona Game
Quillback (Carpoides cyprinus)
 t. Joseph Assessment Appendix Quillback (Carpoides cyprinus) feeding - clear to turbid water - Michigan - sand, sandy gravel, sandy silt, or clay-silt substrate - medium- to low-gradient rivers and streams;
t. Joseph Assessment Appendix Quillback (Carpoides cyprinus) feeding - clear to turbid water - Michigan - sand, sandy gravel, sandy silt, or clay-silt substrate - medium- to low-gradient rivers and streams;
The Round Goby Botulism Connection. Renea A. Ruffing Graduate Research Assistant Penn State University
 The Round Goby Botulism Connection Renea A. Ruffing Graduate Research Assistant Penn State University Round gobies in Pennsylvania waters of Lake Erie First goby caught in Lake Erie was in 1993, in Grand
The Round Goby Botulism Connection Renea A. Ruffing Graduate Research Assistant Penn State University Round gobies in Pennsylvania waters of Lake Erie First goby caught in Lake Erie was in 1993, in Grand
Michigan Dept. of Natural Resources Status of the Fishery Resource Report Page 1. Weber Lake Cheboygan County, T34N, R3W, Sec.
 Michigan Dept. of Natural Resources 2012-143 Status of the Fishery Resource Report Page 1 Weber Lake Cheboygan County, T34N, R3W, Sec. 31 Neal Godby, Fisheries Biologist Environment Weber Lake is a 28.5-acre
Michigan Dept. of Natural Resources 2012-143 Status of the Fishery Resource Report Page 1 Weber Lake Cheboygan County, T34N, R3W, Sec. 31 Neal Godby, Fisheries Biologist Environment Weber Lake is a 28.5-acre
Winter Drawdown Issues of Concern
 Winter Drawdown Issues of Concern Wetland Connectivity Amphibians Fisheries Winter Drawdown Wetland Connectivity Wetland complex near Osprey Creek is not responsive to lake level Wetland at upper end of
Winter Drawdown Issues of Concern Wetland Connectivity Amphibians Fisheries Winter Drawdown Wetland Connectivity Wetland complex near Osprey Creek is not responsive to lake level Wetland at upper end of
Little Calumet River Rapid Response Fish Identification and Enumeration Branch Summary Report
 Little Calumet River Rapid Response Fish Identification and Enumeration Branch Summary Report Introduction A rotenone rapid response was completed on a 2.6-mile section of the Little Calumet River immediately
Little Calumet River Rapid Response Fish Identification and Enumeration Branch Summary Report Introduction A rotenone rapid response was completed on a 2.6-mile section of the Little Calumet River immediately
BENSON PARK POND FISH SPECIES
 BENSON PARK POND FISH SPECIES Bluegill (Lepomis macrochirus) From the Greek, lepomis means scaled gill cover and macrochirus means large hand, in reference to its body shape and size. Average adult size
BENSON PARK POND FISH SPECIES Bluegill (Lepomis macrochirus) From the Greek, lepomis means scaled gill cover and macrochirus means large hand, in reference to its body shape and size. Average adult size
Fall 2017: Problem Set 3 (DUE Oct 26; 50 points)
 ESS 445 Introduction to Fisheries Science and Management: Biology, Ecology, Management, and Conservation of North American Freshwater Fishes and Aquatic Ecosystems Fall 2017: Problem Set 3 (DUE Oct 26;
ESS 445 Introduction to Fisheries Science and Management: Biology, Ecology, Management, and Conservation of North American Freshwater Fishes and Aquatic Ecosystems Fall 2017: Problem Set 3 (DUE Oct 26;
Thunder Bay River Assessment Appendix. Appendix 2
 Appendix 2 Known past and present fish distributions in the River system. Distribution of fishes were compiled from Bailey et al. (2003) and from records located at the Michigan Department of Natural Resources
Appendix 2 Known past and present fish distributions in the River system. Distribution of fishes were compiled from Bailey et al. (2003) and from records located at the Michigan Department of Natural Resources
Fish monitoring requirements of new FERC licenses: are they adequate?
 Fish monitoring requirements of new FERC licenses: are they adequate? Joseph D. Kiernan, Peter B. Moyle and John G. Williams Center for Watershed Sciences University of California, Davis Trends from recently
Fish monitoring requirements of new FERC licenses: are they adequate? Joseph D. Kiernan, Peter B. Moyle and John G. Williams Center for Watershed Sciences University of California, Davis Trends from recently
The Uptake of Mercury and Relationship to Food Habits of Fish in the South River and South Fork Shenandoah River
 The Uptake of Mercury and Relationship to Food Habits of Fish in the South River and South Fork Shenandoah River G. W. Murphy, T. J. Newcomb, and D. J. Orth Dept. of Fisheries and Wildlife Sciences Virginia
The Uptake of Mercury and Relationship to Food Habits of Fish in the South River and South Fork Shenandoah River G. W. Murphy, T. J. Newcomb, and D. J. Orth Dept. of Fisheries and Wildlife Sciences Virginia
Zooplankton Migration Patterns at Scotton Landing: Behavioral Adaptations written by Lauren Zodl, University of Delaware
 Zooplankton Migration Patterns at Scotton Landing: Behavioral Adaptations written by Lauren Zodl, University of Delaware Summary: Zooplankton have evolved specific migration patterns that increase their
Zooplankton Migration Patterns at Scotton Landing: Behavioral Adaptations written by Lauren Zodl, University of Delaware Summary: Zooplankton have evolved specific migration patterns that increase their
LIFE CYCLE AND COMMUNITY STRUCTURE OF CADDISFLIES (INSECTA:TRICHOPTERA) IN THE NAVASOTA RIVER, TEXAS.
 LIFE CYCLE AND COMMUNITY STRUCTURE OF CADDISFLIES (INSECTA:TRICHOPTERA) IN THE NAVASOTA RIVER, TEXAS. An Undergraduate Research Scholars Thesis by SARAH PRUSKI Submitted to Honors and Undergraduate Research
LIFE CYCLE AND COMMUNITY STRUCTURE OF CADDISFLIES (INSECTA:TRICHOPTERA) IN THE NAVASOTA RIVER, TEXAS. An Undergraduate Research Scholars Thesis by SARAH PRUSKI Submitted to Honors and Undergraduate Research
DIET SEASONALITY AND OVERLAP OF CHRYSICHTHYS SPECIES ON LAKE KAINJI, NIGERIA
 Continental J. Fisheries and Aquatic Sciences 1:15-20, 2007. Wilolud Online Journals, 2007. DIET SEASONALITY AND OVERLAP OF CHRYSICHTHYS SPECIES ON LAKE KAINJI, NIGERIA 1 Yem, I.Y, 1 Ago, N.D and 2 Ugoala,
Continental J. Fisheries and Aquatic Sciences 1:15-20, 2007. Wilolud Online Journals, 2007. DIET SEASONALITY AND OVERLAP OF CHRYSICHTHYS SPECIES ON LAKE KAINJI, NIGERIA 1 Yem, I.Y, 1 Ago, N.D and 2 Ugoala,
Stream Insects of the Pacific Northwest
 Stream Insects of the Pacific Northwest Patrick Edwards 2008 Acknowledgments - Jeff Adams, Rick Hafele, Dr. Ian Waite and Dr. Robert W. Wisseman for sharing their knowledge of aquatic insects. Dr. Trygve
Stream Insects of the Pacific Northwest Patrick Edwards 2008 Acknowledgments - Jeff Adams, Rick Hafele, Dr. Ian Waite and Dr. Robert W. Wisseman for sharing their knowledge of aquatic insects. Dr. Trygve
Chinook salmon (photo by Roger Tabor)
 Stream Residence 1. The choice : emigration vs. residence 2. Food: Abundance, density, and variation 3. Territory and Dominance 4. Fish Responses: Distribution, growth, survival 5. Mortality Migration
Stream Residence 1. The choice : emigration vs. residence 2. Food: Abundance, density, and variation 3. Territory and Dominance 4. Fish Responses: Distribution, growth, survival 5. Mortality Migration
MINNESOTA DEPARTMENT OF NATURAL RESOURCES DIVISION OF FISHERIES AND WILDLIFE. Gamefish Assessment Report
 Minnesota DNR F-29-R(P) Area F312 February 1, 2013 MINNESOTA DEPARTMENT OF NATURAL RESOURCES DIVISION OF FISHERIES AND WILDLIFE Gamefish Assessment Report Spring Gamefish Assessment on the Mississippi
Minnesota DNR F-29-R(P) Area F312 February 1, 2013 MINNESOTA DEPARTMENT OF NATURAL RESOURCES DIVISION OF FISHERIES AND WILDLIFE Gamefish Assessment Report Spring Gamefish Assessment on the Mississippi
Finescale Dace. Appendix A: Fish. Chrosomus neogaeus. New Hampshire Wildlife Action Plan Appendix A Fish-60
 Finescale Dace Chrosomus neogaeus Federal Listing State Listing Global Rank State Rank Regional Status SC S3 Photo by John Lyons Justification (Reason for Concern in NH) Finescale dace are vulnerable to
Finescale Dace Chrosomus neogaeus Federal Listing State Listing Global Rank State Rank Regional Status SC S3 Photo by John Lyons Justification (Reason for Concern in NH) Finescale dace are vulnerable to
Fish Community. Fish Habitat, Streams and Rivers
 Clear, Ston(e)y and White Lake Plan Fish Community The lakes support a cool/warm water fishery including muskellunge, walleye, largemouth and smallmouth bass, yellow perch, pumpkinseed, rockbass, blue
Clear, Ston(e)y and White Lake Plan Fish Community The lakes support a cool/warm water fishery including muskellunge, walleye, largemouth and smallmouth bass, yellow perch, pumpkinseed, rockbass, blue
STUDY PERFORMANCE REPORT
 STUDY PERFORMANCE REPORT State: Michigan Project No.: F-53-R-14 Study No.: 466 Title: Fish Community status in Saginaw Bay, Lake Huron Period Covered: April 1, 1997 to March 31, 1998 Study Objective: To
STUDY PERFORMANCE REPORT State: Michigan Project No.: F-53-R-14 Study No.: 466 Title: Fish Community status in Saginaw Bay, Lake Huron Period Covered: April 1, 1997 to March 31, 1998 Study Objective: To
A COMPARISON OF MICROPLASTIC INGESTION BETWEEN FRESHWATER SUNFISH AND MARINE PINFISH
 A COMPARISON OF MICROPLASTIC INGESTION BETWEEN FRESHWATER SUNFISH AND MARINE PINFISH COLLEEN A. PETERS, PEYTON A. THOMAS, KAITLYN B. RIEPER AND SUSAN P. BRATTON*; Fiber BACKGROUND Sphere/microbead Fragment
A COMPARISON OF MICROPLASTIC INGESTION BETWEEN FRESHWATER SUNFISH AND MARINE PINFISH COLLEEN A. PETERS, PEYTON A. THOMAS, KAITLYN B. RIEPER AND SUSAN P. BRATTON*; Fiber BACKGROUND Sphere/microbead Fragment
JadEco, LLC PO BOX 445 Shannon, IL 61078
 Introduction: With the assistance of Lake Holiday staff and volunteers, we were able to conduct an AC electrofishing survey on May 8, 27. Water temperatures were 2.3 C (8.5 F) and water clarity was decent
Introduction: With the assistance of Lake Holiday staff and volunteers, we were able to conduct an AC electrofishing survey on May 8, 27. Water temperatures were 2.3 C (8.5 F) and water clarity was decent
CHANGES IN ZOOPLANKTON COMMUNITY OF THE ILLINOIS RIVER SINCE ESTABLISHMENT OF BIGHEAD AND SILVER CARP
 CHANGES IN ZOOPLANKTON COMMUNITY OF THE ILLINOIS RIVER SINCE ESTABLISHMENT OF BIGHEAD AND SILVER CARP Collin Hinz, Mark Fritts, Greg Sass, Andy Casper Illinois River Biological Station Illinois Natural
CHANGES IN ZOOPLANKTON COMMUNITY OF THE ILLINOIS RIVER SINCE ESTABLISHMENT OF BIGHEAD AND SILVER CARP Collin Hinz, Mark Fritts, Greg Sass, Andy Casper Illinois River Biological Station Illinois Natural
Michigan Department of Natural Resources Status of the Fishery Resource Report Page 1
 Michigan Department of Natural Resources 2008-59 Status of the Fishery Resource Report Page 1 Bowens Manistee County, (T24N R16W Sec. 11, 12, 13, 14, 15, 24, and 30). Last surveyed 2008 Mark A. Tonello
Michigan Department of Natural Resources 2008-59 Status of the Fishery Resource Report Page 1 Bowens Manistee County, (T24N R16W Sec. 11, 12, 13, 14, 15, 24, and 30). Last surveyed 2008 Mark A. Tonello
SUMMARY REPORT FOR LAKE ST. MALO FISHERIES ASSESSMENT. Prepared for the St. Malo and District Wildlife Association
 SUMMARY REPORT FOR LAKE ST. MALO FISHERIES ASSESSMENT Prepared for the St. Malo and District Wildlife Association Presented by August 25, 2014 Study dates thus far: Date Tasks February 16 Fishing derby;
SUMMARY REPORT FOR LAKE ST. MALO FISHERIES ASSESSMENT Prepared for the St. Malo and District Wildlife Association Presented by August 25, 2014 Study dates thus far: Date Tasks February 16 Fishing derby;
Moses Lake Fishery Restoration Project
 Moses Lake Fishery Restoration Project Factors Affecting the Recreational Fishery in Moses Lake Washington Annual Report 2002-2003 DOE/BP-00006320-4 November 2003 Field37: This Document should be cited
Moses Lake Fishery Restoration Project Factors Affecting the Recreational Fishery in Moses Lake Washington Annual Report 2002-2003 DOE/BP-00006320-4 November 2003 Field37: This Document should be cited
Michigan Department of Natural Resources Status of the Fishery Resource Report Page 1
 Michigan Department of Natural Resources 2007-33 Status of the Fishery Resource Report Page 1 Grand Sable Lake Alger County, T49N, R14W, Sec. Many Lake Superior watershed, last year surveyed 2004 James
Michigan Department of Natural Resources 2007-33 Status of the Fishery Resource Report Page 1 Grand Sable Lake Alger County, T49N, R14W, Sec. Many Lake Superior watershed, last year surveyed 2004 James
RECREATIONAL PONDS AND LAKES
 RECREATIONAL PONDS AND LAKES POND ECOLOGY AQUATIC PLANTS & FISH F.S. Conte Department of Animal Science University of California Davis Photos By Flickr AQUATIC PLANTS POND HEALTH Chemical Recycling Oxygen
RECREATIONAL PONDS AND LAKES POND ECOLOGY AQUATIC PLANTS & FISH F.S. Conte Department of Animal Science University of California Davis Photos By Flickr AQUATIC PLANTS POND HEALTH Chemical Recycling Oxygen
Analysis of Fish Harvested During the 2017 Chisago Lakes Lions Club Carp Festival June 3th, 2017
 Analysis of Fish Harvested During the 2017 Chisago Lakes Lions Club Carp Festival June 3th, 2017 Prepared by: Dr. Joshua Lallaman Saint Mary s University of Minnesota Project Background Common carp Cyprinus
Analysis of Fish Harvested During the 2017 Chisago Lakes Lions Club Carp Festival June 3th, 2017 Prepared by: Dr. Joshua Lallaman Saint Mary s University of Minnesota Project Background Common carp Cyprinus
Proposed Changes to Bag and Size Limits Minnesota/Wisconsin Border Waters of the Mississippi River
 Proposed Changes to Bag and Size Limits Minnesota/Wisconsin Border Waters of the Mississippi River The Minnesota and Wisconsin DNRs are jointly reviewing fishing regulations on Pools 3-9 of the Mississippi
Proposed Changes to Bag and Size Limits Minnesota/Wisconsin Border Waters of the Mississippi River The Minnesota and Wisconsin DNRs are jointly reviewing fishing regulations on Pools 3-9 of the Mississippi
ELECTRO-FISHING REPORT 2016 UPPER TWEED
 ELECTRO-FISHING REPORT 2016 UPPER TWEED The electro-fishing programme carried out each summer by The Tweed Foundation is part of our management plan, which details the information that is required to manage
ELECTRO-FISHING REPORT 2016 UPPER TWEED The electro-fishing programme carried out each summer by The Tweed Foundation is part of our management plan, which details the information that is required to manage
Cold-transitional Small River
 Cold-transitional Small River A Brief Ecological Description of this Michigan River Type Cold-transitional Small River segments are defined (by the Michigan Department of Natural Resources, Fisheries Division)
Cold-transitional Small River A Brief Ecological Description of this Michigan River Type Cold-transitional Small River segments are defined (by the Michigan Department of Natural Resources, Fisheries Division)
Preakness Brook - FIBI098
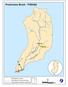 Preakness Brook - FIBI098 Preakn ess B rook Preakness " ( FIBI098 Naachtpunkt Brook 202 80 IBI Ratings FIBI Sampling Location Excellent Small Streams (1st and 2nd Order) Good Fair Large Streams (3rd Order
Preakness Brook - FIBI098 Preakn ess B rook Preakness " ( FIBI098 Naachtpunkt Brook 202 80 IBI Ratings FIBI Sampling Location Excellent Small Streams (1st and 2nd Order) Good Fair Large Streams (3rd Order
POLYCULTURE OF LARGEMOUTH BASS (Micropterus salmoides) WITH BLUE TILAPIA (Oreochromis aurea): USING TILAPIA PROGENY AS FORAGE
 POLYCULTURE OF LARGEMOUTH BASS (Micropterus salmoides) WITH BLUE TILAPIA (Oreochromis aurea): USING TILAPIA PROGENY AS FORAGE William A. Wurts a, D. Allen Davis b, Edwin H. Robinson c a Cooperative Extension
POLYCULTURE OF LARGEMOUTH BASS (Micropterus salmoides) WITH BLUE TILAPIA (Oreochromis aurea): USING TILAPIA PROGENY AS FORAGE William A. Wurts a, D. Allen Davis b, Edwin H. Robinson c a Cooperative Extension
Fisheries Survey of White Rapids Flowage, Marinette County Wisconsin during Waterbody Identification Code
 Fisheries Survey of White Rapids Flowage, Marinette County Wisconsin during 25 Waterbody Identification Code 6343 Michael Donofrio Fisheries Supervisor Wisconsin Department of Natural Resources Peshtigo,
Fisheries Survey of White Rapids Flowage, Marinette County Wisconsin during 25 Waterbody Identification Code 6343 Michael Donofrio Fisheries Supervisor Wisconsin Department of Natural Resources Peshtigo,
Executive Summary. Map 1. The Santa Clara River watershed with topography.
 Santa Clara River Steelhead Trout: Assessment and Recovery Opportunities December 2005 Prepared By: Matt Stoecker and Elise Kelley, Ph.D. University of California, Santa Barbara Prepared For: The Santa
Santa Clara River Steelhead Trout: Assessment and Recovery Opportunities December 2005 Prepared By: Matt Stoecker and Elise Kelley, Ph.D. University of California, Santa Barbara Prepared For: The Santa
Ecological interactions between forage fish, rorquals, and fisheries in Haida Gwaii
 Ecological interactions between forage fish, rorquals, and fisheries in Haida Gwaii Szymon Surma MSc candidate UBC Fisheries Centre The problem rorquals: lunge-feeding baleen whales eat large quantities
Ecological interactions between forage fish, rorquals, and fisheries in Haida Gwaii Szymon Surma MSc candidate UBC Fisheries Centre The problem rorquals: lunge-feeding baleen whales eat large quantities
Relative Weight of Brown Trout and Lake Trout in Blue Mesa Reservoir, Colorado
 Relative Weight of Brown Trout and Lake Trout in Blue Mesa Reservoir, Colorado Jessica Abbott U.S. Air Force Academy 2354 Fairchild Drive, USAF Academy, CO 80840, USA Kerri Schmidt U.S. Air Force Academy
Relative Weight of Brown Trout and Lake Trout in Blue Mesa Reservoir, Colorado Jessica Abbott U.S. Air Force Academy 2354 Fairchild Drive, USAF Academy, CO 80840, USA Kerri Schmidt U.S. Air Force Academy
Management of Small Impoundments
 Management of Small Impoundments Chapter 12 (Lake food webs) Chapter 15 (Natural Lakes) Chapter 16 (Farm Ponds & Small Impoundments) Quote for today: "The gods do not deduct from man's allotted span the
Management of Small Impoundments Chapter 12 (Lake food webs) Chapter 15 (Natural Lakes) Chapter 16 (Farm Ponds & Small Impoundments) Quote for today: "The gods do not deduct from man's allotted span the
CORRESPONDENCE/MEMORANDUM
 CORRESPONDENCE/MEMORANDUM State of Wisconsin DATE: November 13, 2012 FILE REF: [Click here and type file ref.] TO: FROM: Mike Donofrio Harpt Lake File Steve Hogler SUBJECT: 2012 Harpt Lake Electrofishing
CORRESPONDENCE/MEMORANDUM State of Wisconsin DATE: November 13, 2012 FILE REF: [Click here and type file ref.] TO: FROM: Mike Donofrio Harpt Lake File Steve Hogler SUBJECT: 2012 Harpt Lake Electrofishing
Fish Survey Report and Stocking Advice for Loch Milton. (Loch a Mhuilinn), May 2011
 Fish Survey Report and Stocking Advice for Loch Milton (Loch a Mhuilinn), May 2011 Jonah Tosney Freshwater Fisheries Biologist Wester Ross Fisheries Trust 1 Loch Milton (Loch a Mhuilinn) Fish Survey Report
Fish Survey Report and Stocking Advice for Loch Milton (Loch a Mhuilinn), May 2011 Jonah Tosney Freshwater Fisheries Biologist Wester Ross Fisheries Trust 1 Loch Milton (Loch a Mhuilinn) Fish Survey Report
Forage indicators and consumption profiles for Chesapeake Bay fishes
 Forage indicators and consumption profiles for Chesapeake Bay fishes Andre Buchheister Ed Houde Carlos Lozano Presentation to Fisheries GIT Dec 14, 2015 Outline Background & Objectives Result Highlights
Forage indicators and consumption profiles for Chesapeake Bay fishes Andre Buchheister Ed Houde Carlos Lozano Presentation to Fisheries GIT Dec 14, 2015 Outline Background & Objectives Result Highlights
Fish Conservation and Management
 Fish Conservation and Management CONS 486 Managing biotic communities Gardening, poisoning, and shocking what would PETA think? Chapter 10 Ross Managing biotic communities Topics Organism additions Organism
Fish Conservation and Management CONS 486 Managing biotic communities Gardening, poisoning, and shocking what would PETA think? Chapter 10 Ross Managing biotic communities Topics Organism additions Organism
Minnesota Department of Natural Resources Division of Fish and Wildlife Section of Fisheries. Stream Survey Report. Cold Spring Creek.
 Minnesota F-29-R(P)-24 Area 315 Study 3 March 2015 Minnesota Department of Natural Resources Division of Fish and Wildlife Section of Fisheries Stream Survey Report Cold Spring Creek 2014 Mark Pelham Montrose
Minnesota F-29-R(P)-24 Area 315 Study 3 March 2015 Minnesota Department of Natural Resources Division of Fish and Wildlife Section of Fisheries Stream Survey Report Cold Spring Creek 2014 Mark Pelham Montrose
Catlow Valley Redband Trout
 Catlow Valley Redband Trout Existing Populations The Catlow Valley Redband Trout SMU is comprised of five populations (Table 1). Three exist in Home, Threemile, and Skull creeks, located on the east side
Catlow Valley Redband Trout Existing Populations The Catlow Valley Redband Trout SMU is comprised of five populations (Table 1). Three exist in Home, Threemile, and Skull creeks, located on the east side
Largemouth Bass Abundance and Aquatic Vegetation in Florida Lakes: An Alternative Interpretation
 J. Aquat. Plant Manage. 34: 43-47 Largemouth Bass Abundance and Aquatic Vegetation in Florida Lakes: An Alternative Interpretation MICHAEL J. MACEINA 1 INTRODUCTION Hoyer and Canfield (1996) examined relations
J. Aquat. Plant Manage. 34: 43-47 Largemouth Bass Abundance and Aquatic Vegetation in Florida Lakes: An Alternative Interpretation MICHAEL J. MACEINA 1 INTRODUCTION Hoyer and Canfield (1996) examined relations
THE DIET OF Lutra canadensis IN THE UPPER COLORADO RIVER SYSTEM
 THE DIET OF Lutra canadensis IN THE UPPER COLORADO RIVER SYSTEM Judith K. BERG 56 Pinnon Woods Drive, Sedona, AZ 86351-7917, USA. dberg@us.ibm.com Abstract: The North American river otter was reintroduced
THE DIET OF Lutra canadensis IN THE UPPER COLORADO RIVER SYSTEM Judith K. BERG 56 Pinnon Woods Drive, Sedona, AZ 86351-7917, USA. dberg@us.ibm.com Abstract: The North American river otter was reintroduced
FISH COMMUNITIES AND FISHERIES OF THE THOUSAND ISLANDS AND MIDDLE CORRIDOR
 St. Lawrence River Discussion Paper FISH COMMUNITIES AND FISHERIES OF THE THOUSAND ISLANDS AND MIDDLE CORRIDOR Issues Below is a synopsis of fish community issues and proposed management approaches. More
St. Lawrence River Discussion Paper FISH COMMUNITIES AND FISHERIES OF THE THOUSAND ISLANDS AND MIDDLE CORRIDOR Issues Below is a synopsis of fish community issues and proposed management approaches. More
Alcona Dam Pond Alcona County (T25N, R5E, Sections various) Surveyed June 6-12 and September 16, 2003
 Michigan Department of Natural Resources Status of the Fishery Resource Report No. 2004-6, Year 2004 Alcona Dam Pond Alcona County (T25N, R5E, Sections various) Surveyed June 6-12 and September 16, 2003
Michigan Department of Natural Resources Status of the Fishery Resource Report No. 2004-6, Year 2004 Alcona Dam Pond Alcona County (T25N, R5E, Sections various) Surveyed June 6-12 and September 16, 2003
Lower Dolores River Corridor Planning Meeting Jim White Colorado Division of Wildlife
 Lower Dolores River Corridor Planning Meeting Jim White Colorado Division of Wildlife Objectives Overview of cold and warm water fishes in the Dolores River including: Flow and Fish Management Distribution
Lower Dolores River Corridor Planning Meeting Jim White Colorado Division of Wildlife Objectives Overview of cold and warm water fishes in the Dolores River including: Flow and Fish Management Distribution
I Region I Area I DOW Number / County I DOW Lake Name I Acreage I
 LAKE MANAGEMENT PLAN 814 Bp I Region I Area I DOW Number / County I DOW Lake Name I Acreage I I I # I f 2 Duluth F213 690036 St Louis Salo DOW:149 Plan: 137 Long Range Goal: Manage for walleye with a gillnet
LAKE MANAGEMENT PLAN 814 Bp I Region I Area I DOW Number / County I DOW Lake Name I Acreage I I I # I f 2 Duluth F213 690036 St Louis Salo DOW:149 Plan: 137 Long Range Goal: Manage for walleye with a gillnet
Current Status and Management Recommendations for the Fishery in the Cloverleaf Chain of Lakes
 Current Status and Management Recommendations for the Fishery in the Cloverleaf Chain of Lakes Jason Breeggemann Senior Fisheries Biologist Wisconsin Department of Natural Resources Outline DNR fisheries
Current Status and Management Recommendations for the Fishery in the Cloverleaf Chain of Lakes Jason Breeggemann Senior Fisheries Biologist Wisconsin Department of Natural Resources Outline DNR fisheries
2014 Island Lake Survey June 13 th, 2014 Andrew Plauck District Fisheries Biologist Report Prepared 4 March 2015
 2014 Island Lake Survey June 13 th, 2014 Andrew Plauck District Fisheries Biologist Report Prepared 4 March 2015 BACKGROUND - A fish survey was requested due to a moderately severe winterkill in Island
2014 Island Lake Survey June 13 th, 2014 Andrew Plauck District Fisheries Biologist Report Prepared 4 March 2015 BACKGROUND - A fish survey was requested due to a moderately severe winterkill in Island
niche requirements, interspecific
 Invasive salmonids: niche requirements, interspecific interactions and empty niches Timo Muotka University of Oulu Finnish Environment Institute Kai Korsu University of Oulu Ari Huusko Finnish Game & Fisheries
Invasive salmonids: niche requirements, interspecific interactions and empty niches Timo Muotka University of Oulu Finnish Environment Institute Kai Korsu University of Oulu Ari Huusko Finnish Game & Fisheries
EcoLogic Memorandum. TO: Ben Brezell; EDR FROM: Mark Arrigo RE: Possible Impacts of Dredging Snooks Pond DATE: 6/4/07
 EcoLogic Memorandum TO: Ben Brezell; EDR FROM: Mark Arrigo RE: Possible Impacts of Dredging Snooks Pond DATE: 6/4/07 Background EDR P.C. (EDR) has been contracted by a landowner to assess the feasibility
EcoLogic Memorandum TO: Ben Brezell; EDR FROM: Mark Arrigo RE: Possible Impacts of Dredging Snooks Pond DATE: 6/4/07 Background EDR P.C. (EDR) has been contracted by a landowner to assess the feasibility
Cold-transitional Stream
 Cold-transitional Stream A Brief Ecological Description of this Michigan River Type Cold-transitional Stream segments are defined (by the Michigan Department of Natural Resources, Fisheries Division) as
Cold-transitional Stream A Brief Ecological Description of this Michigan River Type Cold-transitional Stream segments are defined (by the Michigan Department of Natural Resources, Fisheries Division) as
LAKE DIANE Hillsdale County (T8-9S, R3W, Sections 34, 3, 4) Surveyed May Jeffrey J. Braunscheidel
 Michigan Department of Natural Resources Status of the Fishery Resource Report No. 2003-4, 2003 LAKE DIANE Hillsdale County (T8-9S, R3W, Sections 34, 3, 4) Surveyed May 2001 Jeffrey J. Braunscheidel Environment
Michigan Department of Natural Resources Status of the Fishery Resource Report No. 2003-4, 2003 LAKE DIANE Hillsdale County (T8-9S, R3W, Sections 34, 3, 4) Surveyed May 2001 Jeffrey J. Braunscheidel Environment
Key words: community similarity; coral patch reef; Enewetak; reeffish; species diversity; Virgin Islands.
 Ecology, 61(5), 1980, pp. 1156-1168 1980 by the Ecological Society of America SIMILARITY AND DIVERSITY AMONG CORAL REEF FISH COMMUNITIES: A COMPARISON BETWEEN TROPICAL WESTERN ATLANTIC (VIRGIN ISLANDS)
Ecology, 61(5), 1980, pp. 1156-1168 1980 by the Ecological Society of America SIMILARITY AND DIVERSITY AMONG CORAL REEF FISH COMMUNITIES: A COMPARISON BETWEEN TROPICAL WESTERN ATLANTIC (VIRGIN ISLANDS)
Conewago Creek Initiative. Fish Survey Report for the Conewago Creek
 Conewago Creek Initiative Fish Survey Report for the Conewago Creek A compilation of historic fish survey data from 97, 973, 7, for three locations along the Conewago Creek Prepared by Kristen Kyler for
Conewago Creek Initiative Fish Survey Report for the Conewago Creek A compilation of historic fish survey data from 97, 973, 7, for three locations along the Conewago Creek Prepared by Kristen Kyler for
DRAFT TECHNICAL MEMORANDUM
 DRAFT TECHNICAL MEMORANDUM From: File No.: 213.001 Re: Beaux Arts Village Shoreline - 2016 Introduction The Washington Academy of Beaux Arts (WABA) completed shoreline habitat restoration at three locations
DRAFT TECHNICAL MEMORANDUM From: File No.: 213.001 Re: Beaux Arts Village Shoreline - 2016 Introduction The Washington Academy of Beaux Arts (WABA) completed shoreline habitat restoration at three locations
INLAND LAKE MANAGEMENT REPORT FY Spring 2008
 INLAND LAKE MANAGEMENT REPORT FY 2008 Spring 2008 Prepared by Robert O. Andress District Fisheries Biologist E. Daniel Catchings District Fisheries Supervisor Kevin W. Baswell District Biologist Aide Department
INLAND LAKE MANAGEMENT REPORT FY 2008 Spring 2008 Prepared by Robert O. Andress District Fisheries Biologist E. Daniel Catchings District Fisheries Supervisor Kevin W. Baswell District Biologist Aide Department
Introduction: JadEco, LLC PO BOX 445 Shannon, IL 61078
 Introduction: was contacted to collected data on the fishery for Lake Holiday. AC Electroshocking was conducted at 2 locations on September 28, 2015. Fish population data was collected for a total of 100
Introduction: was contacted to collected data on the fishery for Lake Holiday. AC Electroshocking was conducted at 2 locations on September 28, 2015. Fish population data was collected for a total of 100
Uptake of Mercury and Relationship to Food Habits of Selected Fish Species in the Shenandoah River Basin, Virginia
 Uptake of Mercury and Relationship to Food Habits of Selected Fish Species in the Shenandoah River Basin, Virginia G. W. Murphy Masters Candidate Department of Fisheries and Wildlife Sciences Virginia
Uptake of Mercury and Relationship to Food Habits of Selected Fish Species in the Shenandoah River Basin, Virginia G. W. Murphy Masters Candidate Department of Fisheries and Wildlife Sciences Virginia
Ecology of Columbia River redband trout (Oncorhynchus mykiss gairdneri) in high desert streams
 Ecology of Columbia River redband trout (Oncorhynchus mykiss gairdneri) in high desert streams Dry Creek October 2012 Chris A. Walser Ph.D., Professor of Biology Department of Biology and Environmental
Ecology of Columbia River redband trout (Oncorhynchus mykiss gairdneri) in high desert streams Dry Creek October 2012 Chris A. Walser Ph.D., Professor of Biology Department of Biology and Environmental
Pequannock River - FIBI077
 Pequannock River - FIBI077 Canistear Rsvr. Clint on Brook Clinton R svr. k R r. idge Rs v Echo Lake Oa 23 Pequann ock R iver FIBI077 Kinnelon 287 23 FIBI Sampling Location Small Streams (1st and 2nd Order)
Pequannock River - FIBI077 Canistear Rsvr. Clint on Brook Clinton R svr. k R r. idge Rs v Echo Lake Oa 23 Pequann ock R iver FIBI077 Kinnelon 287 23 FIBI Sampling Location Small Streams (1st and 2nd Order)
Lect 19 - Populations - Chapter 23. Different Levels of Ecological Organization. Populations
 Lect 19 - - Chapter 23 Different Levels of Ecological Organization Individuals Assemblage Community Ecosystem - all individuals of a particular species in a given area - but need to recognize genetic structure
Lect 19 - - Chapter 23 Different Levels of Ecological Organization Individuals Assemblage Community Ecosystem - all individuals of a particular species in a given area - but need to recognize genetic structure
Predation Cycle-5 parts
 Fish as Predators Predator-prey relations: Predators have to eat (if they don t eat, fitness = 0) Prey die if they are eaten (if they are eaten, fitness = 0) Predator-prey arms race can drive evolution
Fish as Predators Predator-prey relations: Predators have to eat (if they don t eat, fitness = 0) Prey die if they are eaten (if they are eaten, fitness = 0) Predator-prey arms race can drive evolution
NURSERY POND Fish Management Report. Jason C. Doll Assistant Fisheries Biologist
 NURSERY POND 2004 Fish Management Report Jason C. Doll Assistant Fisheries Biologist FISHERIES SECTION INDIANA DEPARTMENT OF NATURAL RESOURCES DIVISION OF FISH AND WILDLIFE I.G.C. South, Room W273 402
NURSERY POND 2004 Fish Management Report Jason C. Doll Assistant Fisheries Biologist FISHERIES SECTION INDIANA DEPARTMENT OF NATURAL RESOURCES DIVISION OF FISH AND WILDLIFE I.G.C. South, Room W273 402
ASSESSMENT OF BLACK CRAPPIE AND WHITE PERCH IN HIGHLAND LAKE, STODDARD-WASHINGTON, NH (2014) New Hampshire
 ASSESSMENT OF BLACK CRAPPIE AND WHITE PERCH IN HIGHLAND LAKE, STODDARD-WASHINGTON, NH (2014) STATE: GRANT: GRANT TITLE: JOB 9: New Hampshire F-50-R-31 Anadromous and Inland Fisheries Operational Management
ASSESSMENT OF BLACK CRAPPIE AND WHITE PERCH IN HIGHLAND LAKE, STODDARD-WASHINGTON, NH (2014) STATE: GRANT: GRANT TITLE: JOB 9: New Hampshire F-50-R-31 Anadromous and Inland Fisheries Operational Management
Predation Cycle-5 parts
 Fish as Predators Predator-prey relations: Predators have to eat (if they don t eat, fitness = 0) Prey die if they are eaten (if they are eaten, fitness = 0) Predator-prey arms race can drive evolution
Fish as Predators Predator-prey relations: Predators have to eat (if they don t eat, fitness = 0) Prey die if they are eaten (if they are eaten, fitness = 0) Predator-prey arms race can drive evolution
