Recruitment of gastrocnemius muscles during the swing phase of stepping following partial denervation of knee flexor muscles in the cat
|
|
|
- Bernadette Ruth Parsons
- 5 years ago
- Views:
Transcription
1 Exp Brain Res (2005) DOI /s RESEARCH ARTICLE A. Tachibana Æ D. A. McVea Æ J. M. Donelan K. G. Pearson Recruitment of gastrocnemius muscles during the swing phase of stepping following partial denervation of knee flexor muscles in the cat Received: 23 May 2005 / Accepted: 24 June 2005 Ó Springer-Verlag 2005 A. Tachibana Æ D. A. McVea Æ J. M. Donelan Æ K. G. Pearson (&) Department of Physiology, University of Alberta, Edmonton, AB, T6G 2H7 Canada keir.pearson@ualberta.ca Tel.: Fax: Present address: A. Tachibana Department of Physiology and Neuroscience, Kanagawa Dental College, Kanagawa, Japan Present address: J. M. Donelan School of Kinesiology, Simon Fraser University, Vancouver, Canada Abstract In walking cats, the biarticular medial and lateral gastrocnemius (MG LG) muscles act to produce extension and flexion torques at the ankle and knee, respectively, and they usually display only one burst of activity beginning just before ground contact and ending near the end of the stance phase. Currently, the MG LG muscles are considered to function primarily to control extension movements around the ankle joint during the stance phase. However, their flexion action at the knee means that they have the capacity to regulate rotations at the knee, but this role has not yet been clearly defined. Following partial denervation of the other muscles that normally act to flex the knee during swing, we observed that the MG LG muscles, but not the Soleus muscle (a pure ankle extensor), often generated strong bursts of activity during early swing. These bursts were enhanced following mechanical stimulation of the paw, and they were especially prominent when the leg trailed over an object. They were absent when the leg led over an object. During treadmill walking the swing-related bursts in MG and LG had little influence on ankle flexion at the beginning of swing, but they were associated with slowing of ankle flexion when the leg trailed over an object. We hypothesized that the recruitment of these bursts functions to partially compensate for the reduction in knee torque resulting from the denervation of other knee flexors. Consistent with this hypothesis was our finding that the magnitude of the swing-related activity in the MG LG muscles was linearly correlated to the extent of the knee flexion and to the peak angular velocity of knee flexion, and that the timing of the bursts was similar to that in the denervated muscles prior to denervation. Our findings suggest that an excitatory pathway exists from the flexor half-center of the central pattern-generating network to MG LG motoneurons, and that this pathway is strongly regulated by central and/or peripheral signals. Keywords Walking Æ Nerve injury Æ Muscle synergies Æ Motor patterns Æ Spinal cord Introduction The patterns of activity of many muscles in the hind legs of walking cats have been well characterized (Engberg and Lundberg 1969; Abraham and Loeb 1985; Rossignol 1996; Yakovenko et al. 2002). The bursts of activity in individual muscles can generally be assigned to a flexor or an extensor synergy, since most muscles exhibit clear bursts of activity associated with either the flexion (F phase) or one or more of the extension phases (E1, E2, and E3) of the step cycle (Engberg and Lundberg 1969). Of special interest is that the burst activity in some muscles can be either recruited to or removed from a synergy when the locomotor task changes. For example, an additional burst is recruited into the extensor synergy in the iliopsoas muscle (a hip flexor) when animals walk down steep slopes (Smith et al. 1998), and in the semitendinosus muscle (knee flexor, hip extensor) when animals increase their walking speed (Smith et al. 1993). Additionally, during fictive locomotion, the bursts of activity in the motoneurons innervating the biarticular muscles acting at the knee and hip (semitendinosus and rectus femoris) can be
2 completely switched from one synergy to another by tonic sensory input (Perret and Cabelguen 1980). These observations clearly demonstrate that the mechanisms establishing membership of flexor and extensor synergies are highly flexible, and they have suggested plausible schemes for the neuronal basis of this flexibility (Perret 1983; Smith et al. 1998). In a preliminary study aimed at examining functional deficits produced by partially denervating muscles that flex the knee in walking cats, we unexpectedly observed that the medial gastrocnemius (MG) and lateral gastrocnemius (LG) muscles (ankle extensors, knee flexors) often produced bursts of activity during late stance and early swing in addition to the bursts that are normally associated with the stance phase. This observation, together with the fact that the MG and LG muscles cross the knee joint, indicated that under some circumstances these muscles might contribute to the production of flexion movements at the knee during the swing phase. Furthermore, it also suggested that a pathway exists from the system generating flexor bursts during early swing onto the motoneurons innervating the MG and LG muscles. The only reported occurrence of bursts of activity in LG during the flexion (F) phase of swing is during the stumbling-corrective reaction produced by mechanical stimulation of the dorsum of the paw (Forssberg 1979; Wand et al. 1980; Buford and Smith 1993). One function of the swing-associated bursts in LG is to assist paw clearance of the perturbing stimulus by stopping or reversing flexion movements at the ankle (Wand et al. 1980; Buford and Smith 1993). Another function may be to act synergistically with other muscles, such as semitendinosus, to enhance knee flexion. Recently, the pathways underlying the stumbling-corrective reaction have been examined during fictive locomotion in decerebrate cats (Quevedo et al. 2005a, b). The primary objectives of the present investigation were to establish some of the conditions that lead to robust burst activity in the LG and MG muscles after partial denervation of knee flexors, and to assess whether these bursts contribute to producing knee flexion during early swing. We focused on two situations that we knew would normally result in enhanced knee flexion movements at the knee: (1) following a repeated sequence of mechanical stimuli to the paw (McVea et al. 2004) and (2) when the animals step over an object (Widajewicz et al. 1994). The rationale for this strategy was that if the bursts in MG and LG during the F phase were substituting functionally for the loss of other knee flexors, then the magnitude of the bursts should be significantly increased in these two situations. Furthermore, we expected to find a strong correlation between the magnitude of the F-related activity in MG and LG and the magnitude and speed of knee flexion. Thus, our main experimental approach was to examine quantitatively the relationships between the kinematics of knee movement and the intensity of activity in the MG and LG muscles during the F phase of swing in the two experimental situations mentioned above. Materials and methods Electromyographic (EMG) and video recordings were made from the muscles of a hind leg in three adult cats ( kg) while walking on a treadmill and stepping over an object on a stationary walkway. All experimental procedures were approved by the Health Sciences Animal Welfare Committee at the University of Alberta. Training Each animal was first trained daily for 1 2 weeks to walk consistently on a treadmill at a speed of about 0.5 m/s and across a stationary walkway. In the latter situation, a block (height 6.5 cm, width 3.5 cm) was sometimes placed in the center of the walkway to force the animal to flex their hind legs more than normal to avoid contacting the block. Surgical procedures Following the initial training, bipolar EMG electrodes (Cooner wire AS632) were implanted into the following muscles of the right hind leg under general anesthetic (isoflurane) and aseptic conditions: the hip flexor iliopsoas (IP) and the ankle extensors soleus (SOL), LG, and MG in all three animals, the knee flexor semitendinosus (ST) in two animals, and the ankle flexor tibialis anterior (TA) in one of the animals with ST implanted. The leads from the EMG electrodes were led under the skin to a multi-pin socket fixed with screws and dental acrylic to the animal s skull. While anesthetized, adhesive reflective markers (diameter 0.5 cm) were placed over the iliac crest, the hip, knee and ankle joints, and on the end of the paw and the fifth digit of the right hind leg. A drop of Krazy glue was placed on the adhesive surface of these markers to help prevent detachment. If lost, these markers were replaced prior to each recording session. A second surgical procedure was performed 10, 35, and 43 days after implantation of the EMG electrodes in the three animals, respectively, to partially denervate the knee flexor muscles. The timing of this second procedure varied because the animals were used to collect data for unrelated projects that took different times to complete. Again, under general anesthesia (isoflurane) and aseptic conditions, two of the three major branches of the hamstring nerve were transected in the right hind leg. These branches contain motor axons innervating two of the main muscles flexing the knee (semitendinosus, ST and posterior biceps, PB) and a muscle extending the hip (anterior biceps, AB). The AB nerve was included because it normally provides some innervation to ST and PB (English and Weeks 1987). Prior to cutting the nerves, EMG and video data were recorded when the animals walked on the treadmill
3 and across the walkway. These recordings were used to provide baseline information on the EMG patterns for the current investigation. Acquisition of EMG and video data While the animals were walking on the treadmill, EMG data were recorded on an eight-channel Vetter 4000A PCM recorder, and the stepping movements of the right hind leg were captured at the same time by video recording at 60 frames/s. One channel of the Vetter recorder was reserved for a signal for later use in synchronizing the EMGs with video data using the Peak Motus 8.2 motion analysis system (ViconPeak, Centennial, Co). The synchronizing signals were delivered at a rate of about one every 10 s when the animals were walking on the treadmill, and close to the times the right leg stepped over the object on the walkway. During some sequences of walking on the treadmill, we examined the aftereffects of mechanically stimulating the paw of the right leg on the patterns of activity in the ankle extensor muscles. This was done by manually positioning a horizontal bar attached to a long stick in front of the paw so that the paw contacted the bar approximately mid-way through the swing phase. Multiple stimuli applied to the paw in a naïve animal eventually led to a persistent increase in knee flexion following the termination of the stimuli. Once conditioned, a single stimulus led to an increase in flexion at the knee that lasted for many hours in intact animals (McVea et al. 2004), and from five to ten cycles following a single stimulus in animals with the knee flexors partially denervated. Data analysis After the EMG and video recordings were made, the two sets of data were synchronized off-line using the Peak Motus system. Files of synchronized EMG and video data were made for selected sequences of treadmill walking and for all the trials where the animals stepped over the object on the treadmill. The positions of the markers on the right leg were digitized and stored in files in the Peak system. Because the knee marker shifted significantly relative to the knee joint position, we calculated the knee joint position by triangulation from the hip and ankle joint positions and measurements of the thigh and shank lengths. This calculation was done using software in the Peak system. The Peak system also calculated joint angles and joint angular velocities, and stored these calculated parameters within the file for each trial. To display the EMG and kinematic data in the Peak files, we used custom-written software (Matlab, Mathworks, Natwick, MA) and plotted the data in formats of our choosing. Custom-written software was also used to determine the relationships between the magnitude of EMG activity and kinematic parameters. The magnitude of EMG activity in the LG and MG muscles was determined by calculating the mean of the full-wave rectified and filtered EMG (50 Hz low pass first-order one-way Butterworth filter) over a defined time interval. In the treadmill walking cats, this interval was from the beginning to the end of bursts in the LG and MG muscles associated with flexion (Fig. 3). When the animals stepped over the block on the walkway, the interval was the first half of the flexion-associated LG and MG bursts (Fig. 6). Results Treadmill walking after partial denervation of knee flexors As expected, the most obvious effect of partially denervating the knee flexor muscles was a marked reduction in the extent of knee flexion during the flexion (F) phase of the swing (Fig. 1). However, an unexpected observation was that strong bursts of activity often occurred in the LG muscle in all the three animals beginning just before the onset of swing and terminating in the latter half of the F phase. Bursts of activity also occurred at the same time in the MG muscle of all three animals, but in two animals the bursts in MG were relatively weak compared to the stance-related EMG activity. Figure 1c (filled sections) shows an example of these flexion-associated bursts in LG and MG in the animal in which both muscles were strongly activated (see Fig. 8 for an example from another animal in which the magnitude of the flexion-associated LG bursts was much larger than in the case of the MG muscle). We refer to these bursts as MG F and LG F bursts. These bursts were never accompanied by bursts of activity in SOL. Because the main focus of this study was on the occurrence and associated movements at the knee and ankle joints, we did not make a detailed comparison of joint movements before and after the partial denervation of the knee flexors. Nevertheless, during the treadmill walking, any changes in the movements at the hip and ankle appeared to be relatively small (compare the hip and ankle movements in Fig. 1a, b). The absence of an obvious influence on hip extension must be reconciled with the fact that the denervated muscles, especially AB, act to extend the hip. A possibility is that hip extension following denervation may rely more on the mechanical effects produced by propulsive forces of the other legs. The duration of the swing phase was also not noticeably influenced by the denervation in two animals, but it did increase in the third (Fig. 1) due to a prolongation of the duration of the first extension phase (defined by extension at the knee and ankle). We were unable to establish any specific condition that led to the occurrence of the MG F and LG F bursts when the animals were walking on the treadmill (and
4 Fig. 1 Electromyographic (EMG) activity in MG and LG muscles during the early swing in a treadmill walking cat following partial denervation of the knee flexors. Top four traces rectified and filtered EMGs from the MG, LG, SOL, and ST muscles. Bottom three traces plots of the corresponding movements at the hip, knee, and ankle joints. Treadmill speed 0.5 m/s. pc paw contact, po paw off. a A typical step cycle recorded before partial denervation of the knee flexors showing burst activity in ST and the absence of activity in the extensor muscles during the flexion (F) phase. b, c Step cycles following partial denervation of the knee flexors, one in which little activity occurred in MG and LG during the F phase (b) and another in which distinct bursts of activity where generated in MG and LG (filled regions) near the time of onset of the F phase (c). Note the absence of bursting in SOL during the F phase, the abolition of ST activity after denervation, the reduction in amplitude of knee flexion following denervation, and the increase in knee flexion associated with the MG F and LG F bursts. All recordings were from the same animal across the walkway without the object being present), but one possibility we did exclude was that dragging of the paw on the treadmill belt caused the bursts due to activation of cutaneous receptors on the dorsum of the paw. It was certainly the case that the paw often dragged along the treadmill belt during early swing, especially in the first few days following partial denervation of the knee flexors. However, we were unable to correlate the occurrence of the MG F and LG F bursts to paw drag, and on most occasions these bursts occurred without any noticeable paw drag. Most importantly, the onset of the bursts preceded the onset of the swing phase by 60 to 80 ms, and the ventral (plantar) side of the paw was always firmly in contact with the supporting surface and moving backwards relative to the hip at the time of onset of MG F and LG F bursts. In addition, it was clear from the analysis of the leg kinematics that the MG F and LG F bursts began before the onset of flexion at the hip, knee, and ankle joints, that is, prior to the time of onset of any forward movement of the paw, and there were no indications that the leg was held in an extended position prior to the initiation of flexion to cause movement of the treadmill belt across the stationary paw. A noteworthy observation was that the timing of the onset of the MG F and LG F bursts relative to the extensor-related activity in the ankle extensors corresponded closely to the time of onset activity of the main knee flexors prior to denervation. This can be seen by comparing the timing of the onset of ST burst relative to the movements at the hip, knee, and ankle in Fig. 1a (before denervation) to the timing of the onset of the MG F and LG F bursts in Fig. 1c (after denervation), and it is shown clearly in averages of the LG F bursts after denervation and the ST bursts before denervation (Fig. 2). Note in Fig. 2 that the time of the onset of the LG F bursts relative to bursts in other muscles (panels B and D) corresponds quite closely to the time of the onset of ST bursts relative to the same muscles (panels A and C). The main implication of the close correspondence of the normal ST bursts and the MG F and LG F bursts is that the MG F and LG F bursts may be recruited in an attempt to substitute for the loss of knee flexors. Another important point to note in Fig. 1 is that the occurrence of the MG F and LG F bursts was associated with a larger flexion movement at the knee (compare knee angle in Fig. 1b, c) without a large influence on
5 Fig. 2 The timing and duration of the LG F bursts (filled in LG traces) after partial denervation of the knee flexors (b, d) are similar to the timing and duration of the ST bursts (filled in ST traces) before denervation (a, c) relative to bursts in other muscles. a, b Same animal as that yielding data from Fig. 1. c, d Same animal as that yielding data for Fig. 5. Each trace is an average of ten steps referenced (vertical lines) to either the onset of the ST (a, c) or LG F (b, d) bursts. Treadmill speed 0.5 m/s movement at the ankle joint (see also Fig. 5 for absence of large effects on the ankle in another animal). This observation is consistent with the notion that the MG F and LG F bursts are recruited to substitute for the loss of other muscles flexing the knee. To assess whether the MG F and LG F bursts could be contributing to knee flexion, we examined the relationships between the magnitude of the bursts and the extent and velocity of knee flexion movements. We chose to do this analysis for the step cycles following a mechanical stimulus to the dorsum of paw because the MG F and LG F bursts were consistently evoked on every step for five to ten steps following the stimulus, and because there was a progressive decline in the amplitude of the MG F and LG F bursts. Figure 3 shows an example of the EMGs in MG, LG, and SOL during eight cycles following the stimulus, as well as the hip and knee angles, knee angular velocity, and toe height during the sequence. These data clearly show that the progressive decline in the magnitude of the MG F and LG F bursts was associated with declines in the extent of knee flexion, the magnitude of knee angular velocity, and the amplitude of toe height. The plots in Fig. 4 show the correlation between these kinematic parameters and the amplitude of the MG F and LG F bursts. Figure 3 also shows that the SOL muscle was never activated (asterisks) at the same time as the MG F and LG F bursts, despite the very large initial amplitudes of the MG F and LG F bursts. A surprising observation was that ankle extension at the end of stance was not noticeably influenced by the occurrence of the MG F and LG F bursts (compare Fig. 1b, c). We examined this more closely by averaging the ankle movements following the onset of the MG F and LG F bursts in one animal in which we had implanted the TA muscle (Fig. 5). This was a different animal from that yielding the data in Figs. 1 and 3, and one in which the LG F bursts were relatively large. These averages clearly showed that the augmented LG F bursts following paw stimulation had a negligible effect on ankle extension. Corresponding to the enhanced activity in LG was an earlier onset of activity in TA, and an increase in the period of coactivation of the LG and TA muscles. It is likely that this increase in coactivation of LG and TA (and possibly other ankle flexor muscles, such as extensor digitorum longus) is the reason for the small influence of the augmented LG F bursts on ankle extension. The most noticeable effect on the ankle movements in the steps following a mechanical stimulus of the paw was an increase in the magnitude of ankle flexion during the F phase (Fig. 5). Correspondingly there was a marked increase in the magnitude of the TA bursts following the termination of the LG F bursts (Fig. 5) that very likely was largely responsible for the enhanced ankle flexion. Recruitment of MG and LG activity during swing when stepping over an object To further investigate the conditions leading to the production of the MG F and LG F bursts after partial denervation of the knee flexors, we examined the activity in the ankle extensors when the animals stepped over an object (Fig. 6). There was a clear difference in the activity in MG and LG depending on whether the leg was trailing or leading relative to the contralateral leg.
6 Fig. 3 The magnitude and velocity of knee flexion and toe height above the treadmill during the swing phase are related to the magnitude of the MG F and LG F bursts. Top three records rectified and filtered EMGs from the MG, LG, and SOL muscles during a sequence of steps following a mechanical stimulus to the paw (vertical dashed line); bottom four records plots of hip angle, knee angle, knee angular velocity, and toe height. Vertical lines indicate the times of onset of the MG F bursts. Treadmill speed 0.5 m/s. Note that the progressive decline in the magnitude of the MG F and LG F bursts following the stimulus was associated with progressive declines in the magnitude of flexion at the knee (distance between short horizontal lines), the maximum knee angular velocity during flexion (short horizontal lines), and the peak toe height during the swing phase. The SOL muscle was not active during the MG F and LG F bursts (asterisks). By the end of this stepping sequence the EMGs and kinematics had returned to values close to those prior to the stimulus When trailing, large MG F and LG F bursts were generated commencing before the onset of swing (Fig. 6a), whereas there was usually no activation of the MG and LG muscles during the F phase when the leg led (Fig. 6b). Similar to our observations in treadmill walking animals, no activity occurred in SOL during the MG F and LG F bursts when the leg trailed (asterisk in Fig. 6a). In addition, the extent of knee flexion and maximum knee angular velocity were correlated with the magnitude of the MG F and LG F bursts (Fig. 7). In all the three animals, the maximum angular velocity of knee flexion was larger when the leg trailed over the object compared to when it led (9, 25 and 37% higher when trailing). This is consistent with the notion that the MG F and LG F bursts contributed to knee flexion, since the MG F and LG F bursts only occurred when the leg trailed. The wide range of the difference in the magnitude of the knee angular velocities of trailing vs leading steps in the three animals may be partially due to differences in the magnitude of the hip angular velocity. In the animal with the smallest difference in knee angular velocity (9%) the difference in maximum hip angular velocity was largest (always higher during leading step), and vice versa for the animal with the largest difference in knee angular velocity. A mechanical consequence of a large difference in the angular velocity of hip flexion would be to provide additional assistance for knee flexion, thus reducing the difference in the knee angular velocity. In all the animals, the onset of activity in the hip flexor IP commenced earlier and was larger in amplitude when the leg was leading compared to when the leg was trailing (Fig. 6). The earlier and larger activity in IP during leading steps was associated with a faster flexion of the hip, as illustrated in the plots of hip angular velocity in Fig. 6. The occurrence of the MG F and LG F bursts during trailing steps was also associated with an obvious slowing and delay in flexion of the ankle joint (Fig. 8). Functionally, this was appropriate because with the weakening of knee flexion it was necessary to keep the ankle extended during early swing on trailing steps to ensure that the toe cleared the object. On trailing
7 Fig. 4 Scatter plots of the relationships between the magnitude of the MG F and LG F bursts (left and right, respectively) and the extent of knee flexion (a), the maximum knee flexion angular velocity (b), and maximum toe height (c) during stepping following a mechanical stimulus to the paw. The line in each plot is the best fitting linear regression line. AU arbitrary units steps, the position of the paw at the onset of swing was close to the object (Fig. 6, top left), so a weak knee flexion combined with an immediate flexion at the ankle would have increased the likelihood of the foot contacting the object. The occurrence of the MG F and LG F bursts likely contributed to the slowing of ankle flexion because the ankle angular velocity during early swing was inversely related to the magnitude of the MG F and LG F bursts (Fig. 9). The association of the MG F and LG F bursts with the slowing of ankle flexion during early swing was distinctly different from our observations during treadmill walking, in which the enhancement of the MG F and LG F bursts had little or no influence on initial part of ankle flexion (Fig. 5). A possible reason for this difference is that when the leg trailed over the object there was a substantial reduction in the magnitude of activity of the main ankle flexor TA at the time of the occurrence of the MG F and LG F bursts compared to the magnitude of the TA bursts when the leg led over the object (Fig. 8). This observation indicates that when the leg trails over an object, TA motoneurons are inhibited by the system generating the MG F and LG F bursts, thus reducing the flexor torque at the ankle joint. A reduction in TA activity was not observed when the MG F and LG F bursts were enhanced following the mechanical stimulation of the paw (Fig. 5). Discussion The main finding of this investigation was that the biarticular MG and LG muscles (ankle extensors/knee flexors) can be briefly activated near the onset of the swing phase in walking cats following partial denervation of muscles flexing the knee. This resulted in two bursts of activity during walking: one commencing just before the onset of stance and associated with the E1, E2, and E3 phases of the step cycle, and the other commencing just before swing and lasting well into the F phase. We refer to the latter bursts in MG and LG as the MG F and LG F bursts. The occurrence of these bursts have not been reported in previous studies on the EMG patterns during unperturbed walking, although it has been known for some time that mechanical stimulation of the dorsum paw during the swing phase can evoke strong bursts of activity in the LG muscles (Forssberg 1979; Wand et al. 1980). Because cutaneous stimulation of the paw evokes responses qualitatively similar to those we found in this study, it is important to consider the possibility that the MG F and LG F bursts were reflexively produced by cutaneous stimulation of the paw. The most compelling observations demonstrating that this was not the case was that these bursts began about ms before the end of stance (Fig. 1c), and
8 Fig. 5 Enhancement of the LG F bursts by mechanical stimulation of the paw during treadmill walking has little effect on late ankle extension and early ankle flexion. Superimposed averages (n=8) of rectified and filtered EMG records in TA and LG (top two sets) and ankle movement (bottom set) before (thin traces) and after (thick traces) a mechanical stimulus to the paw. Note the increase in the duration of the coactivation of the LG and TA muscles following the stimulus. The periods of coactivation before and after the stimulus are indicated by the width of shaded area and the horizontal arrowed line, respectively. Averages were made relative to the onset of the LG F burst. See text for more details at the time of onset of the bursts the ventral (plantar) surface of the paw was firmly in contact with the supporting surface and moving backwards relative to the hip. Furthermore, the MG F and LG F bursts usually occurred in the absence of any noticeable paw drag that might have activated cutaneous receptors to evoke a stumbling-corrective response. Thus, we conclude that the MG F and LG F bursts are produced by a central network linked to the system generating motor output for the flexion component of the swing phase. The first issue we wish to consider is the function of the MG F and LG F bursts. Because the occurrence of the MG F and LG F bursts was a consequence of partial denervation of other muscles producing flexion torques at the knee, the most obvious possibility is that the MG F and LG F bursts are recruited to compensate for the loss of function of the denervated knee flexors. This possibility is strongly supported by the following observations. First, the timing of the MG F and LG F bursts corresponded very closely to the timing of bursts of activity in ST that normally commenced just before the onset of swing and terminated mid-way during the F phase (Figs. 1a, 2). Second, conditions that normally led to an increase in the magnitude of activity in ST prior to denervation, such as enhanced leg flexion following mechanical stimulation of the paw and when the leg trails over an object (unpublished observations and Drew et al. 2002), also led to an increase in the magnitude of the MG F and LG F bursts (Figs. 3, 8). It is also relevant that ST activity increases more when the leg trails than when it leads in normal animals (unpublished observations and Drew et al. 2002), which parallels our finding that the MG F and LG F bursts only occurred when the leg was trailing (Fig. 6a). The third observation supporting the notion that the MG F and LG F bursts function to assist in knee flexion was that the pure ankle extensor muscle SOL was never found to be activated with the MG F and LG F bursts (Figs. 1, 3, 6). This suggests that the MG and LG muscles participate in a specialized function that is not shared with SOL. The obvious possibility for this specialized function is producing a flexion torque at the knee, since the action of all three muscles at the ankle (extension) is similar. On the other hand, it is conceivable that the MG F and LG F bursts could function to control movements at the ankle joint without the participation of SOL during early swing. Indeed, our finding that the velocity of ankle flexion was inversely related to the magnitude of the MG F and LG F bursts when the leg trailed over an object (Fig. 9) indicates that the MG F and LG F bursts are involved in controlling ankle flexion in this situation. The slowing of ankle flexion is functionally appropriate because on trailing steps a rapid ankle flexion at the beginning of swing would increase the likelihood of the paw contacting the object because the swing begins from a paw position close to the object. The slowing of ankle flexion may also be assisted by a reduction in the magnitude of the TA bursts relative to their magnitude during leading steps (Fig. 8). On the other hand, the influence of the MG F and LG F bursts on extension movements at the ankle when the animals were walking on the treadmill was not nearly so obvious (Figs. 1, 5). This was initially surprising because the small inertia of the paw would seem to enable contractions of the MG and LG muscles to have a strong influence on ankle movements. However, recordings from the antagonist TA showed this muscle to be active at the same time as the MG F and LG F bursts and the extent of the overlap
9 Fig. 6 Generation of MG F and LG F bursts when the leg trails over an object (filled sections in a) but not when it leads (b). Top diagrams superimposed stick figures of the hind leg (60/s), top four traces rectified and filtered EMGs from MG, LG, SOL, and IP muscles, bottom four traces corresponding records of knee angle, knee angular velocity, hip angle, and hip angular velocity. No burst activity occurred in SOL at the same time as MG F and increased when the MG F and LG F bursts were enhanced (Fig. 5). This activity in the TA muscle, and perhaps other ankle flexors (such as extensor digitorum longus), would act to resist extensor torques produced by the MG F and LG F bursts. This resistance to extension at the ankle would enable a greater force to be generated in the MG and LG muscles due to a reduction in the velocity of shortening and hence produce a greater flexion torque at the knee. One caveat to these proposals for the role of the TA muscle to regulate ankle movements produced by the MG F and LG F bursts is that our observations on TA burst activity were made in a single animal. To fully explain the neuromuscular mechanisms underlying the modification of ankle movements in the different behavioral tasks requires a more extensive examination of activity of TA and other muscles producing torques at the ankle. One muscle that may be especially interesting to examine would be flexor digitorus longus (FDL). This muscle produces an extensor LG F bursts (asterisk in a), and the extent of knee flexion was larger when the leg trailed over the object. Note also that the IP bursts had a smaller magnitude and later onset time when the leg trailed over the object. The smaller and delayed IP bursts during trailing steps were associated with a longer and slower flexion movement at the hip (horizontal arrowed lines) torque at the ankle in addition to its main function of plantar flexing the toes (Lawrence and Nichols 1999). Of special relevance is that FDL is briefly activated close to the onset of the swing phase (O Donovan et al. 1982). The timing and duration of the bursts in FDL closely resemble the MG F and LG F bursts we observed. Therefore, it is conceivable that the interneuronal system generating the FDL bursts is recruited to produce the MG F and LG F bursts. If this is true, then significant changes may also occur in FDL bursts following denervation of the knee flexors. Another issue we wish to discuss is the mechanism that enables the generation of the MG F and LG F bursts following partial denervation of the knee flexors. The first point to consider is whether these bursts are produced by an immediate reorganization of the patterngenerating network by removal or changes of sensory signals (due to loss of afferent signals from the denervated muscles or modifications in the movement of the
10 Fig. 7 Scatter plots of the relationships between the magnitude of the MG F and LG F bursts (left and right, respectively) and the extent of knee flexion (a) and the maximum knee flexion angular velocity (b) When the hind leg trailed over an object. Each dot is a single trial, and the line in each plot is the best fitting linear regression line. AU arbitrary units Fig. 8 The occurrence of the MG F and LG F bursts when a leg trails over an object is associated with a slowing of ankle flexion. Top three trace rectified and filtered EMGs from MG, LG, and TA; bottom two traces corresponding records of ankle angle and ankle angular velocity. Note that the TA burst was smaller and longer when the leg trailed (a) than when the leg led over the object (b). The dotted line in the TA record in (a) shows the TA burst during the leading step for comparison with the TA burst (filled region) during a trailing step
11 Fig. 9 Ankle flexion when the leg trails over an object is slowed in proportion to the magnitude of the MG F and LG F bursts. Scatter plot showing the inverse relationship between the ankle flexion angular velocity and the amplitude of the LG F bursts. Each dot is a single trial, and the line in the plot is the best fitting linear regression line. AU arbitrary units leg, respectively), or whether the generation of the MG F and LG F bursts develops over time as a result of some sort of learning process. One indication that learning was not a major factor was that MG F and LG F bursts were observed on the first recording session 5 6 h following the denervation surgery. During the short recovery period the animals were confined to a smallanimal transport cage that prevented locomotion. However, all the animals did walk on the treadmill or walkway for short periods (a few seconds each time) while being prepared for each recording session, so there remains the possibility that the animals quickly learned to generate the MG F and LG F bursts during these short periods of walking prior to the first recording session. If this occurred, it would be in contrast to the much slower adaptations in the activity of the MG muscle following denervation or weakening of the LG, SOL, and plantaris muscles (Misiaszek and Pearson 1999; Pearson et al. 1999). At present, however, we favor the notion that the MG F and LG F bursts are produced by an immediate reorganization of the spinal pattern-generating network. The reorganization of the spinal pattern-generating network appears to require input from supraspinal regions because the MG F and LG F bursts have never been reported in walking decerebrate or spinal cats, or during fictive locomotion in these preparations. Nor do they occur in decerebrate walking cats in which the hamstring nerve has been cut (unpublished observations). A likely source of these supraspinal signals is the motor cortex. The activity of many neurons in the hind leg region of the motor cortex is modulated during walking, with maximal activity occurring during the F phase of the step cycle (Widajewicz et al. 1994; Drew et al. 2002). Moreover, the activity in these modulated neurons increases significantly when the contralateral hind leg steps over an object. Thus, it would be of some interest to know whether the activity of these neurons also increases following partial denervation of the contralateral knee flexors. If they do, then the next question is what could be the cause of the increased activity? This question also applies to any other descending pathway showing modified activity after partial denervation of knee flexors. Another fact of potential relevance to understanding the mechanism for the generation of the MG F and LG F bursts is that activation of the LG and MG muscles occurs without activity in SOL during paw shaking (Smith et al. 1980). Thus, we know that a spinal interneuronal network exists that can selectively activate LG and MG motoneurons, so it is reasonable to suppose that this same network is recruited to produce the MG F and LG F bursts following partial denervation of the knee flexors. If, in addition, supraspinal inputs are involved in generating the MG F and LG F bursts, as we suspect (see above), then we predict that some descending pathways must be directed onto the interneuronal system selectively activating the MG and LG motoneurons. It is also likely that this interneuronal system receives input from, or may be part of, the central pattern generating network that produces the basic locomotor rhythm, since the MG F and LG F bursts can be consistently linked with the onset of flexor activity during walking (Fig. 3). The findings of the present investigation add to the growing body of data that has revealed considerable flexibility in the functioning of the interneuronal network controlling burst generation in hind leg motoneurons. It is now quite clear that bursts can be generated in most sets of motoneurons in either the flexion phase, one or more of the extension phases, or both flexion and extension phases of the step cycle, depending on the situation. The expression of additional bursts and/or the switching of bursting activity from one phase to another, has been reported during task-modification during normal walking (Smith et al. 1993, 1998; Carlson-Kuhta et al. 1998), in response to removal or addition of sensory signals (Perret and Cabelguen 1980; Perret 1983; Bouyer and Rossignol 2003), and as a result of weakening of a set of muscles (this study). In no instance have the underlying mechanisms been determined. One proposal is that the basic rhythm generating network consists of mutually inhibiting half-centers (flexor and extensor) and that each half-center is connected to many different groups of motoneurons via pathways that can be open or closed by sensory and/or supraspinal signals (Perret 1983). Another is that the central pattern generating network consists of a set of unit burst generators each of which drives a set of motoneurons and that the coupling between these unit burst generators can be modified by sensory and/or supraspinal signals (Grillner 1981; Smith et al. 1998). Establishing whether either of these proposals is correct is a challenging task. At the very least it requires a much greater understanding of the functional organization of interneurons in the spinal cord, as well as knowledge of the activity patterns of descending interneurons in
12 situations in which burst patterns are modified. Encouragingly, the feasibility of gaining this information has been demonstrated in recent studies on activity patterns of an identified class of interneurons in the lumbar cord (Angel et al. 2005) and recordings from different regions of the brain stem and cortex in behaving cats (Widajewicz et al. 1994; Matsuyama and Drew 2000; Lavoie and Drew 2002). Acknowledgements This work is supported by grants from the Canadian Institutes for Health Research, Alberta Heritage Foundation for Medical Research, and the Natural Science and Engineering Council of Canada. References Abraham LD, Loeb GE (1985) The distal hindlimb musculature of the cat. Patterns of normal use. Exp Brain Res 58: Angel MJ, Jankowska E, McCrea DA (2005) Candidate interneurones mediating group I disynaptic EPSPs in extensor motoneurones during fictive locomotion in the cat. J Physiol 563: Bouyer LJD, Rossignol S (2003) Contribution of cutaneous inputs from the hindpaw to the control of locomotion: 2. Spinal cats. J Neurophysiol 90: Buford JA, Smith JL (1993) Adaptive control for backward quadruped walking. III. Stumbling corrective reactions and cutaneous reflex sensitivity. J Neurophysiol 70: Carlson-Kuhta P, Trank TV, Smith JL (1998) Forms of forward quadrupedal locomotion. II. A comparison of posture, hindlimb kinematics, and motor patterns for upslope and level walking. J Neurophysiol 79: Drew T, Jiang W, Widajewicz W (2002) Contribution of the motor cortex to the control of hindlimbs during locomotion in the cat. Brain Res Rev 40: Engberg I, Lundberg A (1969) An electromyographic analysis of muscular activity in the hindlimb of the cat during unrestrained locomotion. Acta Physiol Scand 75: English AW, Weeks OI (1987) An anatomical and functional analysis of cat biceps femoris and semitendinosus muscle. J Morphol 191: Forssberg H (1979) Stumbling corrective reaction: a phase dependent compensatory reaction during locomotion. J Neurophysiol 42: Grillner S (1981) Control of locomotion in bipeds, tetrapods and fish. In: Brooks VB (ed) Handbook of physiology, sect 1, vol 2. The nervous system, motor control. American Physiological Society, Bethesda, MD, pp Lavoie S, Drew T (2002) Discharge characteristics of neurons in the red nucleus during voluntary gait modifications: a comparison with motor cortex. J Neurophysiol 88: Lawrence JH, Nichols TR (1999) A three-dimensional biomechanical analysis of the cat ankle joint complex: II. Effects of ankle joint orientation on evoked isometric joint torque. J Appl Biomech 15: Matsuyama K, Drew T (2000) Vestibulospinal and reticulospinal activity during locomotion in the intact cat. I. Walking on a level surface. J Neurophysiol 84: McVea D, Tachibana A, Donelan JM, Hulliger M, Pearson KG (2004) Long-term adaptation to persistent environmental perturbations in the walking cat. Neurosci Soc Abstr 34:180.8 Misiaszek JE, Pearson KG (1999) Injecting botulinum toxin into ankle extensors mimics the effects of axotomy. Neurosci Soc Abstr 25:122 O Donovan MJ, Pinter MJ, Dum RP, Burke RE (1982) Actions of FDL and FHL muscles in intact cats: functional dissociation between anatomical synergists. J Neurophysiol 47: Pearson KG, Fouad K, Misiaszek JE (1999) Adaptive changes in motor activity associated with functional recovery following muscle denervation in walking cats. J Neurophysiol 82: Perret C (1983) Centrally generated pattern of motoneuron activity during locomotion in the cat. In: Roberts A, Roberts B (eds) Neural origin of rhythmic movements. Cambridge University Press, Cambridge, pp Perret C, Cabelguen JM (1980) Main characteristics of the hindlimb locomotor cycle in the decorticate cat with special reference to bifunctional muscles. Brain Res 187: Quevedo J, Stecina K, Gosgnach S, McCrea DA (2005a) The stumbling corrective reaction during fictive locomotion in the cat. J Neurophysiol 94(3): Quevedo J, Stecina K, McCrea DA (2005b) Intracellular analysis of reflex pathways underlying the stumbling corrective reaction during fictive locomotion in the cat. J Neurophysiol 94(3): Rossignol S (1996) Control of stereotypic limb movements. In: Rowell LB, Shepard JT (eds) Handbook of physiology, sect 12. Exercise: regulation and integration of multiple systems. American Physiological Society, Bethesda, MD, pp Smith JL, Betts B, Edgerton VR, Zernicke RF (1980) Rapid ankle extension during paw shakes: selective recruitment of fast ankle extensors. J Neurophysiol 43: Smith JL, Carlson-Kuhta P, Trank TV (1998) Forms of forward quadrupedal locomotion. III. A comparison of posture, hindlimb kinematics, and motor patterns for downslope and level walking. J Neurophysiol 79: Smith JL, Chung SH, Zernicke RF (1993) Gait-related motor patterns and hindlimb kinetics for the cat trot and gallop. Exp Brain Res 94: Wand P, Prochazka A, Sontag KH (1980) Neuromuscular responses to gait perturbations in freely moving cats. Exp Brain Res 38: Widajewicz W, Kably B, Drew T (1994) Motor cortical activity during voluntary gait modifications in the cat. II. Cells related to the hindlimbs. J Neurophysiol 72: Yakovenko S, Mushahwar V, Vanderhorst VGJM, Holstege G, Prochazka A (2002) Spatiotemporal activation of lumbosacral motoneurons in the locomotor step cycle. J Neurophysiol 87:
Transformation of nonfunctional spinal circuits into functional states after the loss of brain input
 Transformation of nonfunctional spinal circuits into functional states after the loss of brain input G. Courtine, Y. P. Gerasimenko, R. van den Brand, A. Yew, P. Musienko, H. Zhong, B. Song, Y. Ao, R.
Transformation of nonfunctional spinal circuits into functional states after the loss of brain input G. Courtine, Y. P. Gerasimenko, R. van den Brand, A. Yew, P. Musienko, H. Zhong, B. Song, Y. Ao, R.
Adaptive Locomotion Controller for a Quadruped Robot
 Adaptive Locomotion Controller for a Quadruped Robot Step 1: Model of sensory feedback in animals during locomotion Simon Ruffieux 07 1 Introduction Sensory feedback is an important component of locomotion
Adaptive Locomotion Controller for a Quadruped Robot Step 1: Model of sensory feedback in animals during locomotion Simon Ruffieux 07 1 Introduction Sensory feedback is an important component of locomotion
Computer simulation of stepping in the hind legs of the cat: an examination of mechanisms regulating the stance-to-swing transition
 1 Computer simulation of stepping in the hind legs of the cat: an examination of mechanisms regulating the stance-to-swing transition Örjan Ekeberg Department of Computer Science, Royal Institute of Technology,
1 Computer simulation of stepping in the hind legs of the cat: an examination of mechanisms regulating the stance-to-swing transition Örjan Ekeberg Department of Computer Science, Royal Institute of Technology,
Neurorehabil Neural Repair Oct 23. [Epub ahead of print]
![Neurorehabil Neural Repair Oct 23. [Epub ahead of print] Neurorehabil Neural Repair Oct 23. [Epub ahead of print]](/thumbs/86/94477916.jpg) APPENDICE Neurorehabil Neural Repair. 2009 Oct 23. [Epub ahead of print] Segmental Muscle Vibration Improves Walking in Chronic Stroke Patients With Foot Drop: A Randomized Controlled Trial. Paoloni M,
APPENDICE Neurorehabil Neural Repair. 2009 Oct 23. [Epub ahead of print] Segmental Muscle Vibration Improves Walking in Chronic Stroke Patients With Foot Drop: A Randomized Controlled Trial. Paoloni M,
Walking. Zachary Zeidler Graduate Program in Neuroscience University of Minnesota
 Walking Zachary Zeidler Graduate Program in Neuroscience University of Minnesota 1 Locomotion Gaits Walking Running Skipping Hopping Trotting 2 Notice the: - Rhythmic pattern of limb activation while walking
Walking Zachary Zeidler Graduate Program in Neuroscience University of Minnesota 1 Locomotion Gaits Walking Running Skipping Hopping Trotting 2 Notice the: - Rhythmic pattern of limb activation while walking
Supplementary Figure S1
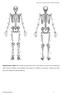 Supplementary Figure S1: Anterior and posterior views of the marker set used in the running gait trials. Forty-six markers were attached to the subject (15 markers on each leg, 4 markers on each arm, and
Supplementary Figure S1: Anterior and posterior views of the marker set used in the running gait trials. Forty-six markers were attached to the subject (15 markers on each leg, 4 markers on each arm, and
Sensory Gating for the Initiation of the Swing Phase in Different Directions of Human Infant Stepping
 The Journal of Neuroscience, July 1, 2002, 22(13):5734 5740 Sensory Gating for the Initiation of the Swing Phase in Different Directions of Human Infant Stepping Marco Y. C. Pang 1 and Jaynie F. Yang 1,2
The Journal of Neuroscience, July 1, 2002, 22(13):5734 5740 Sensory Gating for the Initiation of the Swing Phase in Different Directions of Human Infant Stepping Marco Y. C. Pang 1 and Jaynie F. Yang 1,2
ASSISTED AND RESISTED METHODS FOR SPEED DEVELOPMENT (PART 1)
 ASSISTED AND RESISTED METHODS FOR SPEED DEVELOPMENT (PART 1) By Adrian Faccioni Adrian Faccioni, a lecturer at the Centre of Sports Studies, University of Canberra, Australia, presents a detailed evaluation
ASSISTED AND RESISTED METHODS FOR SPEED DEVELOPMENT (PART 1) By Adrian Faccioni Adrian Faccioni, a lecturer at the Centre of Sports Studies, University of Canberra, Australia, presents a detailed evaluation
Coordination of medial gastrocnemius and soleus forces during cat locomotion
 The Journal of Experimental Biology 6, 6-6 The Company of Biologists Ltd doi:./jeb. 6 Coordination of medial gastrocnemius and soleus forces during cat locomotion Motoshi Kaya, Tim Leonard and Walter Herzog
The Journal of Experimental Biology 6, 6-6 The Company of Biologists Ltd doi:./jeb. 6 Coordination of medial gastrocnemius and soleus forces during cat locomotion Motoshi Kaya, Tim Leonard and Walter Herzog
Chapter 2 A Neuromechanical Model of Spinal Control of Locomotion
 Chapter 2 A Neuromechanical Model of Spinal Control of Locomotion Sergey N. Markin, Alexander N. Klishko, Natalia A. Shevtsova, Michel A. Lemay, Boris I. Prilutsky and Ilya A. Rybak Abstract We have developed
Chapter 2 A Neuromechanical Model of Spinal Control of Locomotion Sergey N. Markin, Alexander N. Klishko, Natalia A. Shevtsova, Michel A. Lemay, Boris I. Prilutsky and Ilya A. Rybak Abstract We have developed
Gait. Kinesiology RHS 341 Lecture 12 Dr. Einas Al-Eisa
 Gait Kinesiology RHS 341 Lecture 12 Dr. Einas Al-Eisa Definitions Locomotion = the act of moving from one place to the other Gait = the manner of walking Definitions Walking = a smooth, highly coordinated,
Gait Kinesiology RHS 341 Lecture 12 Dr. Einas Al-Eisa Definitions Locomotion = the act of moving from one place to the other Gait = the manner of walking Definitions Walking = a smooth, highly coordinated,
Spatiotemporal Activation of Lumbosacral Motoneurons in the Locomotor Step Cycle
 J Neurophysiol 87: 1542 1553, 2002; 10.1152/jn.00479.2001. Spatiotemporal Activation of Lumbosacral Motoneurons in the Locomotor Step Cycle SERGIY YAKOVENKO, 1 VIVIAN MUSHAHWAR, 1 VERONIQUE VANDERHORST,
J Neurophysiol 87: 1542 1553, 2002; 10.1152/jn.00479.2001. Spatiotemporal Activation of Lumbosacral Motoneurons in the Locomotor Step Cycle SERGIY YAKOVENKO, 1 VIVIAN MUSHAHWAR, 1 VERONIQUE VANDERHORST,
-Elastic strain energy (duty factor decreases at higher speeds). Higher forces act on feet. More tendon stretch. More energy stored in tendon.
 As velocity increases ( ) (i.e. increasing Froude number v 2 / gl) the component of the energy cost of transport associated with: -Internal kinetic energy (limbs accelerated to higher angular velocity).
As velocity increases ( ) (i.e. increasing Froude number v 2 / gl) the component of the energy cost of transport associated with: -Internal kinetic energy (limbs accelerated to higher angular velocity).
Positive running posture sums up the right technique for top speed
 Positive running, a model for high speed running Frans Bosch positive running posture sums up the right technique for top speed building blocks in running: Pelvic rotation for- and backward and hamstring
Positive running, a model for high speed running Frans Bosch positive running posture sums up the right technique for top speed building blocks in running: Pelvic rotation for- and backward and hamstring
Artifacts Due to Filtering Mismatch in Drop Landing Moment Data
 Camenga et al. UW-L Journal of Undergraduate Research XVI (213) Artifacts Due to Filtering Mismatch in Drop Landing Moment Data Elizabeth T. Camenga, Casey J. Rutten, Brendan D. Gould, Jillian T. Asmus,
Camenga et al. UW-L Journal of Undergraduate Research XVI (213) Artifacts Due to Filtering Mismatch in Drop Landing Moment Data Elizabeth T. Camenga, Casey J. Rutten, Brendan D. Gould, Jillian T. Asmus,
Dynamic Control of Location-Specific Information in Tactile Cutaneous Reflexes from the Foot during Human Walking
 The Journal of Neuroscience, May 15, 1997, 17(10):3804 3814 Dynamic Control of Location-Specific Information in Tactile Cutaneous Reflexes from the Foot during Human Walking Bart M. H. Van Wezel, Frans
The Journal of Neuroscience, May 15, 1997, 17(10):3804 3814 Dynamic Control of Location-Specific Information in Tactile Cutaneous Reflexes from the Foot during Human Walking Bart M. H. Van Wezel, Frans
Control principles for locomotion looking toward biology
 Control principles for locomotion looking toward biology Avis H. Cohen University of Maryland, Biology Department and Institute for Systems Research, College Park, MD 20742 Abstract Presented here is an
Control principles for locomotion looking toward biology Avis H. Cohen University of Maryland, Biology Department and Institute for Systems Research, College Park, MD 20742 Abstract Presented here is an
Today: the first activity system. Neural Control of Movement LOCOMOTION GAIT DESCRIPTIONS. Review: that amazing spinal cord! What we do: gait patterns
 Neural Control of Movement LOCOMOTION Today: the first activity system Review: that amazing spinal cord! What we do: gait patterns How we do it: neural circuitry and the role of sensory feedback and higher
Neural Control of Movement LOCOMOTION Today: the first activity system Review: that amazing spinal cord! What we do: gait patterns How we do it: neural circuitry and the role of sensory feedback and higher
Muscular Responses and Movement Strategies During Stumbling Over Obstacles
 Muscular Responses and Movement Strategies During Stumbling Over Obstacles A. M. SCHILLINGS, 1,2 B.M.H. VAN WEZEL, 1 TH. MULDER, 2,3 AND J. DUYSENS 1 1 Department of Medical Physics and Biophysics, University
Muscular Responses and Movement Strategies During Stumbling Over Obstacles A. M. SCHILLINGS, 1,2 B.M.H. VAN WEZEL, 1 TH. MULDER, 2,3 AND J. DUYSENS 1 1 Department of Medical Physics and Biophysics, University
The Journal of Physiology
 J Physiol 590.7 (2012) pp 1721 1736 1721 The upright posture improves plantar stepping and alters responses to serotonergic drugs in spinal rats Urszula Sławińska 1, Henryk Majczyński 1, Yue Dai 2 and
J Physiol 590.7 (2012) pp 1721 1736 1721 The upright posture improves plantar stepping and alters responses to serotonergic drugs in spinal rats Urszula Sławińska 1, Henryk Majczyński 1, Yue Dai 2 and
Supplementary Information
 Supplementary Information Novel robotic interface to evaluate, enable, and train locomotion and balance after neuromotor disorders Nadia Dominici, Urs Keller, Heike Vallery, Lucia Friedli, Rubia van den
Supplementary Information Novel robotic interface to evaluate, enable, and train locomotion and balance after neuromotor disorders Nadia Dominici, Urs Keller, Heike Vallery, Lucia Friedli, Rubia van den
Supplementary Materials for
 www.sciencetranslationalmedicine.org/cgi/content/full/6/255/255ra133/dc1 for Closed-loop neuromodulation of spinal sensorimotor circuits controls refined locomotion after complete spinal cord injury Nikolaus
www.sciencetranslationalmedicine.org/cgi/content/full/6/255/255ra133/dc1 for Closed-loop neuromodulation of spinal sensorimotor circuits controls refined locomotion after complete spinal cord injury Nikolaus
Assessments SIMPLY GAIT. Posture and Gait. Observing Posture and Gait. Postural Assessment. Postural Assessment 6/28/2016
 Assessments 2 SIMPLY GAIT Understanding movement Evaluations of factors that help therapist form professional judgments Include health, palpatory, range of motion, postural, and gait assessments Assessments
Assessments 2 SIMPLY GAIT Understanding movement Evaluations of factors that help therapist form professional judgments Include health, palpatory, range of motion, postural, and gait assessments Assessments
Parallel facilitatory reflex pathways from the foot and hip to flexors and extensors in the injured human spinal cord
 Experimental Neurology 206 (2007) 146 158 www.elsevier.com/locate/yexnr Parallel facilitatory reflex pathways from the foot and hip to flexors and extensors in the injured human spinal cord Maria Knikou
Experimental Neurology 206 (2007) 146 158 www.elsevier.com/locate/yexnr Parallel facilitatory reflex pathways from the foot and hip to flexors and extensors in the injured human spinal cord Maria Knikou
GROUND REACTION FORCE DOMINANT VERSUS NON-DOMINANT SINGLE LEG STEP OFF
 GROUND REACTION FORCE DOMINANT VERSUS NON-DOMINANT SINGLE LEG STEP OFF Sara Gharabaghli, Rebecca Krogstad, Sara Lynch, Sofia Saavedra, and Tamara Wright California State University, San Marcos, San Marcos,
GROUND REACTION FORCE DOMINANT VERSUS NON-DOMINANT SINGLE LEG STEP OFF Sara Gharabaghli, Rebecca Krogstad, Sara Lynch, Sofia Saavedra, and Tamara Wright California State University, San Marcos, San Marcos,
TECHNICAL CONSIDERATIONS FOR THE 100M HURDLES
 TECHNICAL CONSIDERATIONS FOR THE 100M HURDLES Thanks & Appreciation Vince Anderson, Texas A&M Andreas Behm, ALTIS Andy Eggerth, Kennesaw State University Erik Jenkins, University of Western Kentucky Glenn
TECHNICAL CONSIDERATIONS FOR THE 100M HURDLES Thanks & Appreciation Vince Anderson, Texas A&M Andreas Behm, ALTIS Andy Eggerth, Kennesaw State University Erik Jenkins, University of Western Kentucky Glenn
The Journal of Physiology Neuroscience
 J Physiol 595.1 (217) pp 341 361 341 The Journal of Physiology Neuroscience Left right coordination from simple to extreme conditions during split-belt locomotion in the chronic spinal adult cat Alain
J Physiol 595.1 (217) pp 341 361 341 The Journal of Physiology Neuroscience Left right coordination from simple to extreme conditions during split-belt locomotion in the chronic spinal adult cat Alain
A Pilot Study on Electromyographic Analysis of Single and Double Revolution Jumps in Figure Skating
 Journal of Exercise Science and Physiotherapy, Vol. 5, No. 1: 14-19, 2009 A Pilot Study on Electromyographic Analysis of Single and Double Revolution Jumps in Figure Skating Taylor¹, C. L. and Psycharakis²,
Journal of Exercise Science and Physiotherapy, Vol. 5, No. 1: 14-19, 2009 A Pilot Study on Electromyographic Analysis of Single and Double Revolution Jumps in Figure Skating Taylor¹, C. L. and Psycharakis²,
MOTOR ACTIVITY DURING SEARCHING AND WALKING MOVEMENTS OF COCKROACH LEGS
 J. exp. Biol. 133, 111-120 (1987) \\\ Printed in Great Britain The Company of Biologists Limited 1987 MOTOR ACTIVITY DURING SEARCHING AND WALKING MOVEMENTS OF COCKROACH LEGS BY FRED DELCOMYN Department
J. exp. Biol. 133, 111-120 (1987) \\\ Printed in Great Britain The Company of Biologists Limited 1987 MOTOR ACTIVITY DURING SEARCHING AND WALKING MOVEMENTS OF COCKROACH LEGS BY FRED DELCOMYN Department
Athlete Profiling. Injury Prevention
 Athlete Profiling Injury Prevention Fraser McKinney Physiotherapist Special interest in: Basketball Athletics Race Walking Research Performance markers (screening / HR assessments / biomechanics) Athlete
Athlete Profiling Injury Prevention Fraser McKinney Physiotherapist Special interest in: Basketball Athletics Race Walking Research Performance markers (screening / HR assessments / biomechanics) Athlete
Sensitivity of toe clearance to leg joint angles during extensive practice of obstacle crossing: Effects of vision and task goal
 Sensitivity of toe clearance to leg joint angles during extensive practice of obstacle crossing: Effects of vision and task goal Sérgio Tosi Rodrigues 1, Valéria Duarte Garcia 1,2, Arturo Forner- Cordero
Sensitivity of toe clearance to leg joint angles during extensive practice of obstacle crossing: Effects of vision and task goal Sérgio Tosi Rodrigues 1, Valéria Duarte Garcia 1,2, Arturo Forner- Cordero
Mechanics and energetics of level walking with powered ankle exoskeletons
 142 The Journal of Experimental Biology 211, 142-1413 Published by The Company of Biologists 28 doi:1.1242/jeb.9241 Mechanics and energetics of level walking with ed ankle exoskeletons Gregory S. Sawicki
142 The Journal of Experimental Biology 211, 142-1413 Published by The Company of Biologists 28 doi:1.1242/jeb.9241 Mechanics and energetics of level walking with ed ankle exoskeletons Gregory S. Sawicki
INTERACTION OF STEP LENGTH AND STEP RATE DURING SPRINT RUNNING
 INTERACTION OF STEP LENGTH AND STEP RATE DURING SPRINT RUNNING Joseph P. Hunter 1, Robert N. Marshall 1,, and Peter J. McNair 3 1 Department of Sport and Exercise Science, The University of Auckland, Auckland,
INTERACTION OF STEP LENGTH AND STEP RATE DURING SPRINT RUNNING Joseph P. Hunter 1, Robert N. Marshall 1,, and Peter J. McNair 3 1 Department of Sport and Exercise Science, The University of Auckland, Auckland,
Walking Simulator Mechanism
 The Downtown Review Volume 2 Issue 2 Article 4 2015 Walking Simulator Mechanism Titus Lungu Cleveland State University Igor Tachynskyy Cleveland State University Omri Tayyara Cleveland State University
The Downtown Review Volume 2 Issue 2 Article 4 2015 Walking Simulator Mechanism Titus Lungu Cleveland State University Igor Tachynskyy Cleveland State University Omri Tayyara Cleveland State University
Posture influences ground reaction force: implications for crouch gait
 University of Tennessee, Knoxville From the SelectedWorks of Jeffrey A. Reinbolt July 14, 2010 Posture influences ground reaction force: implications for crouch gait H. X. Hoang Jeffrey A. Reinbolt, University
University of Tennessee, Knoxville From the SelectedWorks of Jeffrey A. Reinbolt July 14, 2010 Posture influences ground reaction force: implications for crouch gait H. X. Hoang Jeffrey A. Reinbolt, University
DIFFERENCE BETWEEN TAEKWONDO ROUNDHOUSE KICK EXECUTED BY THE FRONT AND BACK LEG - A BIOMECHANICAL STUDY
 268 Isas 2000! Hong Kong DIFFERENCE BETWEEN TAEKWONDO ROUNDHOUSE KICK EXECUTED BY THE FRONT AND BACK LEG - A BIOMECHANICAL STUDY Pui-Wah Kong, Tze-Chung Luk and Youlian Hong The Chinese University of Hong
268 Isas 2000! Hong Kong DIFFERENCE BETWEEN TAEKWONDO ROUNDHOUSE KICK EXECUTED BY THE FRONT AND BACK LEG - A BIOMECHANICAL STUDY Pui-Wah Kong, Tze-Chung Luk and Youlian Hong The Chinese University of Hong
RESEARCH ARTICLE Effect of slope and sciatic nerve injury on ankle muscle recruitment and hindlimb kinematics during walking in the rat
 7 The Journal of Experimental Biology 214, 7-16 211. Published by The Company of Biologists Ltd doi:.1242/jeb.5158 RESEARCH ARTICLE Effect of slope and sciatic nerve injury on ankle muscle recruitment
7 The Journal of Experimental Biology 214, 7-16 211. Published by The Company of Biologists Ltd doi:.1242/jeb.5158 RESEARCH ARTICLE Effect of slope and sciatic nerve injury on ankle muscle recruitment
Regulation of Arm and Leg Movement during Human Locomotion E. PAUL ZEHR AND JACQUES DUYSENS
 REVIEW Regulation of Arm and Leg Movement during Human Locomotion E. PAUL ZEHR AND JACQUES DUYSENS Walking can be a very automated process, and it is likely that central pattern generators (CPGs) play
REVIEW Regulation of Arm and Leg Movement during Human Locomotion E. PAUL ZEHR AND JACQUES DUYSENS Walking can be a very automated process, and it is likely that central pattern generators (CPGs) play
Transfer of Motor Performance in an Obstacle Avoidance Task to Different Walking Conditions
 J Neurophysiol 92: 2010 2016, 2004; 10.1152/jn.00397.2004. Transfer of Motor Performance in an Obstacle Avoidance Task to Different Walking Conditions Tania Lam and Volker Dietz Spinal Cord Injury Center,
J Neurophysiol 92: 2010 2016, 2004; 10.1152/jn.00397.2004. Transfer of Motor Performance in an Obstacle Avoidance Task to Different Walking Conditions Tania Lam and Volker Dietz Spinal Cord Injury Center,
Rehabilitation of Non-operative Hamstring Injuries
 Rehabilitation of Non-operative Hamstring Injuries 12 th Annual Colorado University Sports Medicine Fall Symposium Robert A. Panariello MS, PT, ATC, CSCS Founding Partner, Chief Clinical Officer Professional
Rehabilitation of Non-operative Hamstring Injuries 12 th Annual Colorado University Sports Medicine Fall Symposium Robert A. Panariello MS, PT, ATC, CSCS Founding Partner, Chief Clinical Officer Professional
video Purpose Pathological Gait Objectives: Primary, Secondary and Compensatory Gait Deviations in CP AACPDM IC #3 1
 s in CP Disclosure Information AACPDM 71st Annual Meeting September 13-16, 2017 Speaker Names: Sylvia Ounpuu, MSc and Kristan Pierz, MD Differentiating Between, Secondary and Compensatory Mechanisms in
s in CP Disclosure Information AACPDM 71st Annual Meeting September 13-16, 2017 Speaker Names: Sylvia Ounpuu, MSc and Kristan Pierz, MD Differentiating Between, Secondary and Compensatory Mechanisms in
Characteristics of ball impact on curve shot in soccer
 Available online at www.sciencedirect.com Procedia Engineering 60 (2013 ) 249 254 6 th Asia-Pacific Congress on Sports Technology (APCST) Characteristics of ball impact on curve shot in soccer Sungchan
Available online at www.sciencedirect.com Procedia Engineering 60 (2013 ) 249 254 6 th Asia-Pacific Congress on Sports Technology (APCST) Characteristics of ball impact on curve shot in soccer Sungchan
The Journal of Physiology
 J Physiol 595.17 (2017) pp 5987 06 5987 Lack of adaptation during prolonged split-belt locomotion in the intact and spinal cat Victoria Kuczynski, Alessandro Telonio, Yann Thibaudier, Marie-France Hurteau,
J Physiol 595.17 (2017) pp 5987 06 5987 Lack of adaptation during prolonged split-belt locomotion in the intact and spinal cat Victoria Kuczynski, Alessandro Telonio, Yann Thibaudier, Marie-France Hurteau,
Toward a Human-like Biped Robot with Compliant Legs
 Book Title Book Editors IOS Press, 2003 1 Toward a Human-like Biped Robot with Compliant Legs Fumiya Iida a,b,1, Yohei Minekawa a Juergen Rummel a and Andre Seyfarth a a Locomotion Laboratory, University
Book Title Book Editors IOS Press, 2003 1 Toward a Human-like Biped Robot with Compliant Legs Fumiya Iida a,b,1, Yohei Minekawa a Juergen Rummel a and Andre Seyfarth a a Locomotion Laboratory, University
An investigation of kinematic and kinetic variables for the description of prosthetic gait using the ENOCH system
 An investigation of kinematic and kinetic variables for the description of prosthetic gait using the ENOCH system K. OBERG and H. LANSHAMMAR* Amputee Training and Research Unit, University Hospital, Fack,
An investigation of kinematic and kinetic variables for the description of prosthetic gait using the ENOCH system K. OBERG and H. LANSHAMMAR* Amputee Training and Research Unit, University Hospital, Fack,
Supplementary materials
 Supplementary materials I. Pressure sensor calibration Our analysis is based on identification of the onset and offset of the inhalation relatively to the electrophysiological recordings. Onset and offset
Supplementary materials I. Pressure sensor calibration Our analysis is based on identification of the onset and offset of the inhalation relatively to the electrophysiological recordings. Onset and offset
Dynamically stepping over large obstacle utilizing PSO optimization in the B4LC system
 1 Dynamically stepping over large obstacle utilizing PSO optimization in the B4LC system QI LIU, JIE ZHAO, KARSTEN BERNS Robotics Research Lab, University of Kaiserslautern, Kaiserslautern, 67655, Germany
1 Dynamically stepping over large obstacle utilizing PSO optimization in the B4LC system QI LIU, JIE ZHAO, KARSTEN BERNS Robotics Research Lab, University of Kaiserslautern, Kaiserslautern, 67655, Germany
Sample Solution for Problem 1.a
 Sample Solution for Problem 1.a 1 Inverted Pendulum Model (IPM) 1.1 Equations of Motion and Ground Reaction Forces Figure 1: Scheme of the Inverted Pendulum Model (IPM). The equations of motion of this
Sample Solution for Problem 1.a 1 Inverted Pendulum Model (IPM) 1.1 Equations of Motion and Ground Reaction Forces Figure 1: Scheme of the Inverted Pendulum Model (IPM). The equations of motion of this
VARIATIONS IN FORCE TIME HISTORIES OF CAT GASTROCNEMIUS, SOLEUS AND PLANTARIS MUSCLES FOR CONSECUTIVE WALKING STEPS
 J. exp. iol. 191, 19 36 (1994) Printed in Great ritain The Company of iologists Limited 1994 19 VARIATIONS IN FORCE TIME HISTORIES OF CAT GASTROCNEMIUS, SOLEUS AND PLANTARIS MUSCLES FOR CONSECUTIVE WALKING
J. exp. iol. 191, 19 36 (1994) Printed in Great ritain The Company of iologists Limited 1994 19 VARIATIONS IN FORCE TIME HISTORIES OF CAT GASTROCNEMIUS, SOLEUS AND PLANTARIS MUSCLES FOR CONSECUTIVE WALKING
Neuromuscular Coordination during Slope Walking. Andrea N. Lay
 Neuromuscular Coordination during Slope Walking A Thesis Presented to The Academic Faculty by Andrea N. Lay In Partial Fulfillment Of the Requirements for the Degree Doctor of Philosophy in Bioengineering
Neuromuscular Coordination during Slope Walking A Thesis Presented to The Academic Faculty by Andrea N. Lay In Partial Fulfillment Of the Requirements for the Degree Doctor of Philosophy in Bioengineering
A Bio-inspired Behavior Based Bipedal Locomotion Control B4LC Method for Bipedal Upslope Walking
 1 A Bio-inspired Behavior Based Bipedal Locomotion Control B4LC Method for Bipedal Upslope Walking JIE ZHAO, QI LIU, STEFFEN SCHUETZ, and KARSTEN BERNS Robotics Research Lab, University of Kaiserslautern,
1 A Bio-inspired Behavior Based Bipedal Locomotion Control B4LC Method for Bipedal Upslope Walking JIE ZHAO, QI LIU, STEFFEN SCHUETZ, and KARSTEN BERNS Robotics Research Lab, University of Kaiserslautern,
Does Ski Width Influence Muscle Action in an Elite Skier? A Case Study. Montana State University Movement Science Laboratory Bozeman, MT 59717
 Does Ski Width Influence Muscle Action in an Elite Skier? A Case Study John G. Seifert 1, Heidi Nunnikhoven 1, Cory Snyder 1, Ronald Kipp 2 1 Montana State University Movement Science Laboratory Bozeman,
Does Ski Width Influence Muscle Action in an Elite Skier? A Case Study John G. Seifert 1, Heidi Nunnikhoven 1, Cory Snyder 1, Ronald Kipp 2 1 Montana State University Movement Science Laboratory Bozeman,
Biomechanical analysis of the medalists in the 10,000 metres at the 2007 World Championships in Athletics
 STUDY Biomechanical analysis of the medalists in the 10,000 metres at the 2007 World Championships in Athletics by IAAF 23:3; 61-66, 2008 By Yasushi Enomoto, Hirosuke Kadono, Yuta Suzuki, Tetsu Chiba,
STUDY Biomechanical analysis of the medalists in the 10,000 metres at the 2007 World Championships in Athletics by IAAF 23:3; 61-66, 2008 By Yasushi Enomoto, Hirosuke Kadono, Yuta Suzuki, Tetsu Chiba,
Reactive balance adjustments to unexpected perturbations during human walking
 Gait and Posture 16 (2002) 238/248 www.elsevier.com/locate/gaitpost Reactive balance adjustments to unexpected perturbations during human walking Reed Ferber a, *, Louis R. Osternig b, Marjorie H. Woollacott
Gait and Posture 16 (2002) 238/248 www.elsevier.com/locate/gaitpost Reactive balance adjustments to unexpected perturbations during human walking Reed Ferber a, *, Louis R. Osternig b, Marjorie H. Woollacott
+ t1 t2 moment-time curves
 Part 6 - Angular Kinematics / Angular Impulse 1. While jumping over a hurdle, an athlete s hip angle was measured to be 2.41 radians. Within 0.15 seconds, the hurdler s hip angle changed to be 3.29 radians.
Part 6 - Angular Kinematics / Angular Impulse 1. While jumping over a hurdle, an athlete s hip angle was measured to be 2.41 radians. Within 0.15 seconds, the hurdler s hip angle changed to be 3.29 radians.
Supplementary Figure 1 An insect model based on Drosophila melanogaster. (a)
 Supplementary Figure 1 An insect model based on Drosophila melanogaster. (a) Side and ventral images of adult female flies used to calculate the sizes of body and leg segments. Scale bar is 0.3 mm. Green,
Supplementary Figure 1 An insect model based on Drosophila melanogaster. (a) Side and ventral images of adult female flies used to calculate the sizes of body and leg segments. Scale bar is 0.3 mm. Green,
Muscle force redistributes segmental power for body progression during walking
 Gait and Posture 19 (2004) 194 205 Muscle force redistributes segmental power for body progression during walking R.R. Neptune a,b,, F.E. Zajac b,c,d, S.A. Kautz b,e,f,g a Department of Mechanical Engineering,
Gait and Posture 19 (2004) 194 205 Muscle force redistributes segmental power for body progression during walking R.R. Neptune a,b,, F.E. Zajac b,c,d, S.A. Kautz b,e,f,g a Department of Mechanical Engineering,
GAIT RECOVERY IN HEALTHY SUBJECTS: PERTURBATIONS TO THE KNEE MOTION WITH A SMART KNEE BRACE. by Mehmet Temel
 GAIT RECOVERY IN HEALTHY SUBJECTS: PERTURBATIONS TO THE KNEE MOTION WITH A SMART KNEE BRACE by Mehmet Temel A thesis submitted to the Faculty of the University of Delaware in partial fulfillment of the
GAIT RECOVERY IN HEALTHY SUBJECTS: PERTURBATIONS TO THE KNEE MOTION WITH A SMART KNEE BRACE by Mehmet Temel A thesis submitted to the Faculty of the University of Delaware in partial fulfillment of the
Interlimb Coordination During Locomotion: What Can be Adapted and Stored?
 J Neurophysiol 94: 2403 2415, 2005. First published June 15, 2005; doi:10.1152/jn.00089.2005. Interlimb Coordination During Locomotion: What Can be Adapted and Stored? Darcy S. Reisman, 1,2 Hannah J. Block,
J Neurophysiol 94: 2403 2415, 2005. First published June 15, 2005; doi:10.1152/jn.00089.2005. Interlimb Coordination During Locomotion: What Can be Adapted and Stored? Darcy S. Reisman, 1,2 Hannah J. Block,
Neuromuscular Reflexes
 Name: Neuromuscular Reflexes Experiment 14A The automatic response of a muscle to a stimulus is called a reflex. The patellar reflex results from tapping the patellar tendon below the knee with a reflex
Name: Neuromuscular Reflexes Experiment 14A The automatic response of a muscle to a stimulus is called a reflex. The patellar reflex results from tapping the patellar tendon below the knee with a reflex
Leg kinematics and muscle activity during treadmill running in the cockroach, Blaberus discoidalis : II. Fast running
 J Comp Physiol A (1998) 182: 23±33 Ó Springer-Verlag 1998 ORIGINAL PAPER J. T. Watson á R. E. Ritzmann Leg kinematics and muscle activity during treadmill running in the cockroach, Blaberus discoidalis
J Comp Physiol A (1998) 182: 23±33 Ó Springer-Verlag 1998 ORIGINAL PAPER J. T. Watson á R. E. Ritzmann Leg kinematics and muscle activity during treadmill running in the cockroach, Blaberus discoidalis
Coaching the Triple Jump Boo Schexnayder
 I. Understanding the Event A. The Run and Its Purpose B. Hip Undulation and the Phases C. Making the Connection II. III. IV. The Approach Run A. Phases B. Technical Features 1. Posture 2. Progressive Body
I. Understanding the Event A. The Run and Its Purpose B. Hip Undulation and the Phases C. Making the Connection II. III. IV. The Approach Run A. Phases B. Technical Features 1. Posture 2. Progressive Body
SIMULTANEOUS RECORDINGS OF VELOCITY AND VIDEO DURING SWIMMING
 Portuguese Journal of Sport Sciences. 6:supl. 2, 32-35, 2006 SIMULTANEOUS RECORDINGS OF VELOCITY AND VIDEO DURING SWIMMING Albert B. Craig 1, Budd Termin2, and David R. Pendergast 2 1University of Rochester,
Portuguese Journal of Sport Sciences. 6:supl. 2, 32-35, 2006 SIMULTANEOUS RECORDINGS OF VELOCITY AND VIDEO DURING SWIMMING Albert B. Craig 1, Budd Termin2, and David R. Pendergast 2 1University of Rochester,
25 Lower-Limb Muscle Function in Human Running
 25 Lower-Limb Muscle Function in Human Running Anthony G. Schache, Tim W. Dorn, and Marcus G. Pandy * Abstract. This paper provides a brief summary of work completed to date in our research laboratory
25 Lower-Limb Muscle Function in Human Running Anthony G. Schache, Tim W. Dorn, and Marcus G. Pandy * Abstract. This paper provides a brief summary of work completed to date in our research laboratory
Available online at Prediction of energy efficient pedal forces in cycling using musculoskeletal simulation models
 Available online at www.sciencedirect.com Engineering 2 00 (2010) (2009) 3211 3215 000 000 Engineering www.elsevier.com/locate/procedia 8 th Conference of the International Sports Engineering Association
Available online at www.sciencedirect.com Engineering 2 00 (2010) (2009) 3211 3215 000 000 Engineering www.elsevier.com/locate/procedia 8 th Conference of the International Sports Engineering Association
C. P. McGowan, 1 R. R. Neptune, 1 and R. Kram 2 1
 J Appl Physiol 105: 486 494, 2008. First published June 12, 2008; doi:10.1152/japplphysiol.90448.2008. Independent effects of weight and mass on plantar flexor activity during walking: implications for
J Appl Physiol 105: 486 494, 2008. First published June 12, 2008; doi:10.1152/japplphysiol.90448.2008. Independent effects of weight and mass on plantar flexor activity during walking: implications for
Experimental and Computational Assessment of Locomotor Coordination and. Complexity Following Incomplete Spinal Cord Injury in the Rat.
 Experimental and Computational Assessment of Locomotor Coordination and Complexity Following Incomplete Spinal Cord Injury in the Rat by Brian Hillen A Dissertation Presented in Partial Fulfillment of
Experimental and Computational Assessment of Locomotor Coordination and Complexity Following Incomplete Spinal Cord Injury in the Rat by Brian Hillen A Dissertation Presented in Partial Fulfillment of
THE ANKLE-HIP TRANSVERSE PLANE COUPLING DURING THE STANCE PHASE OF NORMAL WALKING
 THE ANKLE-HIP TRANSVERSE PLANE COUPLING DURING THE STANCE PHASE OF NORMAL WALKING Thales R. Souza, Rafael Z. Pinto, Renato G. Trede, Nadja C. Pereira, Renata N. Kirkwood and Sérgio T. Fonseca. Movement
THE ANKLE-HIP TRANSVERSE PLANE COUPLING DURING THE STANCE PHASE OF NORMAL WALKING Thales R. Souza, Rafael Z. Pinto, Renato G. Trede, Nadja C. Pereira, Renata N. Kirkwood and Sérgio T. Fonseca. Movement
MECHANISMS OF COUPLING BETWEEN THE IPSILATERAL LEGS OF A WALKING INSECT (CARAUSIUS MOROSUS)
 U. exp. Biol. 138, 455-469 (1988) 455 fprinted in Great Britain The Company of Biologists Limited 1988 MECHANISMS OF COUPLING BETWEEN THE IPSILATERAL LEGS OF A WALKING INSECT (CARAUSIUS MOROSUS) BY H.
U. exp. Biol. 138, 455-469 (1988) 455 fprinted in Great Britain The Company of Biologists Limited 1988 MECHANISMS OF COUPLING BETWEEN THE IPSILATERAL LEGS OF A WALKING INSECT (CARAUSIUS MOROSUS) BY H.
Gait Analysis by High School Students
 Gait Analysis by High School Students André Heck 1 and Caroline van Dongen 2 1 AMSTEL Institute, University of Amsterdam, Amsterdam, The Netherlands, heck@science.uva.nl 2 St. Antoniuscollege, Gouda, The
Gait Analysis by High School Students André Heck 1 and Caroline van Dongen 2 1 AMSTEL Institute, University of Amsterdam, Amsterdam, The Netherlands, heck@science.uva.nl 2 St. Antoniuscollege, Gouda, The
Changes in muscle activity with increase in leg stiffness during hopping
 Neuroscience Letters 418 (2007) 55 59 Changes in muscle activity with increase in leg stiffness during hopping Hiroaki Hobara, Kazuyuki Kanosue, Shuji Suzuki Graduate School of Human Sciences, Waseda University,
Neuroscience Letters 418 (2007) 55 59 Changes in muscle activity with increase in leg stiffness during hopping Hiroaki Hobara, Kazuyuki Kanosue, Shuji Suzuki Graduate School of Human Sciences, Waseda University,
Factors that Determine the Magnitude and Time Course of Human H-Reflexes in Locomotion
 The Journal of Neuroscience, February 1991, 17(2): 42-427 Factors that Determine the Magnitude and Time Course of Human H-Reflexes in Locomotion M. Edamura, J. F. Yang, and R. B. Stein Division of Neuroscience
The Journal of Neuroscience, February 1991, 17(2): 42-427 Factors that Determine the Magnitude and Time Course of Human H-Reflexes in Locomotion M. Edamura, J. F. Yang, and R. B. Stein Division of Neuroscience
Maria Knikou. Experimental Brain Research. ISSN Volume 228 Number 3. Exp Brain Res (2013) 228: DOI /s y
 Functional reorganization of soleus H- reflex modulation during stepping after robotic-assisted step training in people with complete and incomplete spinal cord injury Maria Knikou Experimental Brain Research
Functional reorganization of soleus H- reflex modulation during stepping after robotic-assisted step training in people with complete and incomplete spinal cord injury Maria Knikou Experimental Brain Research
SWING- AND SUPPORT-RELATED MUSCLE ACTIONS DIFFERENTIALLY TRIGGER HUMAN WALK RUN AND RUN WALK TRANSITIONS
 The Journal of Experimental Biology 24, 2277 2287 (21) Printed in Great Britain The Company of Biologists Limited 21 JEB389 2277 SWING- AND SUPPORT-RELATED MUSCLE ACTIONS DIFFERENTIALLY TRIGGER HUMAN WALK
The Journal of Experimental Biology 24, 2277 2287 (21) Printed in Great Britain The Company of Biologists Limited 21 JEB389 2277 SWING- AND SUPPORT-RELATED MUSCLE ACTIONS DIFFERENTIALLY TRIGGER HUMAN WALK
Normal and Abnormal Gait
 Normal and Abnormal Gait Adrielle Fry, MD EvergreenHealth, Division of Sport and Spine University of Washington Board Review Course March 6, 2017 What are we going to cover? Definitions and key concepts
Normal and Abnormal Gait Adrielle Fry, MD EvergreenHealth, Division of Sport and Spine University of Washington Board Review Course March 6, 2017 What are we going to cover? Definitions and key concepts
SPRINTING CHARACTERISTICS OF WOMEN S 100 METER FINALS AT THE IAAF WORLD CHAMPIONSHOPS DAEGU 2011
 9:30-9:45 am Sang-Kyoon Park. Sprinting characteristics of women's 100 meter finals at the IAAF world championships Daegu 2011. (228) SPRINTING CHARACTERISTICS OF WOMEN S 100 METER FINALS AT THE IAAF WORLD
9:30-9:45 am Sang-Kyoon Park. Sprinting characteristics of women's 100 meter finals at the IAAF world championships Daegu 2011. (228) SPRINTING CHARACTERISTICS OF WOMEN S 100 METER FINALS AT THE IAAF WORLD
Detection of Forces and Body Load in Standing and Walking in the American Cockroach
 Marshall University Marshall Digital Scholar Theses, Dissertations and Capstones 2003 Detection of Forces and Body Load in Standing and Walking in the American Cockroach J. Adam Noah Follow this and additional
Marshall University Marshall Digital Scholar Theses, Dissertations and Capstones 2003 Detection of Forces and Body Load in Standing and Walking in the American Cockroach J. Adam Noah Follow this and additional
Spastic Reflexes Triggered by Ankle Load Release in Human Spinal Cord Injury
 J Neurophysiol 96: 2941 2950, 2006; doi:10.1152/jn.00186.2006. Spastic Reflexes Triggered by Ankle Load Release in Human Spinal Cord Injury Ming Wu 1,2 and Brian D. Schmit 1,2,3 1 Sensory Motor Performance
J Neurophysiol 96: 2941 2950, 2006; doi:10.1152/jn.00186.2006. Spastic Reflexes Triggered by Ankle Load Release in Human Spinal Cord Injury Ming Wu 1,2 and Brian D. Schmit 1,2,3 1 Sensory Motor Performance
Kinematic, Kinetic & Electromyographic Characteristics of the Sprinting Stride of Top Female Sprinters
 Kinematic, Kinetic & Electromyographic Characteristics of the Sprinting Stride of Top Female Sprinters Milan Coh, Ales Dolenec & Bojan Jost University of Ljubljana, Slovenia The main purpose of this study
Kinematic, Kinetic & Electromyographic Characteristics of the Sprinting Stride of Top Female Sprinters Milan Coh, Ales Dolenec & Bojan Jost University of Ljubljana, Slovenia The main purpose of this study
Motion Analysis on Backward Walking: Kinetics, Kinematics, and Electromyography
 Motion Analysis on Backward Walking: Kinetics, Kinematics, and Electromyography Min Hyeon Lee The Graduate School Yonsei University Department of Biomedical Engineering Motion Analysis on Backward Walking:
Motion Analysis on Backward Walking: Kinetics, Kinematics, and Electromyography Min Hyeon Lee The Graduate School Yonsei University Department of Biomedical Engineering Motion Analysis on Backward Walking:
USA Track & Field Heptathlon Summit- November
 USA Track & Field Heptathlon Summit- November 1994 1 I. Technical considerations in the sprint hurdles Practical Biomechanics For the 100m Hurdles By Gary Winckler University of Illinois A. General flow
USA Track & Field Heptathlon Summit- November 1994 1 I. Technical considerations in the sprint hurdles Practical Biomechanics For the 100m Hurdles By Gary Winckler University of Illinois A. General flow
RESEARCH ARTICLE Modulation of joint moments and work in the goat hindlimb with locomotor speed and surface grade
 22 The Journal of Experimental Biology 26, 22-222 23. Published by The Company of Biologists Ltd doi:.242/jeb.8249 RESEARCH ARTICLE Modulation of joint moments and work in the goat hindlimb with locomotor
22 The Journal of Experimental Biology 26, 22-222 23. Published by The Company of Biologists Ltd doi:.242/jeb.8249 RESEARCH ARTICLE Modulation of joint moments and work in the goat hindlimb with locomotor
14A. Neuromuscular Reflexes. Experiment
 Experiment 14A The automatic response of a muscle to a stimulus is called a reflex. The patellar reflex results from tapping the patellar tendon below the knee with a reflex hammer. This causes contraction
Experiment 14A The automatic response of a muscle to a stimulus is called a reflex. The patellar reflex results from tapping the patellar tendon below the knee with a reflex hammer. This causes contraction
The EMG activity and mechanics of the running jump as a function of takeoff angle
 Journal of Electromyography and Kinesiology 11 (2001) 365 372 www.elsevier.com/locate/jelekin The EMG activity and mechanics of the running jump as a function of takeoff angle W. Kakihana *, S. Suzuki
Journal of Electromyography and Kinesiology 11 (2001) 365 372 www.elsevier.com/locate/jelekin The EMG activity and mechanics of the running jump as a function of takeoff angle W. Kakihana *, S. Suzuki
Muscle force distribution during forward and backward locomotion
 Acta of Bioengineering and Biomechanics Vol. 15, No. 3, 2013 Original paper DOI: 10.5277/abb130301 First award of Morecki & Fidelus Scientific Awards Muscle force distribution during forward and backward
Acta of Bioengineering and Biomechanics Vol. 15, No. 3, 2013 Original paper DOI: 10.5277/abb130301 First award of Morecki & Fidelus Scientific Awards Muscle force distribution during forward and backward
Modulation of hamstrings reflexive responses during human gait
 University of Iowa Iowa Research Online Theses and Dissertations Spring 2012 Modulation of hamstrings reflexive responses during human gait Brad Wayne Floy University of Iowa Copyright 2012 Brad Floy This
University of Iowa Iowa Research Online Theses and Dissertations Spring 2012 Modulation of hamstrings reflexive responses during human gait Brad Wayne Floy University of Iowa Copyright 2012 Brad Floy This
Myths and Science in Cycling
 Myths and Science in Cycling John McDaniel, PhD Kent State University Jim Martin, PhD - U of Utah Steve Elmer, PhD- Michigan Tech Who am I PhD in Exercise Physiology under Dr. Jim Martin at the University
Myths and Science in Cycling John McDaniel, PhD Kent State University Jim Martin, PhD - U of Utah Steve Elmer, PhD- Michigan Tech Who am I PhD in Exercise Physiology under Dr. Jim Martin at the University
Adaptation to Knee Flexion Torque Assistance in Double Support Phase
 Adaptation to Knee Flexion Torque Assistance in Double Support Phase James S. Sulzer, Keith E. Gordon, T. George Hornby, Michael A. Peshkin and James L. Patton Abstract Studies have shown locomotor adaptation
Adaptation to Knee Flexion Torque Assistance in Double Support Phase James S. Sulzer, Keith E. Gordon, T. George Hornby, Michael A. Peshkin and James L. Patton Abstract Studies have shown locomotor adaptation
(2) BIOMECHANICS of TERRESTRIAL LOCOMOTION
 (2) BIOMECHANICS of TERRESTRIAL LOCOMOTION Questions: - How does size influence the mode and speed of locomotion? - What determines the energy cost of locomotion? - Why do humans walk and run the way we
(2) BIOMECHANICS of TERRESTRIAL LOCOMOTION Questions: - How does size influence the mode and speed of locomotion? - What determines the energy cost of locomotion? - Why do humans walk and run the way we
RUNNING SHOE STIFFNESS: THE EFFECT ON WALKING GAIT
 RUNNING SHOE STIFFNESS: THE EFFECT ON WALKING GAIT Stephen N Stanley, Peter J M c Nair, Angela G Walker, & Robert N Marshall Auckland Institute of Technology, Auckland, New Zealand University of Auckland,
RUNNING SHOE STIFFNESS: THE EFFECT ON WALKING GAIT Stephen N Stanley, Peter J M c Nair, Angela G Walker, & Robert N Marshall Auckland Institute of Technology, Auckland, New Zealand University of Auckland,
Megan E. Krause, BSBSE, Young Hui Chang, Ph.D. Comparative Neuromechanics Laboratory. Georgia Institute of Technology
 Megan E. Krause, BSBSE, Young Hui Chang, Ph.D. Comparative Neuromechanics Laboratory Sh School of Applied Physiology Georgia Institute of Technology 1 Variety of ankle constraints during locomotion: Fashion
Megan E. Krause, BSBSE, Young Hui Chang, Ph.D. Comparative Neuromechanics Laboratory Sh School of Applied Physiology Georgia Institute of Technology 1 Variety of ankle constraints during locomotion: Fashion
a. Specific Aims The Specific Aims are unchanged from the original application. There have been no staff changes during this past year.
 g-lio-6/9) a. Specific Aims The Specific Aims are unchanged from the original application. b. Studies and Results This report presents work conducted from April 15, 2004 to March 31, 2005. During this
g-lio-6/9) a. Specific Aims The Specific Aims are unchanged from the original application. b. Studies and Results This report presents work conducted from April 15, 2004 to March 31, 2005. During this
INTRODUCTION TO GAIT ANALYSIS DATA
 INTRODUCTION TO GAIT ANALYSIS DATA 1. Phases of gait a. Stance (% gc) i. Loading response (10%) ii. Mid- and terminal stance (%) iii. Pre-swing (10%) b. Swing (% gc) i. Initial swing ii. Mid-swing iii.
INTRODUCTION TO GAIT ANALYSIS DATA 1. Phases of gait a. Stance (% gc) i. Loading response (10%) ii. Mid- and terminal stance (%) iii. Pre-swing (10%) b. Swing (% gc) i. Initial swing ii. Mid-swing iii.
Analysis of Backward Falls Caused by Accelerated Floor Movements Using a Dummy
 Original Article Analysis of Backward Falls Caused by Accelerated Floor Movements Using a Dummy Hisao NAGATA 1 * and Hisato OHNO 2 1 National Institute of Occupational Safety and Health, 1 4 6 Umezono,
Original Article Analysis of Backward Falls Caused by Accelerated Floor Movements Using a Dummy Hisao NAGATA 1 * and Hisato OHNO 2 1 National Institute of Occupational Safety and Health, 1 4 6 Umezono,
Analysis of Skip Motion as a Recovery Strategy after an Induced Trip
 2015 IEEE International Conference on Systems, Man, and Cybernetics Analysis of Skip Motion as a Recovery Strategy after an Induced Trip Kento Mitsuoka, Yasuhiro Akiyama, Yoji Yamada, and Shogo Okamoto
2015 IEEE International Conference on Systems, Man, and Cybernetics Analysis of Skip Motion as a Recovery Strategy after an Induced Trip Kento Mitsuoka, Yasuhiro Akiyama, Yoji Yamada, and Shogo Okamoto
1. Hip flexion Muscles: Iliopsoas (psoas major + iliacus)
 Chap. 5 Testing the muscles of the Lower Extremity Part I. Manual Muscle Testing of the hip joint muscles 1. Hip flexion Muscles: Iliopsoas (psoas major + iliacus) Rectus femoris Sartorius Tensor fascia
Chap. 5 Testing the muscles of the Lower Extremity Part I. Manual Muscle Testing of the hip joint muscles 1. Hip flexion Muscles: Iliopsoas (psoas major + iliacus) Rectus femoris Sartorius Tensor fascia
Supplementary,Material,!
 Supplementary,Material, MTJ,Tracking,Methods, WeestimatedAT,muscle,andMTUlengthchangesusingpreviouslypublishedMTJtrackingmethods (Hoffrén et al., 212; Lichtwark, 25; Lichtwark and Wilson, 26). The ultrasound
Supplementary,Material, MTJ,Tracking,Methods, WeestimatedAT,muscle,andMTUlengthchangesusingpreviouslypublishedMTJtrackingmethods (Hoffrén et al., 212; Lichtwark, 25; Lichtwark and Wilson, 26). The ultrasound
Impact of heel position on leg muscles during walking
 Original article Niigata Journal of Health and Welfare Vol. 14, No. 1 Impact of heel position on leg muscles during walking Koichi Akaishi Graduate School of Health and Welfare, Niigata University of Health
Original article Niigata Journal of Health and Welfare Vol. 14, No. 1 Impact of heel position on leg muscles during walking Koichi Akaishi Graduate School of Health and Welfare, Niigata University of Health
Nature Neuroscience: doi: /nn Supplementary Figure 1. Visual responses of the recorded LPTCs
 Supplementary Figure 1 Visual responses of the recorded LPTCs (a) The mean±sd (n=3 trials) of the direction-selective (DS) responses (i.e., subtracting the null direction, ND, from the preferred direction,
Supplementary Figure 1 Visual responses of the recorded LPTCs (a) The mean±sd (n=3 trials) of the direction-selective (DS) responses (i.e., subtracting the null direction, ND, from the preferred direction,
COMPARISON STUDY BETWEEN THE EFFICIENY OF THE START TECHNIQUES IN THE ROMANIAN COMPETITIVE SWIMMING
 Bulletin of the Transilvania University of Braşov Series IX: Sciences of Human Kinetics Vol. 6 (55) No. 1 2013 COMPARISON STUDY BETWEEN THE EFFICIENY OF THE START TECHNIQUES IN THE ROMANIAN COMPETITIVE
Bulletin of the Transilvania University of Braşov Series IX: Sciences of Human Kinetics Vol. 6 (55) No. 1 2013 COMPARISON STUDY BETWEEN THE EFFICIENY OF THE START TECHNIQUES IN THE ROMANIAN COMPETITIVE
