How To Use Getters and Getter Pumps
|
|
|
- Bartholomew Carpenter
- 6 years ago
- Views:
Transcription
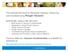
1 A Journal of Practical and Useful Vacuum Technology From By Phil Danielson How To Use Getters and Getter Pumps Gettering is a vacuum pumping technology that has been with us, in many forms, for almost as long as vacuum technology itself has been in existence. Historically, especially in the early days of electron tubes, vacuum practitioners were concerned with clean-up of the residual gases and needed to get them. Obviously, then, the gases would be gotten by getters. This would mean tying up gases by some means that would result in removing them from the vacuum vessel. This would put getter pumps within the overall category of capture pumps along with cryopumps and sputter-ion pumps. Capture pumps can be taken as an analog of a garbage can. Gettering, as a category, must also be broken down into physical getters and chemical getters, or some nomenclature problems will result. Physical getters are often found in cryostats or dewars where a zeolite material such as molecular sieve is used to physically absorb and hold water vapor. If, as is often the case, the molecular sieve is placed against a surface at liquid nitrogen temperature with good thermal contact, it will absorb and hold the common air gases as well. This is the same pumping technique used by cryosorption roughing pumps where the pumped gases will be pumped and held as long as the sieve is maintained at a low enough temperature. Chemical getters, though, are what is most often meant when the term getter is used. A chemical getter provides a pumping action by a chemical reaction where a chemically active gas combines with a chemically active metal to form a low vapor pressure solid compound. The chemically active metal can be either an element or an alloy that, for convenience, can be called a getter metal (GM) which provides pumping actions which can be shown in the following simplified chemical equations: GM + O 2 GMO GM + N 2 GMN GM + CO 2 CO + GMO GMC + GMO GM + CO GMC + GMO GM + H 2 O H + GMO GMO + H (bulk) GM + H 2 GM + H (bulk) GM + Hydrocarbons, C x H x, etc. GMC + H (bulk) GM + He, Ne, Ar, Kr, Xe (inert gases) No Reaction
2 The reactions of the chemically active gases with the getter metal are fairly straightforward. They all form a low vapor pressure ceramic compound where the active gas is permanently removed from the vacuum chamber. The inert gases are not pumped at all due to the obvious reason that they are inert and will not react with the getter metal. Hydrogen (H 2 ), though, is the odd gas out in that it does not react to form a chemical compound but merely dissolves in the getter metal to form a solid solution. Chemical getters can be broken down into two separate categories; evaporable and non-evaporable. Chamber Ti Schematic of TSP in Chamb er Getter Surface TSP Cartridge Evaporable getters are often used in electron tubes where a mirror-like metal coating can be easily observed on the inside of the glass envelope. When the tube has been pumped down and ready for tip-off to seal it, a slug of getter metal, often barium or a barium alloy, is heated to a high enough temperature to evaporate it so that it can subsequently condense on the tube s inner surfaces. There it forms a high surface area reactive coating that will remain as an in situ pump after the tube is pinched off. Another common example is the titanium sublimation pump (TSP.) A TSP operates by heating either a titanium/molybdenum alloy filament directly or a slug of titanium indirectly by radiation from a filament to about 1450 o C where the titanium sublimes. Sublimation is defined as going directly from the solid state to the vapor state without passing through the liquid state. A common example of sublimation is hanging out a wet piece of cloth to dry in the winter where the water freezes but still evaporates. Once sublimed into the vapor state, the titanium is allowed to condense on an internal array or on a portion of the inner surface of the chamber. As it condenses into a thin film, the titanium becomes a pump for active gases and H 2. The pumping speed of the condensed film is directly proportional to the total surface area of the film. The pumping speed of a fresh film will decline as it reacts (pumps) the gas in the chamber, and the lifetime of the film is dependent upon the gas load it is required to pump. The higher the gas load, the shorter the lifetime will be. Under high gas load conditions, the titanium source will need to periodically replenish the film to maintain pumping speed, and this can be a problem. The titanium source will have become saturated with hydrogen when it cooled after reaching sublimation temperature, and re-heating releases the hydrogen. This up and down temperature cycle will result in pressure spikes as further cycling continues. The solution for this potential problem is to maintain the source at an elevated, but below sublimation temperature, at all times. Additional problems result from the layering of fresh titanium over saturated titanium. The film layers become highly stressed to the point that cracking and
3 peeling can result. H 2, dissolving in the titanium, adds to this effect since the bulk volume of the film increases with the amount of dissolved H 2. Both of these effects can cause particles to form within the vacuum chamber with disastrous results to some processes. Conversely, TSPs are an efficient and economical source of high speed and clean pumping, and this is especially true for ultrahigh vacuum applications where the low gas load will allow very long film lifetimes. Non-evaporable getters (NEG), as can be assumed from their name, remain in the solid state instead of being evaporated and condensed on a surface. This family of getters is usually, but not always, alloys of zirconium. Although they can be in any solid form, they are most often found as either chunks or pellets, or they are found as thin films bonded to metallic substrates. Activation is a process that s required for NEGs. When the getter material is exposed to air for handling or loading into a system or device, the material s surface will skin over with reacted gases. This means that the NEG will be totally enclosed in an envelope of oxides, Getter pellet with clean nitrides, etc. as shown by the gettering surface, active gas equations shown above. Additionally, the bulk of molecule approaching the material will be saturated with dissolved H 2. Since, under these conditions, the getter material will be essentially inert, it will not provide an active getter-pumping surface. Activation, then, is the process to prepare the getter surface for pumping. This is done in situ Getter pellet with active by heating under vacuum after being installed. gas molecule reacted This process is two-fold. During heating, the on surface reacted skin layer will diffuse into the NEGs bulk in an attempt to achieve a constant concentration equilibrium throughout the bulk and the H 2 will be driven out of solid solution into the chamber where it can be pumped away by Getter pellet with active gas molecule diffused an appropriate high vacuum pump. The time into bulk by heating and temperature required for activation will vary with the specific getter alloy, and the procedure recommended by the manufacturer should be followed. The vacuum level required will depend Getter pellets can react with gas molecules, whichusually form a low upon the actual application, but a pressure of at vapor pressure ceramic compound. least 10-4 torr is usually required and pumping time must be long enough to ensure that the released H 2 is pumped away. Any H 2 remaining in the chamber will be re-pumped by the getter, and this will limit the amount of H 2 the getter can subsequently pump. As the newly
4 activated NEG cools, the newly clean and active surface will begin to react with the active gases and the bulk will begin to dissolve H 2 into the bulk. Pumping action, following activation, follows the reactions shown in the chemical equations. Gases will be removed from the vessel by reaction on the NEG s surface until enough gas is reacted to once again skin over the surface. At this point, pumping action for active gases ceases and the reacted skin begins to act as a barrier for further dissolution of H 2. The time required for this to happen is a function of the gas load to be pumped. In ultrahigh vacuum applications, this might be years, but might be minutes or hours in a high vacuum process with its attendant high gas loads. Gas loads that would result in a unacceptably short period of surface saturation are usually dealt with by raising the temperature of the NEG to some point above room temperature. The higher temperature will result in pumping and diffusion into the bulk taking place at the same time as sort of a continuous activation cycle. The higher the temperature, the faster the diffusion. As in most engineering considerations, compromise is required. A higher temperature that would result in very fast diffusion also reduces the amount of H 2 that can be contained in solid solution. Depending upon the actual NEG alloy in use, a compromise temperature must be chosen that trades-off diffusion rate for H 2 retention. The high H 2 Pumping speed often allows NEGs to be used as a specific H 2 booster pump in tandem with cryo or turbo pumps. Batch systems that are cycled repeatedly to atmospheric pressure require careful thought. Each time the NEG material sees air or any other active gas such as N 2,, re-activation is required. Additionally, if the gas loads are high enough to require that the material be heated in order to meet the gas loads, the material has to be cooled to room temperature before exposure. When an active gas encounters a hot NEG material, an exothermic reaction occurs that can run away in terms of temperature and present a very real safety problem. In a cycled system, the chamber can be released to argon instead of an active gas, or the getter can be contained within a separate housing that can be isolated from the chamber by an appropriate valve. The use of O-rings in getter-pumped systems presents a very specific problem. Since the O-rings are slightly gas-permeable, some ambient air will continually permeate into the chamber. This small level of gas load presents no problem in terms of the active air gases, but air contains about 0.9% of inert argon. Although this seems like such a minuscule amount of gas that it can be ignored, the argon will enter the chamber on a continuous basis. This means that a single ISO/KF-10 Viton O-ring will allow the argon pressure to rise from about 1 x 10-6 torr to above 1 x 10-4 torr in 100 hours in a one liter chamber or 10 hours in a 100 ml chamber. Obviously, using metallic gaskets instead of Viton is often a requirement. Another option is to provide an additional pump, such as a small appendage sputter-ion pump, that can handle the small argon gas load.
5 Getters and getter pumps can provide very economical and compact pumping in many applications if they are used properly. Understanding the pumping process and the behavior of the getter material is essential if good results are to be expected and attained. Reprinted with permission by R&D Magazine, all rights reserved. Copyright Cahners Business Information. A shorter version appeared in R&D Magazine, February 2001.
Matching Vacuum Pump to Process
 A Journal of Practical and Useful Vacuum Technology From By Phil Danielson Matching Vacuum Pump to Process What s the best vacuum pump? is one of those maddening questions that can only be answered with
A Journal of Practical and Useful Vacuum Technology From By Phil Danielson Matching Vacuum Pump to Process What s the best vacuum pump? is one of those maddening questions that can only be answered with
Introduction of Vacuum Science & Technology. Diffusion pumps used on the Calutron mass spectrometers during the Manhattan Project.
 Introduction of Vacuum Science & Technology Diffusion pumps used on the Calutron mass spectrometers during the Manhattan Project. 1 What is a vacuum? 760 mm Hg Vacuum ATM A vacuum is defined as less than
Introduction of Vacuum Science & Technology Diffusion pumps used on the Calutron mass spectrometers during the Manhattan Project. 1 What is a vacuum? 760 mm Hg Vacuum ATM A vacuum is defined as less than
How To Choose a Roughing/Backing Pump for the Turbo and Drag Family
 A Journal of Practical and Useful Vacuum Technology From By Phil Danielson How To Choose a Roughing/Backing Pump for the Turbo and Drag Family The process of choosing the right high vacuum pump to suit
A Journal of Practical and Useful Vacuum Technology From By Phil Danielson How To Choose a Roughing/Backing Pump for the Turbo and Drag Family The process of choosing the right high vacuum pump to suit
A Journal of Practical and Useful Vacuum Technology. By Phil Danielson
 A Journal of Practical and Useful Vacuum Technology From By Phil Danielson Thermal Conductivity Gauges Thermal conductivity pressure gauges are extremely common in vacuum technology, but an understanding
A Journal of Practical and Useful Vacuum Technology From By Phil Danielson Thermal Conductivity Gauges Thermal conductivity pressure gauges are extremely common in vacuum technology, but an understanding
E SC 412 Nanotechnology: Materials, Infrastructure, and Safety Wook Jun Nam The Pennsylvania State University
 E SC 412 Nanotechnology: Materials, Infrastructure, and Safety Wook Jun Nam Unit 2 Infrastructure Lecture 6 More on Vacuum Systems, Part III Unit Outline Infrastructure * What is it? * Why do we need it
E SC 412 Nanotechnology: Materials, Infrastructure, and Safety Wook Jun Nam Unit 2 Infrastructure Lecture 6 More on Vacuum Systems, Part III Unit Outline Infrastructure * What is it? * Why do we need it
Both physical and chemical effects come into play either separately or in combination.
 A Journal of Practical and Useful Vacuum Technology By Phil Danielson WHY CREATE A VACUUM? From Creating a vacuum has become a fairly common and often routine undertaking, but the commonness of the activity
A Journal of Practical and Useful Vacuum Technology By Phil Danielson WHY CREATE A VACUUM? From Creating a vacuum has become a fairly common and often routine undertaking, but the commonness of the activity
Chapter 8: Cryo-sorption pumps
 Chapter 8: Cryo-sorption pumps Cryo-sorption pumps offer a clean, quiet, safe, vibration free and inexpensive way to rough pump a vacuum system. They are often used on vacuum systems that are sensitive
Chapter 8: Cryo-sorption pumps Cryo-sorption pumps offer a clean, quiet, safe, vibration free and inexpensive way to rough pump a vacuum system. They are often used on vacuum systems that are sensitive
Short Introduction to Cryo-Pumps and Refrigerators. Dr Graham Rogers Leybold UK Ltd.
 Short Introduction to Cryo-Pumps and Refrigerators Dr Graham Rogers Leybold UK Ltd. Cryo Pumps Topics Physical principles of cryo-pumping Design and control of modern refrigerator cryo-pumps UHV and XHV
Short Introduction to Cryo-Pumps and Refrigerators Dr Graham Rogers Leybold UK Ltd. Cryo Pumps Topics Physical principles of cryo-pumping Design and control of modern refrigerator cryo-pumps UHV and XHV
The Principles of Vacuum Technology
 The Principles of Vacuum Technology Vacuum Terminology Vacuum units Vacuum regimes How to measure vacuum. Gauge designs. How to create vacuum Pump classifications and designs UHV compatibility considerations
The Principles of Vacuum Technology Vacuum Terminology Vacuum units Vacuum regimes How to measure vacuum. Gauge designs. How to create vacuum Pump classifications and designs UHV compatibility considerations
The Experts in Vacuum Solutions
 By Woodrow Farrow - Reprinted with permission from Specialty Gas Report Vacuum: a space that is relatively empty of matter, especially when that void has been created through artificial means. The earth
By Woodrow Farrow - Reprinted with permission from Specialty Gas Report Vacuum: a space that is relatively empty of matter, especially when that void has been created through artificial means. The earth
Vacuum Systems and Cryogenics for Integrated Circuit Fabrication Technology 01
 INAOE. Tonantzintla, Mexico. 2010-06-23. June 23 rd, 2010 Vacuum Systems and Cryogenics for Integrated Circuit Fabrication Technology 01 Joel Molina INAOE Microelectronics Group jmolina@inaoep.mx 1 Vacuum
INAOE. Tonantzintla, Mexico. 2010-06-23. June 23 rd, 2010 Vacuum Systems and Cryogenics for Integrated Circuit Fabrication Technology 01 Joel Molina INAOE Microelectronics Group jmolina@inaoep.mx 1 Vacuum
PURE SUBSTANCE. Nitrogen and gaseous air are pure substances.
 CLASS Third Units PURE SUBSTANCE Pure substance: A substance that has a fixed chemical composition throughout. Air is a mixture of several gases, but it is considered to be a pure substance. Nitrogen and
CLASS Third Units PURE SUBSTANCE Pure substance: A substance that has a fixed chemical composition throughout. Air is a mixture of several gases, but it is considered to be a pure substance. Nitrogen and
Honors Chemistry - Problem Set Chapter 13 Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT.
 Honors Chemistry - Problem Set Chapter 13 Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT. 1. Atmospheric pressure is 760 mm Hg. 2. The SI unit of pressure is
Honors Chemistry - Problem Set Chapter 13 Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT. 1. Atmospheric pressure is 760 mm Hg. 2. The SI unit of pressure is
This educational seminar discusses creating, measuring, and troubleshooting Rough Vacuum.
 This educational seminar discusses creating, measuring, and troubleshooting Rough Vacuum. Specifically, today s talk will cover: Brief review of Vacuum Fundamentals Applications Using Rough Vacuum Rough
This educational seminar discusses creating, measuring, and troubleshooting Rough Vacuum. Specifically, today s talk will cover: Brief review of Vacuum Fundamentals Applications Using Rough Vacuum Rough
Kinetic-Molecular Theory
 GASES Chapter Eleven Kinetic-Molecular Theory! Recall that our only previous description of gases stated that gases completely fill and take the shape of their containers.! The Kinetic-Molecular Theory
GASES Chapter Eleven Kinetic-Molecular Theory! Recall that our only previous description of gases stated that gases completely fill and take the shape of their containers.! The Kinetic-Molecular Theory
Chemistry Chapter 11 Test Review
 Chemistry Chapter 11 Test Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Pressure is the force per unit a. volume. c. length. b. surface area.
Chemistry Chapter 11 Test Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Pressure is the force per unit a. volume. c. length. b. surface area.
Under pressure pushing down
 Under pressure pushing down on me When Dalton was conducting his studies, which led him to the atomic-molecular theory of matter, he also included studies of the behaviour of gases. These led him to propose,
Under pressure pushing down on me When Dalton was conducting his studies, which led him to the atomic-molecular theory of matter, he also included studies of the behaviour of gases. These led him to propose,
Vacuum Science Techniques and Applications Dan Dessau Adv. Lab 2007
 Vacuum Science Techniques and Applications Dan Dessau Adv. Lab 2007 Vacuum increases the mean-free-path of gas molecules. Vacuum prevents chemical reaction. Vacuum removes contaminants from surfaces. Vacuum
Vacuum Science Techniques and Applications Dan Dessau Adv. Lab 2007 Vacuum increases the mean-free-path of gas molecules. Vacuum prevents chemical reaction. Vacuum removes contaminants from surfaces. Vacuum
This educational seminar discusses creating, measuring, and troubleshooting Ultra-High Vacuum ( Torr) systems.
 This educational seminar discusses creating, measuring, and troubleshooting UltraHigh Vacuum ( 10 8 10 12 Torr) systems. Specifically, today s talk will cover: Brief review of High Vacuum (Characteristics,
This educational seminar discusses creating, measuring, and troubleshooting UltraHigh Vacuum ( 10 8 10 12 Torr) systems. Specifically, today s talk will cover: Brief review of High Vacuum (Characteristics,
Name /74. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Ch 11 Gases STUDY GUIDE Accelerated Chemistry SCANTRON Name /74 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following statements
Ch 11 Gases STUDY GUIDE Accelerated Chemistry SCANTRON Name /74 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following statements
Oxford Instruments Austin, Inc.
 User User Manual Manual Issue 01 / November 2013 / Oxford Instruments Austin, Inc. Issue 01 / November 2013 / Original Instructions Original Instructions Issue 01 / November 2013 / Oxford Instruments Austin,
User User Manual Manual Issue 01 / November 2013 / Oxford Instruments Austin, Inc. Issue 01 / November 2013 / Original Instructions Original Instructions Issue 01 / November 2013 / Oxford Instruments Austin,
You should be able to: Describe Equipment Barometer Manometer. 5.1 Pressure Read and outline 5.1 Define Barometer
 A P CHEMISTRY - Unit 5: Gases Unit 5: Gases Gases are distinguished from other forms of matter, not only by their power of indefinite expansion so as to fill any vessel, however large, and by the great
A P CHEMISTRY - Unit 5: Gases Unit 5: Gases Gases are distinguished from other forms of matter, not only by their power of indefinite expansion so as to fill any vessel, however large, and by the great
Inert Atmosphere Guide
 Inert Atmosphere Guide The UV curing of acrylate monomers is a free radical polymerization process in which UV radiation is absorbed and activates a certain type of molecules, photoinitiators. The activated
Inert Atmosphere Guide The UV curing of acrylate monomers is a free radical polymerization process in which UV radiation is absorbed and activates a certain type of molecules, photoinitiators. The activated
ANL/APS/TB-16 ADVANCED PHOTON SOURCE ACCELERATOR ULTRAHIGH VACUUM GUIDE. Chian Liu and John Noonan
 ANL/APS/TB-16 ADVANCED PHOTON SOURCE ACCELERATOR ULTRAHIGH VACUUM GUIDE Chian Liu and John Noonan CONTENTS 1. Ultrahigh Vacuum Overview..... 1 1.1 Vacuum..... 1 1.2 Sources of Residual Gas... 2 1.3 Material
ANL/APS/TB-16 ADVANCED PHOTON SOURCE ACCELERATOR ULTRAHIGH VACUUM GUIDE Chian Liu and John Noonan CONTENTS 1. Ultrahigh Vacuum Overview..... 1 1.1 Vacuum..... 1 1.2 Sources of Residual Gas... 2 1.3 Material
Ultrahigh Vacuum in Superconducting Synchrotrons A.Smirnov, JINR, Dubna, Russia
 Ultrahigh Vacuum in Superconducting Synchrotrons A.Smirnov, JINR, Dubna, Russia Topics: - Pumping & outgasing of hydrogen - Cryogenic pumps & non evaporable getters for UHV - Superconducting accelerators:
Ultrahigh Vacuum in Superconducting Synchrotrons A.Smirnov, JINR, Dubna, Russia Topics: - Pumping & outgasing of hydrogen - Cryogenic pumps & non evaporable getters for UHV - Superconducting accelerators:
School of Chemistry SOP For Operation Of Glove Boxes
 School SOP for Operation of Glove Boxes: The following SOP provides guidelines on how to adequately and safely operating a standard laboratory glove box and its associated devices. General Information:
School SOP for Operation of Glove Boxes: The following SOP provides guidelines on how to adequately and safely operating a standard laboratory glove box and its associated devices. General Information:
REPRODUCIBILITY IN OPTICAL THIN FILM PROCESSING PART 1, THE VACUUM AND PUMPING
 ABSTRACT REPRODUCIBILITY IN OPTICAL THIN FILM PROCESSING PART 1, THE VACUUM AND PUMPING Ronald R. Willey Willey Optical, Consultants, 13039 Cedar Street, Charlevoix, MI 49720, USA Ph 231-237-9392, ron@willeyoptical.com
ABSTRACT REPRODUCIBILITY IN OPTICAL THIN FILM PROCESSING PART 1, THE VACUUM AND PUMPING Ronald R. Willey Willey Optical, Consultants, 13039 Cedar Street, Charlevoix, MI 49720, USA Ph 231-237-9392, ron@willeyoptical.com
Cryo-Evaporator Operation
 Cryo-Evaporator Operation Cara Ricci November 25, 2003 Thin Film Deposition THE UNIVERSITY OF TEXAS AT DALLAS ERIK JOHNSON SCHOOL OF ENGINEERING DOCUMENT NUMBER: FA2003-TF-008 EDITION: 1.1 PAGE: 1 of 28
Cryo-Evaporator Operation Cara Ricci November 25, 2003 Thin Film Deposition THE UNIVERSITY OF TEXAS AT DALLAS ERIK JOHNSON SCHOOL OF ENGINEERING DOCUMENT NUMBER: FA2003-TF-008 EDITION: 1.1 PAGE: 1 of 28
Dissolved Oxygen Guide
 Educat i onser i es Di ssol vedoxygengui de Dissolved Oxygen Guide Introduction Dissolved oxygen probes provide a convenient approach to essentially direct measurement of molecular oxygen. The membrane
Educat i onser i es Di ssol vedoxygengui de Dissolved Oxygen Guide Introduction Dissolved oxygen probes provide a convenient approach to essentially direct measurement of molecular oxygen. The membrane
Chapter 13 Temperature, Kinetic Theory, and the Gas Laws 497
 Chapter 13 Temperature, Kinetic Theory, and the Gas Laws 497 Figure 13.25 This photograph of Apollo 17 Commander Eugene Cernan driving the lunar rover on the Moon in 1972 looks as though it was taken at
Chapter 13 Temperature, Kinetic Theory, and the Gas Laws 497 Figure 13.25 This photograph of Apollo 17 Commander Eugene Cernan driving the lunar rover on the Moon in 1972 looks as though it was taken at
Cryogenics is the science of ultra low temperatures. Low temperatures are achieved by the liquefaction of gases.
 CRYOGENICS SAFETY Introduction Cryogenics is the science of ultra low temperatures. Low temperatures are achieved by the liquefaction of gases. The gases which are most widely used in industry and research
CRYOGENICS SAFETY Introduction Cryogenics is the science of ultra low temperatures. Low temperatures are achieved by the liquefaction of gases. The gases which are most widely used in industry and research
General Chemistry I Percent Yield of Hydrogen Gas From Magnesium and HCl
 Introduction For chemical reactions involving gases, gas volume measurements provide a convenient means of determining stoichiometric relationships. A gaseous product is collected in a long, thin graduated
Introduction For chemical reactions involving gases, gas volume measurements provide a convenient means of determining stoichiometric relationships. A gaseous product is collected in a long, thin graduated
Gases NO CALCULATORS MAY BE USED FOR THESE QUESTIONS
 NO CALCULATORS MAY BE USED FOR THESE QUESTIONS Questions 1-3 refer to the following gases at 0 C and 1 atm. (A) Ar (B) NO 2 (C) Xe (D) H 2 (E) N 2 1. Has an average atomic or molecular speed closest to
NO CALCULATORS MAY BE USED FOR THESE QUESTIONS Questions 1-3 refer to the following gases at 0 C and 1 atm. (A) Ar (B) NO 2 (C) Xe (D) H 2 (E) N 2 1. Has an average atomic or molecular speed closest to
Experiment 18 Properties of Gases
 Experiment 18 Properties of Gases E18-1 E18-2 The Task In this experiment you will investigate some of the properties of gases, i.e. how gases flow, their phase changes and chemical reactivity. Skills
Experiment 18 Properties of Gases E18-1 E18-2 The Task In this experiment you will investigate some of the properties of gases, i.e. how gases flow, their phase changes and chemical reactivity. Skills
The Origin and Time Dependence of the Amount and Composition of Non- Constituent Gases Present in Crystal Growth Systems
 The Origin and Time Dependence of the Amount and Composition of Non- Constituent Gases Present in Crystal Growth Systems Witold Palosz A Desorption of inert gases from, and their diffusion through the
The Origin and Time Dependence of the Amount and Composition of Non- Constituent Gases Present in Crystal Growth Systems Witold Palosz A Desorption of inert gases from, and their diffusion through the
Detector Carrier Gas Comments Detector anode purge or reference gas. Electron Capture Nitrogen Maximum sensitivity Nitrogen Argon/Methane
 Gas requirements Gases for packed columns The carrier gas you use depends upon the type of detector and the performance requirements. Table 520-1 lists gas recommendations for packed column use. In general,
Gas requirements Gases for packed columns The carrier gas you use depends upon the type of detector and the performance requirements. Table 520-1 lists gas recommendations for packed column use. In general,
Usage Policies Notebook for Xenon Difluoride (XeF 2 ) Isotropic Si Etch
 Usage Policies Notebook for Xenon Difluoride (XeF 2 ) Isotropic Si Etch Revision date September 2014 2 Emergency Plan for XeF 2 Si Etcher Standard Operating Procedures for Emergencies Contact information
Usage Policies Notebook for Xenon Difluoride (XeF 2 ) Isotropic Si Etch Revision date September 2014 2 Emergency Plan for XeF 2 Si Etcher Standard Operating Procedures for Emergencies Contact information
A. What are the three states of matter chemists work with?
 Chapter 10 and 12 The Behavior of Gases Chapter 10 The States of Matter A. What are the three states of matter chemists work with? Section 10.1 Pg 267 B. We will explain the behavior of gases using the
Chapter 10 and 12 The Behavior of Gases Chapter 10 The States of Matter A. What are the three states of matter chemists work with? Section 10.1 Pg 267 B. We will explain the behavior of gases using the
Chemistry 1B Chapter 10 Worksheet - Daley. Name
 Name 1) The National Weather Service routinely supplies atmospheric pressure data to help pilots set their altimeters. The units the NWS uses for atmospheric pressure are inches of mercury. A barometric
Name 1) The National Weather Service routinely supplies atmospheric pressure data to help pilots set their altimeters. The units the NWS uses for atmospheric pressure are inches of mercury. A barometric
Each gas sample has the same A) density B) mass C) number of molecules D) number of atoms
 1. A real gas behaves most like an ideal gas at A) low pressure and high temperature B) average potential energy of its particles C) ionization energy of its particles D) activation energy of its particles
1. A real gas behaves most like an ideal gas at A) low pressure and high temperature B) average potential energy of its particles C) ionization energy of its particles D) activation energy of its particles
Operating Procedures for Metal Evaporator I
 Operating Procedures for Metal Evaporator I Metal Evaporator I is intended as a tool and a training device. Understanding the operation of this equipment should give you a basic knowledge of vacuum and
Operating Procedures for Metal Evaporator I Metal Evaporator I is intended as a tool and a training device. Understanding the operation of this equipment should give you a basic knowledge of vacuum and
NITROGEN GENERATION FOR INDUSTRIAL APPLICATIONS
 TRIDENT NOTES: NUMBER 5 DECEMBER 2017 NITROGEN GENERATION FOR INDUSTRIAL APPLICATIONS Industry requires nitrogen Dozens of gases are used by industry. First among these in terms of quantity consumed is
TRIDENT NOTES: NUMBER 5 DECEMBER 2017 NITROGEN GENERATION FOR INDUSTRIAL APPLICATIONS Industry requires nitrogen Dozens of gases are used by industry. First among these in terms of quantity consumed is
Oxford Instruments Austin, Inc.
 User's User s Manual Issue 01 / March 2013 / OIAI Cryo-Plex 10 Oxford Instruments Austin, Inc. Issue 01 / March 2013 / Original Instructions Oxford Instruments Austin, Inc. is a wholly-owned subsidiary
User's User s Manual Issue 01 / March 2013 / OIAI Cryo-Plex 10 Oxford Instruments Austin, Inc. Issue 01 / March 2013 / Original Instructions Oxford Instruments Austin, Inc. is a wholly-owned subsidiary
ZUERCHER TANK REGULATORS PRINCIPLES OF TANK VENTILATION
 Why Inertisation (blanketing) of Tanks? General Tank inertisation or blanketing is required in many applications where products or liquids stored in the tank are processed and/or filled into and emptied
Why Inertisation (blanketing) of Tanks? General Tank inertisation or blanketing is required in many applications where products or liquids stored in the tank are processed and/or filled into and emptied
Phase Changes * OpenStax
 OpenStax-CNX module: m42218 1 Phase Changes * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 Abstract Interpret a phase diagram. State Dalton's
OpenStax-CNX module: m42218 1 Phase Changes * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 Abstract Interpret a phase diagram. State Dalton's
Standard Operating Procedure Inert Vacuum Line. Ryan Mulvenna, May 2012
 Standard Operating Procedure Inert Vacuum Line Ryan Mulvenna, May 2012 Description of Process The inert vacuum lines located in the fume hoods one (Conjugated Polymer Synthesis: CPS) and two (ATRP polymer
Standard Operating Procedure Inert Vacuum Line Ryan Mulvenna, May 2012 Description of Process The inert vacuum lines located in the fume hoods one (Conjugated Polymer Synthesis: CPS) and two (ATRP polymer
Cryogenic Materials Operating Procedure and Safety Requirements
 VERSION 2.0 JANUARY 2, 2018 Cryogenic Materials Operating Procedure and Safety Requirements UNC CHARLOTTE 9201 UNIVERSITY CITY BLVD., CHARLOTTE, NC 28223 1 I. Overview of Liquid Nitrogen and other Cryogenic
VERSION 2.0 JANUARY 2, 2018 Cryogenic Materials Operating Procedure and Safety Requirements UNC CHARLOTTE 9201 UNIVERSITY CITY BLVD., CHARLOTTE, NC 28223 1 I. Overview of Liquid Nitrogen and other Cryogenic
Achieving Ultra-High Vacuum without (in-situ) Bakeout
 Achieving Ultra-High Vacuum without (in-situ) Bakeout Matthew Cox Diamond Light Source, UK WS 63 Ávila September 2010 1 What is Diamond Light Source? The UK national synchrotron facility 3 rd generation
Achieving Ultra-High Vacuum without (in-situ) Bakeout Matthew Cox Diamond Light Source, UK WS 63 Ávila September 2010 1 What is Diamond Light Source? The UK national synchrotron facility 3 rd generation
UV Curing in an Inerted Atmosphere Equipment Update
 UV Curing in an Inerted Atmosphere Equipment Update Jim Borsuk Senior Application Engineer Nordson UV Systems Group 09Apr14 Abstract Quality is never an accident. It is the result of intelligent effort
UV Curing in an Inerted Atmosphere Equipment Update Jim Borsuk Senior Application Engineer Nordson UV Systems Group 09Apr14 Abstract Quality is never an accident. It is the result of intelligent effort
BEST KNOWN METHODS. Transpector XPR3 Gas Analysis System. 1 of 6 DESCRIPTION XPR3 APPLICATIONS PHYSICAL INSTALLATION
 BEST KNOWN METHODS Transpector XPR3 Gas Analysis System DESCRIPTION The Transpector XPR3 is a third-generation, quadrupole-based residual gas analyzer that operates at PVD process pressures and is the
BEST KNOWN METHODS Transpector XPR3 Gas Analysis System DESCRIPTION The Transpector XPR3 is a third-generation, quadrupole-based residual gas analyzer that operates at PVD process pressures and is the
Another convenient term is gauge pressure, which is a pressure measured above barometric pressure.
 VACUUM Theory and Applications Vacuum may be defined as the complete emptiness of a given volume. It is impossible to obtain a perfect vacuum, but it is possible to obtain a level of vacuum, defined as
VACUUM Theory and Applications Vacuum may be defined as the complete emptiness of a given volume. It is impossible to obtain a perfect vacuum, but it is possible to obtain a level of vacuum, defined as
CHEM 355 EXPERIMENT 7. Viscosity of gases: Estimation of molecular diameter
 CHEM 355 EXPERIMENT 7 Viscosity of gases: Estimation of molecular diameter Expressed most simply, the viscosity of a fluid (liquid or gas) relates to its resistance to flow. The viscosity of a gas is determined
CHEM 355 EXPERIMENT 7 Viscosity of gases: Estimation of molecular diameter Expressed most simply, the viscosity of a fluid (liquid or gas) relates to its resistance to flow. The viscosity of a gas is determined
- Introduction: a) Metallized Paper & Film :The market Outlook The following diagram shows the estimated usage of the main metallized materials
 Vacuum Metallization of Paper and Outgassing Materials By Fabiano Rimediotti Galileo Vacuum Systems Prato Italy Phone : +390574564380 - f.rimediotti@galileovacuum.com Abstract Materials like paper, some
Vacuum Metallization of Paper and Outgassing Materials By Fabiano Rimediotti Galileo Vacuum Systems Prato Italy Phone : +390574564380 - f.rimediotti@galileovacuum.com Abstract Materials like paper, some
1. [Chang7 5.P.013.] Convert 295 mmhg to kpa. kpa Convert 2.0 kpa to mmhg. mmhg
![1. [Chang7 5.P.013.] Convert 295 mmhg to kpa. kpa Convert 2.0 kpa to mmhg. mmhg 1. [Chang7 5.P.013.] Convert 295 mmhg to kpa. kpa Convert 2.0 kpa to mmhg. mmhg](/thumbs/76/74310398.jpg) Score 1. [Chang7 5.P.013.] Convert 295 mmhg to kpa. kpa Convert 2.0 kpa to mmhg. mmhg 2. [Chang7 5.P.019.] The volume of a gas is 5.80 L, measured at 1.00 atm. What is the pressure of the gas in mmhg if
Score 1. [Chang7 5.P.013.] Convert 295 mmhg to kpa. kpa Convert 2.0 kpa to mmhg. mmhg 2. [Chang7 5.P.019.] The volume of a gas is 5.80 L, measured at 1.00 atm. What is the pressure of the gas in mmhg if
Oxygen Contamination
 MPMS Application Note 1014-210 Oxygen Contamination This application note describes potential sources for oxygen contamination in the sample chamber and discusses its possible effects. Molecular oxygen,
MPMS Application Note 1014-210 Oxygen Contamination This application note describes potential sources for oxygen contamination in the sample chamber and discusses its possible effects. Molecular oxygen,
Low-Power Atmospheric Gas Sampling System Based on ART MS Sensor and NEG-Ion Pump
 8 th Harsh Environment Mass Spectrometry Wokshop Low-Power Atmospheric Gas Sampling System Based on ART MS Sensor and NEG-Ion Pump Sept 22, 2011 Gerardo A. Brucker and G. Jeffery Rathbone Granville-Phillips
8 th Harsh Environment Mass Spectrometry Wokshop Low-Power Atmospheric Gas Sampling System Based on ART MS Sensor and NEG-Ion Pump Sept 22, 2011 Gerardo A. Brucker and G. Jeffery Rathbone Granville-Phillips
DEVICES FOR FIELD DETERMINATION OF WATER VAPOR IN NATURAL GAS Betsy Murphy MNM Enterprises 801 N. Riverside Drive Fort Worth, Texas 76111
 INTRODUCTION Water vapor in natural gas has more than a substantial effect on the quality of the gas stream. Without quality measurement of water vapor the gas is basically not saleable. Contracts are
INTRODUCTION Water vapor in natural gas has more than a substantial effect on the quality of the gas stream. Without quality measurement of water vapor the gas is basically not saleable. Contracts are
Question McGraw-Hill Ryerson Limited
 Question 1 Which of the following cannot be explained by considering the empty space between the particles of a gas? A) Gases are more compressible than liquids. B) Gases have lower viscosities than liquids.
Question 1 Which of the following cannot be explained by considering the empty space between the particles of a gas? A) Gases are more compressible than liquids. B) Gases have lower viscosities than liquids.
Automatic Permeability Testing: The Challenges and Solutions Author: Alyce Hartvigsen, PBI-Dansensor A/S, Ringsted, Denmark
 Automatic Permeability Testing: The Challenges and Solutions Author: Alyce Hartvigsen, PBI-Dansensor A/S, Ringsted, Denmark In recent years, numerous product developments and major changes in the distribution
Automatic Permeability Testing: The Challenges and Solutions Author: Alyce Hartvigsen, PBI-Dansensor A/S, Ringsted, Denmark In recent years, numerous product developments and major changes in the distribution
Usage Policies Notebook for Parylene Coating System
 Usage Policies Notebook for Parylene Coating System Revision date September 2014 2 Emergency Plan for Parylene Coating System Standard Operating Procedures for Emergencies Contact information Person Lab
Usage Policies Notebook for Parylene Coating System Revision date September 2014 2 Emergency Plan for Parylene Coating System Standard Operating Procedures for Emergencies Contact information Person Lab
Gas Sampling Fitting with MK2.1 and L-Probe
 measuring controlling automation measuring controlling automation measuring controlling automation Page i von 8 Operation Manual Gas Sampling Fitting with MK2.1 and L-Probe Page ii von 8 Manufacturer MESA
measuring controlling automation measuring controlling automation measuring controlling automation Page i von 8 Operation Manual Gas Sampling Fitting with MK2.1 and L-Probe Page ii von 8 Manufacturer MESA
DRAFT. Operating Procedures for the NPDGamma Liquid Hydrogen Target in TA-53, Building MPF-35
 Operating Procedures for the NPDGamma Liquid Hydrogen Target V0.03 11/26/05 1 DRAFT Operating Procedures for the NPDGamma Liquid Hydrogen Target in TA-53, Building MPF-35 Version 0.03 November 26, 2005
Operating Procedures for the NPDGamma Liquid Hydrogen Target V0.03 11/26/05 1 DRAFT Operating Procedures for the NPDGamma Liquid Hydrogen Target in TA-53, Building MPF-35 Version 0.03 November 26, 2005
Markus Hofer (Chair WHS committee) & Dianne Fisher (Safety Officer)
 Use this form to assist you to complete risk assessments for hazardous activities and processes. Any serious or ongoing hazards should be reported via RiskWare to ensure that appropriate corrective actions
Use this form to assist you to complete risk assessments for hazardous activities and processes. Any serious or ongoing hazards should be reported via RiskWare to ensure that appropriate corrective actions
CHM 111 Unit 5 Sample Questions
 Name: Class: Date: As you work these problems, consider and explain: A. What type of question is it? B. How do you know what type of question it is? C. What information are you looking for? D. What information
Name: Class: Date: As you work these problems, consider and explain: A. What type of question is it? B. How do you know what type of question it is? C. What information are you looking for? D. What information
Plasma Sources and Feedback Control in Pretreatment Web Coating Applications
 Plasma Sources and Feedback Control in Pretreatment Web Coating Applications Joseph Brindley, Benoit Daniel, Victor Bellido-Gonzalez, Dermot Monaghan Gencoa Ltd., Physics Rd, L24 9HP Liverpool, UK (+44)
Plasma Sources and Feedback Control in Pretreatment Web Coating Applications Joseph Brindley, Benoit Daniel, Victor Bellido-Gonzalez, Dermot Monaghan Gencoa Ltd., Physics Rd, L24 9HP Liverpool, UK (+44)
PSI Chemistry: Gases Multiple Choice Review
 PSI Chemistry: Gases Multiple Choice Review Name Kinetic Molecular Theory 1. According to the kinetic-molecular theory, particles of matterare in constant motion (A) have different shapes (B) have different
PSI Chemistry: Gases Multiple Choice Review Name Kinetic Molecular Theory 1. According to the kinetic-molecular theory, particles of matterare in constant motion (A) have different shapes (B) have different
Gases and Pressure SECTION 11.1
 SECTION 11.1 Gases and In the chapter States of Matter, you read about the kineticmolecular theory of matter. You were also introduced to how this theory explains some of the properties of ideal gases.
SECTION 11.1 Gases and In the chapter States of Matter, you read about the kineticmolecular theory of matter. You were also introduced to how this theory explains some of the properties of ideal gases.
States of Matter Review
 States of Matter Review May 13 8:16 PM Physical States of Matter (Phases) Solid Liquid Melting Gas Condensation Freezing Evaporation Deposition Sublimation Sep 13 6:04 PM 1 May 13 8:11 PM Gases Chapter
States of Matter Review May 13 8:16 PM Physical States of Matter (Phases) Solid Liquid Melting Gas Condensation Freezing Evaporation Deposition Sublimation Sep 13 6:04 PM 1 May 13 8:11 PM Gases Chapter
Vacuum System at IUAC
 Vacuum System at IUAC A. Mandal Inter-University Accelerator Centre, Aruna Asaf Ali Marg, Post Box - 10502, New Delhi 110067, Fax:011-26893666. email: mandal@iuac.res.in Abstract: Vacuum technology is
Vacuum System at IUAC A. Mandal Inter-University Accelerator Centre, Aruna Asaf Ali Marg, Post Box - 10502, New Delhi 110067, Fax:011-26893666. email: mandal@iuac.res.in Abstract: Vacuum technology is
Storage and transfer of cryofluids
 Storage and transfer of cryofluids Christian Gianese Guilty 1: Arsène d Arsonval (1851-1940) He invented a glass container with double wall, the vacuum being done in the space between the outer and inner
Storage and transfer of cryofluids Christian Gianese Guilty 1: Arsène d Arsonval (1851-1940) He invented a glass container with double wall, the vacuum being done in the space between the outer and inner
University of Minnesota Nano Center Standard Operating Procedure
 University of Minnesota Nano Center Standard Operating Procedure Equipment Name: Controlled Atmosphere Glove Box Model: Labconco Protector Location: PAN 185 Badger Name: Not on Badger Revision Number:
University of Minnesota Nano Center Standard Operating Procedure Equipment Name: Controlled Atmosphere Glove Box Model: Labconco Protector Location: PAN 185 Badger Name: Not on Badger Revision Number:
A NOVEL SENSOR USING REMOTE PLASMA EMISSION SPECTROSCOPY FOR MONITORING AND CONTROL OF VACUUM WEB COATING PROCESSES
 A NOVEL SENSOR USING REMOTE PLASMA EMISSION SPECTROSCOPY FOR MONITORING AND CONTROL OF VACUUM WEB COATING PROCESSES F. Papa 1, J. Brindley 2, T. Williams 2, B. Daniel 2, V. Bellido-Gonzalez 2, Dermot Monaghan
A NOVEL SENSOR USING REMOTE PLASMA EMISSION SPECTROSCOPY FOR MONITORING AND CONTROL OF VACUUM WEB COATING PROCESSES F. Papa 1, J. Brindley 2, T. Williams 2, B. Daniel 2, V. Bellido-Gonzalez 2, Dermot Monaghan
OXY Integral. INTERCON ENTERPRISES INC Tel: Fax: Internet:
 OXY Integral INTERCON ENTERPRISES INC Tel: 800 665 6655 Fax: 604 946 5340 E-Mail: sales@intercononline.com Internet: www.intercononline.com Manual Integral 2006 1 INDEX 2-3 PREFACE 4 INTRODUCTION 5 Principle
OXY Integral INTERCON ENTERPRISES INC Tel: 800 665 6655 Fax: 604 946 5340 E-Mail: sales@intercononline.com Internet: www.intercononline.com Manual Integral 2006 1 INDEX 2-3 PREFACE 4 INTRODUCTION 5 Principle
Chapter 13. Gases. Copyright Cengage Learning. All rights reserved 1
 Chapter 13 Gases Copyright Cengage Learning. All rights reserved 1 Section 13.1 Pressure Why study gases? An understanding of real world phenomena. An understanding of how science works. Copyright Cengage
Chapter 13 Gases Copyright Cengage Learning. All rights reserved 1 Section 13.1 Pressure Why study gases? An understanding of real world phenomena. An understanding of how science works. Copyright Cengage
Canadian Light Source Inc. Vacuum Component Leak Test Technical Procedure
 Canadian Light Source Inc. Vacuum Component Leak Test Technical Procedure CLSI Document 8.7.33.2 Rev. 2 Date: 2006-03-08 Copyright 2006, Canadian Light Source Inc. This document is the property of Canadian
Canadian Light Source Inc. Vacuum Component Leak Test Technical Procedure CLSI Document 8.7.33.2 Rev. 2 Date: 2006-03-08 Copyright 2006, Canadian Light Source Inc. This document is the property of Canadian
Earth s Atmosphere. Atmospheric Gases. Other Gases. Solids in the Atmosphere
 Earth s Atmosphere 1-1 I Atmospheric Gases Earth s Atmosphere extends from earth s surface to outer space. It is made up of a mixture of gases with some solids and liquids. Other Gases Water Vapor in the
Earth s Atmosphere 1-1 I Atmospheric Gases Earth s Atmosphere extends from earth s surface to outer space. It is made up of a mixture of gases with some solids and liquids. Other Gases Water Vapor in the
Vacuum Science and Technology in Accelerators
 Vacuum Science and Technology in Accelerators Ron Reid Consultant ASTeC Vacuum Science Group (ron.reid@stfc.ac.uk) Session 5 Materials Properties Relevant to Vacuum Aims To understand which mechanical
Vacuum Science and Technology in Accelerators Ron Reid Consultant ASTeC Vacuum Science Group (ron.reid@stfc.ac.uk) Session 5 Materials Properties Relevant to Vacuum Aims To understand which mechanical
Final Gas Law Review
 Name: ate: 1 t which temperature is the vapor pressure of ethanol equal to 80 kpa?. 48. 73. 80. 101 4 Gas Molecular Mass (g/mol) 2 4 17 20 The table shown lists four gases and their molecular mass. Which
Name: ate: 1 t which temperature is the vapor pressure of ethanol equal to 80 kpa?. 48. 73. 80. 101 4 Gas Molecular Mass (g/mol) 2 4 17 20 The table shown lists four gases and their molecular mass. Which
4.) There are no forces of attraction or repulsion between gas particles. This means that
 KINETIC MOLECULAR (K-M) THEORY OF MATTER NOTES - based on the idea that particles of matter are always in motion - assumptions of the K-M Theory 1.) Gases consist of large numbers of tiny particles that
KINETIC MOLECULAR (K-M) THEORY OF MATTER NOTES - based on the idea that particles of matter are always in motion - assumptions of the K-M Theory 1.) Gases consist of large numbers of tiny particles that
DOWNLOAD OR READ : HIGH PRESSURE LIQUID OXYGEN KEROSENE ENGINE COMBUSTION PDF EBOOK EPUB MOBI
 DOWNLOAD OR READ : HIGH PRESSURE LIQUID OXYGEN KEROSENE ENGINE COMBUSTION PDF EBOOK EPUB MOBI Page 1 Page 2 high pressure liquid oxygen kerosene engine combustion high pressure liquid oxygen pdf high pressure
DOWNLOAD OR READ : HIGH PRESSURE LIQUID OXYGEN KEROSENE ENGINE COMBUSTION PDF EBOOK EPUB MOBI Page 1 Page 2 high pressure liquid oxygen kerosene engine combustion high pressure liquid oxygen pdf high pressure
Characterizing a Cu/Mn Alloy for Extracting Oxygen from Inert Gas. Streams
 Characterizing a Cu/Mn Alloy for Extracting Oxygen from Inert Gas Streams Grace Lenhard Prattsburgh Central School LLE advisors: Walter Shmayda and Matthew Sharpe Laboratory for Laser Energetics University
Characterizing a Cu/Mn Alloy for Extracting Oxygen from Inert Gas Streams Grace Lenhard Prattsburgh Central School LLE advisors: Walter Shmayda and Matthew Sharpe Laboratory for Laser Energetics University
PRACTICAL TIPS FOR TRACE-LEVEL OXYGEN MONITORING WITH ELECTRO-CHEMICAL SENSORS
 APPLICATION NOTE PRACTICAL TIPS FOR TRACE-LEVEL OXYGEN MONITORING WITH ELECTRO-CHEMICAL SENSORS Improved sensor performance yields longer intervals between service, consistent on-line monitoring, and increased
APPLICATION NOTE PRACTICAL TIPS FOR TRACE-LEVEL OXYGEN MONITORING WITH ELECTRO-CHEMICAL SENSORS Improved sensor performance yields longer intervals between service, consistent on-line monitoring, and increased
Chapter 3. Solids, Liquids, and Gases
 Chapter 3 Solids, Liquids, and Gases Section 1: States of Matter Learning Objectives: Describe the characteristics of a solid Describe the characteristics of a liquid Describe the characteristics of a
Chapter 3 Solids, Liquids, and Gases Section 1: States of Matter Learning Objectives: Describe the characteristics of a solid Describe the characteristics of a liquid Describe the characteristics of a
ASPECTS OF VACUUM TECHNOLOGY. (a) To see something of the operation of vacuum pumps; Then, we will use the vacuum system for one of two experiments:
 ASPECTS OF VACUUM TECHNOLOGY In this exercise we wish (a) To see something of the operation of vacuum pumps; (b) To look at various vacuum gauges; Then, we will use the vacuum system for one of two experiments:
ASPECTS OF VACUUM TECHNOLOGY In this exercise we wish (a) To see something of the operation of vacuum pumps; (b) To look at various vacuum gauges; Then, we will use the vacuum system for one of two experiments:
MMRC PVD Evaporator Instructions
 Warnings MMRC PVD Evaporator Instructions (Updated Aug 2016) Under PVD conditions, even oils and greases are volatile. WEAR GLOVES, and if you touch your skin or anything else oily or wet, change them.
Warnings MMRC PVD Evaporator Instructions (Updated Aug 2016) Under PVD conditions, even oils and greases are volatile. WEAR GLOVES, and if you touch your skin or anything else oily or wet, change them.
Detecting Hydrogen in Helium Streams. Samuel Goodman. Pittsford Mendon High School. Rochester, New York. Advisor: Dr.
 1 Detecting Hydrogen in Helium Streams Samuel Goodman Pittsford Mendon High School Rochester, New York Advisor: Dr. Walter Shmayda Laboratory for Laser Energetics University of Rochester Rochester, New
1 Detecting Hydrogen in Helium Streams Samuel Goodman Pittsford Mendon High School Rochester, New York Advisor: Dr. Walter Shmayda Laboratory for Laser Energetics University of Rochester Rochester, New
Faculty/School: Pharmacy Initial Issue Date: Oct 2016
 Use this form to assist you to complete risk assessments for hazardous activities and processes. Any serious or ongoing hazards should be reported via RiskWare to ensure that appropriate corrective actions
Use this form to assist you to complete risk assessments for hazardous activities and processes. Any serious or ongoing hazards should be reported via RiskWare to ensure that appropriate corrective actions
Chapter 4: Moisture and Atmospheric Stability The hydrologic cycle
 Chapter 4: Moisture and Atmospheric Stability The hydrologic cycle from: USGS http://water.usgs.gov/edu/watercycle.html Evaporation: enough water to cover the entire surface of Earth to 1 meter cycles
Chapter 4: Moisture and Atmospheric Stability The hydrologic cycle from: USGS http://water.usgs.gov/edu/watercycle.html Evaporation: enough water to cover the entire surface of Earth to 1 meter cycles
Basic Concepts of Chemistry Notes for Students [Chapter 10, page 1] D J Weinkauff - Nerinx Hall High School. Chapter 10 Gases
![Basic Concepts of Chemistry Notes for Students [Chapter 10, page 1] D J Weinkauff - Nerinx Hall High School. Chapter 10 Gases Basic Concepts of Chemistry Notes for Students [Chapter 10, page 1] D J Weinkauff - Nerinx Hall High School. Chapter 10 Gases](/thumbs/78/77082586.jpg) Basic Concepts of Chemistry Notes for Students [Chapter 10, page 1] Chapter 10 Gases We have talked a little about gases in Chapter 3 and we dealt briefly with them in our stoichiometric calculations in
Basic Concepts of Chemistry Notes for Students [Chapter 10, page 1] Chapter 10 Gases We have talked a little about gases in Chapter 3 and we dealt briefly with them in our stoichiometric calculations in
Procedure for Operating Columbia (Nevis) LHe Electron Bubble Chamber Cryostat. Version 5.1
 Project: Novel Electron Bubble Particle Detector Procedure for Operating Columbia (Nevis) LHe Electron Bubble Chamber Cryostat Version 5.1 (Using side filling line) Hand Processed Changes HPC No. Date
Project: Novel Electron Bubble Particle Detector Procedure for Operating Columbia (Nevis) LHe Electron Bubble Chamber Cryostat Version 5.1 (Using side filling line) Hand Processed Changes HPC No. Date
Lecture Handout 5: Gases (Online Text Chapter 6)
 Lecture Handout 5: Gases (Online Text Chapter 6) I. The Structure of a Gas A. Gases are composed of particles that are flying around very fast in their container(s). 1. The particles travel in straight
Lecture Handout 5: Gases (Online Text Chapter 6) I. The Structure of a Gas A. Gases are composed of particles that are flying around very fast in their container(s). 1. The particles travel in straight
Section 5.1 Pressure. Why study gases? An understanding of real world phenomena. An understanding of how science works.
 Chapter 5 Gases Section 5.1 Pressure Why study gases? An understanding of real world phenomena. An understanding of how science works. Copyright Cengage Learning. All rights reserved 2 Section 5.1 Pressure
Chapter 5 Gases Section 5.1 Pressure Why study gases? An understanding of real world phenomena. An understanding of how science works. Copyright Cengage Learning. All rights reserved 2 Section 5.1 Pressure
To compare one gas to another, it is convenient to define a set of conditions: Standard Temperature and Pressure
 Standard Molar Volume To compare one gas to another, it is convenient to define a set of conditions: Standard Temperature and Pressure At STP, one mole of any gas has a volume of: 22.4 L = (This is a cube
Standard Molar Volume To compare one gas to another, it is convenient to define a set of conditions: Standard Temperature and Pressure At STP, one mole of any gas has a volume of: 22.4 L = (This is a cube
Plasma Cleaner. Yamato Scientific America. Contents. Innovating Science for Over 125 Years. Gas Plasma Dry Cleaner PDC200/210/510 PDC610G.
 Yamato Scientific America Innovating Science for Over 125 Years Plasma Cleaner Contents Gas Plasma Dry Cleaner PDC200/210/510 PDC610G Gas Plasma Reactor 145 146 147 149 144 Gas Plasma Dry Cleaner Plasma
Yamato Scientific America Innovating Science for Over 125 Years Plasma Cleaner Contents Gas Plasma Dry Cleaner PDC200/210/510 PDC610G Gas Plasma Reactor 145 146 147 149 144 Gas Plasma Dry Cleaner Plasma
RC102 & RC110 Sample in Vacuum Continuous Flow Cryogenic Workstation Cryostats
 RC102 & RC110 Sample in Vacuum Continuous Flow Cryogenic Workstation Cryostats www.cryoindustries.com INDUSTRIES RC102 & RC110 Sample in Vacuum Continuous Flow Cryogenic Workstation Cryostats Designed
RC102 & RC110 Sample in Vacuum Continuous Flow Cryogenic Workstation Cryostats www.cryoindustries.com INDUSTRIES RC102 & RC110 Sample in Vacuum Continuous Flow Cryogenic Workstation Cryostats Designed
Chemistry Chapter 12. Characteristics of Gases. Characteristics of Gases 1/31/2012. Gases and Liquids
 Importance of Gases Chemistry Chapter 12 Gases and Liquids Airbags fill with N 2 gas in an accident. Gas is generated by the decomposition of sodium azide, NaN 3. 2 NaN 3 ---> 2 Na + 3 N 2 THREE STATES
Importance of Gases Chemistry Chapter 12 Gases and Liquids Airbags fill with N 2 gas in an accident. Gas is generated by the decomposition of sodium azide, NaN 3. 2 NaN 3 ---> 2 Na + 3 N 2 THREE STATES
Name Hour. The Behavior of Gases. Practice B
 Name Hour The Behavior of Gases Practice B B 1 Objective 1: Apply Boyle s Law, Charles s Law, and Gay-Lussac s Law to solve problems involving pressure and volume and temperature. 1. A high-altitude balloon
Name Hour The Behavior of Gases Practice B B 1 Objective 1: Apply Boyle s Law, Charles s Law, and Gay-Lussac s Law to solve problems involving pressure and volume and temperature. 1. A high-altitude balloon
Name Gas Law Date. Version 3
 Name Gas Law Date 1. A real gas behaves least like an ideal gas under the conditions of 1) low temperature and high pressure 2) high temperature and low pressure 3) high temperature and high pressure 4)
Name Gas Law Date 1. A real gas behaves least like an ideal gas under the conditions of 1) low temperature and high pressure 2) high temperature and low pressure 3) high temperature and high pressure 4)
Chemistry Honors - Gases
 Name: Class: Date: ID: A Chemistry Honors - Gases Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Why does a can collapse when a vacuum pump removes air
Name: Class: Date: ID: A Chemistry Honors - Gases Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Why does a can collapse when a vacuum pump removes air
EXERCISE 2: THE VACUUM SYSTEM
 EXERCISE 2: THE VACUUM SYSTEM Because vacuum system techniques are important to all of experimental physics, every undergraduate student should have had some experience in them. This exercise ensures that
EXERCISE 2: THE VACUUM SYSTEM Because vacuum system techniques are important to all of experimental physics, every undergraduate student should have had some experience in them. This exercise ensures that
