The units are determined by the standard states used in the Table: NH 3 (g) NH 3 (aq), means Q =[CO 2]/ p CO2 = M/bar.
|
|
|
- Vernon Atkins
- 6 years ago
- Views:
Transcription
1 CHMY 36 Nov 5, 26 6 problems on 2 pages HOMEWOR #4: SOLUIONS. (a) From able A.5 (back of text), calculate the Henry s Law constant (i.e., equilibrium constant) for dissolving CO2(g) in water (i.e., solubility) at 25 o C. What units does this equilibrium constant have? (b) Do the same as (a) at 37 o C (c) Convert the Henry s Law constants you obtained in (a) and (b) to the units of able 6. (bars/mol fraction) and compare with the numbers found in able 6. G f from A.5 H f kj/mol kj/mol CO 2(aq) CO 2(g) aq gas diff (a) 298=exp(-G /R) = exp(-85/(8.345*298) =.322 moll - bar - he units are determined by the standard states used in the able: NH 3 (g) NH 3 (aq), means Q =[CO 2]/ p CO2 = M/bar. (b)lechatlier says exothermic reactions shift left with increasing (i.e., get smaller, i.e., the solubility of all gases is smaller at higher temperatures). herefore H is absolutely required to find the at 3. (G changes as changes, so that just changing only from 298 to 3 in the above equation is wrong, and worth no credit.) Use Van t Hoff equation for temperature dependence of all equilibrium constants (e.g., vapor pressure, Henry Law constants, solubilities, partition coefficients, etc): 3 H R exp(.37).235m/bar 3 Note: a simple sign error will result in a higher value than 298.You should recognize that this is wrong by LeChatlier s principle, and find the problem. (c) First turn the upside down because the reaction is reversed. for evaporation = Because L of liquid water contains g/8 = mol, a solution has a mole fraction = /.322 =3.2 bar/m. XCO2 = [CO2] / ([CO2]+55.55). hus,.322 moll - becomes.322/( ) =.5793 mol fraction. 298 = 3.2 bar/(m *.5793 mol fract/.322m) = 725 bar/molefraction, which is not too far from 59 bar/molfraction in able 6.. Page of 5
2 At 3, mole fraction changes to.235/( ) =.4229 and 3 = /.235 = bar/m. his becomes *.235/.4229 = 2369 bar/mol fraction compared to 23 in the table. 2. A 4 lb person contains about 5 L of water (about 4/5 of which is intracellular). Assuming all of this water is saturated with dissolved N2 gas, answer the following questions: (a) How many mols of N2 gas are dissolved in a person when the air pressure = bar (assuming that the solubility is the same as in pure water? (Use able 6. for 37 o C.) (b) How many liters of gas at bar and 3 does this number of mols equal? (c) atmosphere = 76 mm of Hg, and the density of Hg is 3.6 times that of water. What is bar in units of meters of water, assuming the density of water is. kg/l? (d) If a diver stays at a depth of 4 m in water until equilibrium is reached, how many liters of N2 gas would potentially be released in the form of microbubbles if the diver came to the surface very quickly? (Remember, Henry s Law is just saying that the amount of a particular gas dissolved is directly proportional to the partial pressure of that gas in equilibrium with the liquid.) (a) he partial pressure of N2 is.78 bar for bar air pressure assumed at sea level. he amount dissolved is /(98 x 3 ) molefraction/bar x.78 bar = mol fraction = 7.96 x -6. he moles N2 dissolved = 7.96 x -6 mol N2/(mol N2 + mol water) = very closely 7.96 x -6 * 5 L water x 55.5 mol water/l water) =.22 mol N2 gas dissolved. (b) = pv = nr V=nR/p =.22 x.8345 x 3/ =.57 L N2 gas. (c) bar = bar /(.3 bar/atm) x.76 mhg/atm x 3.6 m H2O/m Hg =.2 m H2O (d) 4 m H2O / (.2 m H2O bar - ) = 3.92 bar of additional air pressure. his means 3.92 *.78 = 3.6 additional pn2 which equates to 3.6 x.57 =.74 L of bubbles that would be produced upon coming to the surface. 3. Consider a x -5 M polymer solution inside a dialysis bag that is permeable to the ligand, A, but not to the polymer. he following measurements of the free ligand outside the bag and total ligand inside the bag are measured at equilibrium. he units are M. From a Scatchard plot find d for the binding of A to its binding sites on the polymer and the number of binding sites per polymer molecule. (the notation 5E-7 means 5 x -7, etc.) [A]out [A]in total [A bound]=sa SA/A 5.E-7 5.5E-5 5.E-5.E+2.E-7 3.E-5 3.E-5 3.E+2 2.E-8.E E E+2 Plot of [SA] vs [A] has slope = -E7 = -/d and intercept = = [otal Sites]/d = Stot/d [otal Sites] = * E-7 = 6 x -5 M [Polymer] = e-5 M Sites/polymer = 6 x -5 / x -5 = 6 Plot of [SA] vs [A] 6.E+2 4.E+2 2.E+2 y = -E+7x E+.E+ 2.E-5 4.E-5 6.E-5 Page 2 of 5
3 4. he partition coefficient of solute B between water and ethanol cannot be directly measured because water and ethanol are completely miscible. (a) Calculate this partition coefficient ([B(aq)]/ [B(ethanol)]) if the solubility of B in water =. M and the solubility of B in ethanol = 5. M at 25 o C. From the data in (a), calculate the following: (b) What is ΔG o for the process ([B(aq)]/ [B(ethanol)]? (c) What is ΔG o for the process B(aq) --> B(s)? (a) Solubility = for B(s) --> B(aq) = [B(aq)]/X B(s) =. M/ for pure solid B Solubility = for B(s) --> B(ethanol) = [B(ethanol)]/X B(s) =5 M/ for pure solid B Partition Coef. [B(aq)]/ [B(ethanol)] = for B(ethanol) B(aq) = ([B(aq)]/X B(s))/ [B(ethanol)]/X B(s) )=./5 =.2 (b) ΔG o = -R = *298(.2) = 9693 (c) his is reverse of solubility so = /. = ΔG o = -R = *298() = -575 J/mol 5. Suppose in certain mitochondria, the oxidation of glucose products pumps protons into the intermembrane space between the double membrane to reach a ph of 5.. In the interior of the mitochondrion, the ph is 7.. In addition, the intermembrane space has an electric potential difference of +75 mv relative to the interior. AP is synthesized from the combined Gibbs energy change per mol of protons (Δ) flowing down the concentration gradient from the intermembrane space (out) to the interior (in) and from the change of electrical free energy coming from the voltage difference. (a)what is Δ for H + (out) --> H + (in) for this mitochondrion? (b) Calculate the ΔG if a total of 4 moles of protons are transferred. (c) What is the maximum number of moles of AP that can be obtained from ADP and P at ph 7 when 4 moles of protons are transferred if [AP]/[ADP] = 2. and [P].5 M? (a) C R C in out zf ( 7. in out ,5coulombs ( (.75J / coulomb) J ) (b) ΔG = 4 x Δ =4 x (-8649) = -74,595 J Page 3 of 5
4 (c) AP reaction = ADP + Pi AP ΔG = +3, J ΔGproton-transfer = n protons Δproton-transfer ΔGAP = ΔG + RQ/Q = 3, + R(2./.5) = +45,845 J/mol (at ph 7 the Q cancels [H + ] in Q) Maximum will be when reversible, i.e., at equilibrium, i.e., when ΔGtotal = ΔGproton-transfer + ΔGAP =. moles protons x (-8649) ) = moles protons =45845/8649 = 2.46 on average 6. he osmotic pressure of sea water is known to be 25 bar at. Using this number: (a) what is the activity of water in sea water at? (b) what is the vapor pressure of sea water at, given that for pure water it is 62 Pa? (c) what is the melting point of pure water ice in seawater? (d) what is the point of this sea water, assuming the activity is the same as at.? (a) a - exp( V R m 25.bar.8L ) exp( ) (b) Using Rauolt s Law, ph2o(g) / ah2o(l) = vapor pressure of pure liquid = 62 Pa, (i.e., the equilibrium constant for H2O(l,a)H2O(g, psea water ) : psea water = 62 *.98 = 6. Pa (c) he reaction is H2O(s,pure) H2O(l,a) Q = a/. At, this is spontaneous to right because a <. (salt lowers the melting point) o reach equilibrium we must decrease the temperature to the melting point of the sea water i.e., make the process less spontaneous until equilibrium is reached, i.e., until the equilibrium constant = a/. So, use Vant Hoff with H fusion = 67 J/mol m a H R fusion melting melting melting Page 4 of 5
5 (d) Similarly, for the point raising: he reaction is H2O(l,a) H2O(g, ph2ogas) Q = /a he point = with applied pressure = bar because the vapor pressure of pure water =. bar at this temperature. For sea water at, this is spontaneous to LEF because the applied pressure = at sea level, causing ph2o in any bubbles in the water to also =, which exceeds the vapor pressure of the sea water, which has vapor pressure less than because of the salt as shown in 6.(b). o reach equilibrium we must increase the temperature to make the process more spontaneous until the equilibrium constant = /a, i.e., so that the vapor pressure of the solution =. bar = the applied pressure. Again, use Vant Hoff with H vaporization = 4,657 J/mol bp a H vaporazation R , Page 5 of 5
Figure Vapor-liquid equilibrium for a binary mixture. The dashed lines show the equilibrium compositions.
 Another way to view this problem is to say that the final volume contains V m 3 of alcohol at 5.93 kpa and 20 C V m 3 of air at 94.07 kpa and 20 C V m 3 of air plus alcohol at 100 kpa and 20 C Thus, the
Another way to view this problem is to say that the final volume contains V m 3 of alcohol at 5.93 kpa and 20 C V m 3 of air at 94.07 kpa and 20 C V m 3 of air plus alcohol at 100 kpa and 20 C Thus, the
CHEM1901/3 Worksheet 8: The Ideal Gas Law: PV = nrt
 CHEM1901/3 Worksheet 8: The Ideal Gas Law: PV = nrt The Ideal Gas Law Model 1: The Gas Laws T (K) Kelvin or absolute temperature = T ( C) + 273. T(K) is always 0 K Boyle s Law (1660). The volume of a gas
CHEM1901/3 Worksheet 8: The Ideal Gas Law: PV = nrt The Ideal Gas Law Model 1: The Gas Laws T (K) Kelvin or absolute temperature = T ( C) + 273. T(K) is always 0 K Boyle s Law (1660). The volume of a gas
1. [Chang7 5.P.013.] Convert 295 mmhg to kpa. kpa Convert 2.0 kpa to mmhg. mmhg
![1. [Chang7 5.P.013.] Convert 295 mmhg to kpa. kpa Convert 2.0 kpa to mmhg. mmhg 1. [Chang7 5.P.013.] Convert 295 mmhg to kpa. kpa Convert 2.0 kpa to mmhg. mmhg](/thumbs/76/74310398.jpg) Score 1. [Chang7 5.P.013.] Convert 295 mmhg to kpa. kpa Convert 2.0 kpa to mmhg. mmhg 2. [Chang7 5.P.019.] The volume of a gas is 5.80 L, measured at 1.00 atm. What is the pressure of the gas in mmhg if
Score 1. [Chang7 5.P.013.] Convert 295 mmhg to kpa. kpa Convert 2.0 kpa to mmhg. mmhg 2. [Chang7 5.P.019.] The volume of a gas is 5.80 L, measured at 1.00 atm. What is the pressure of the gas in mmhg if
Gases. Chapter 5: Gas Laws Demonstration. September 10, Chapter 5 Gasses.notebook. Dec 18 10:23 AM. Jan 1 4:11 PM. Crushing 55 gallon drum
 Chapter 5: Gases Dec 18 10:23 AM Gas Laws Demonstration Crushing 55 gallon drum Egg in a bottle Student in a bag Boiling Water Charles gas Law Water in a flask Ballon in a bottle Jan 1 4:11 PM 1 5.1 Pressure
Chapter 5: Gases Dec 18 10:23 AM Gas Laws Demonstration Crushing 55 gallon drum Egg in a bottle Student in a bag Boiling Water Charles gas Law Water in a flask Ballon in a bottle Jan 1 4:11 PM 1 5.1 Pressure
Example: 25 C = ( ) K = 298 K. Pressure Symbol: p Units: force per area 1Pa (Pascal) = 1 N/m 2
 Chapter 6: Gases 6.1 Measurements on Gases MH5, Chapter 5.1 Let s look at a certain amount of gas, i.e. trapped inside a balloon. To completely describe the state of this gas one has to specify the following
Chapter 6: Gases 6.1 Measurements on Gases MH5, Chapter 5.1 Let s look at a certain amount of gas, i.e. trapped inside a balloon. To completely describe the state of this gas one has to specify the following
Chapter 5. Pressure. Atmospheric Pressure. Gases. Force Pressure = Area
 Chapter 5 Gases Water for many homes is supplied by a well The pump removes air from the pipe, decreasing the air pressure in the pipe The pressure then pushes the water up the pipe Pressure Atmospheric
Chapter 5 Gases Water for many homes is supplied by a well The pump removes air from the pipe, decreasing the air pressure in the pipe The pressure then pushes the water up the pipe Pressure Atmospheric
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Test General Chemistry CH116 UMass Boston Summer 2013 Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) The pressure exerted by a column of
Test General Chemistry CH116 UMass Boston Summer 2013 Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) The pressure exerted by a column of
Chapter 13 Gases, Vapors, Liquids, and Solids
 Chapter 13 Gases, Vapors, Liquids, and Solids Property is meaning any measurable characteristic of a substance, such as pressure, volume, or temperature, or a characteristic that can be calculated or deduced,
Chapter 13 Gases, Vapors, Liquids, and Solids Property is meaning any measurable characteristic of a substance, such as pressure, volume, or temperature, or a characteristic that can be calculated or deduced,
Chemistry 101 Chapter 5 GAS MIXTURES
 GAS MIXTURES Consider mixing equal volumes of 3 different gases, all at the same temperature and pressure in a container of the same size. 1 L He 1 L N 2 1 L O 2 1 L mixture t = 0 0 C t = 0 0 C t = 0 0
GAS MIXTURES Consider mixing equal volumes of 3 different gases, all at the same temperature and pressure in a container of the same size. 1 L He 1 L N 2 1 L O 2 1 L mixture t = 0 0 C t = 0 0 C t = 0 0
Name /74. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Ch 11 Gases STUDY GUIDE Accelerated Chemistry SCANTRON Name /74 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following statements
Ch 11 Gases STUDY GUIDE Accelerated Chemistry SCANTRON Name /74 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following statements
Gas Law Worksheets - WS: Boyle s and Charles Law
 Gas Law Worksheets - WS: Boyle s and Charles Law Boyle s Law states that the volume of a gas varies inversely with its pressure if temperature is held constant. (If one goes up the, other goes down.) We
Gas Law Worksheets - WS: Boyle s and Charles Law Boyle s Law states that the volume of a gas varies inversely with its pressure if temperature is held constant. (If one goes up the, other goes down.) We
Gilbert Kirss Foster. Chapter 10. Properties of Gases The Air We Breathe
 Gilbert Kirss Foster Chapter 10 Properties of Gases The Air We Breathe Chapter Outline 10.1 The Properties of Gases 10.2 Effusion and the Kinetic Molecular Theory of Gases 10.3 Atmospheric Pressure 10.4
Gilbert Kirss Foster Chapter 10 Properties of Gases The Air We Breathe Chapter Outline 10.1 The Properties of Gases 10.2 Effusion and the Kinetic Molecular Theory of Gases 10.3 Atmospheric Pressure 10.4
Section 8.1 Properties of Gases Goal: Describe the kinetic molecular theory of gases and the units of measurement used for gases.
 Chapter 8 Gases Practice Problems Section 8.1 Properties of Gases Goal: Describe the kinetic molecular theory of gases and the units of measurement used for gases. Summary: In a gas, particles are so far
Chapter 8 Gases Practice Problems Section 8.1 Properties of Gases Goal: Describe the kinetic molecular theory of gases and the units of measurement used for gases. Summary: In a gas, particles are so far
You should be able to: Describe Equipment Barometer Manometer. 5.1 Pressure Read and outline 5.1 Define Barometer
 A P CHEMISTRY - Unit 5: Gases Unit 5: Gases Gases are distinguished from other forms of matter, not only by their power of indefinite expansion so as to fill any vessel, however large, and by the great
A P CHEMISTRY - Unit 5: Gases Unit 5: Gases Gases are distinguished from other forms of matter, not only by their power of indefinite expansion so as to fill any vessel, however large, and by the great
Chapter 13 Gases and Pressure. Pressure and Force. Pressure is the force per unit area on a surface. Force Area. Pressure =
 Chapter 13 Gas Laws Chapter 13 Gases and Pressure Pressure and Force Pressure is the force per unit area on a surface. Pressure = Force Area Chapter 13 Gases and Pressure Gases in the Atmosphere The atmosphere
Chapter 13 Gas Laws Chapter 13 Gases and Pressure Pressure and Force Pressure is the force per unit area on a surface. Pressure = Force Area Chapter 13 Gases and Pressure Gases in the Atmosphere The atmosphere
Elements that exist as gases at 25 o C and 1 atmosphere H 2, N 2, O 2, F 2, Cl 2, He, Ne, Ar, Kr, Xe, Rn
 AP Chemistry Chapter 5 Sections 5. 5.9 Note Organizer Pressure, The Gas Laws of Boyle, Charles, and Avogadro, The Ideal Gas Law, Gas Stoichiometry, Dalton s Law of Partial Pressure, The Kinetic olecular
AP Chemistry Chapter 5 Sections 5. 5.9 Note Organizer Pressure, The Gas Laws of Boyle, Charles, and Avogadro, The Ideal Gas Law, Gas Stoichiometry, Dalton s Law of Partial Pressure, The Kinetic olecular
AP TOPIC 6: Gases. Revised August General properties and kinetic theory
 AP OPIC 6: Gases General properties and kinetic theory Gases are made up of particles that have (relatively) large amounts of energy. A gas has no definite shape or volume and will expand to fill as much
AP OPIC 6: Gases General properties and kinetic theory Gases are made up of particles that have (relatively) large amounts of energy. A gas has no definite shape or volume and will expand to fill as much
Judith Herzfeld 1996,1998. These exercises are provided here for classroom and study use only. All other uses are copyright protected.
 Judith Herzfeld 1996,1998 These exercises are provided here for classroom and study use only. All other uses are copyright protected. 2.7-110 A yardstick laid on a table with a few inches hanging over
Judith Herzfeld 1996,1998 These exercises are provided here for classroom and study use only. All other uses are copyright protected. 2.7-110 A yardstick laid on a table with a few inches hanging over
States of Matter Review
 States of Matter Review May 13 8:16 PM Physical States of Matter (Phases) Solid Liquid Melting Gas Condensation Freezing Evaporation Deposition Sublimation Sep 13 6:04 PM 1 May 13 8:11 PM Gases Chapter
States of Matter Review May 13 8:16 PM Physical States of Matter (Phases) Solid Liquid Melting Gas Condensation Freezing Evaporation Deposition Sublimation Sep 13 6:04 PM 1 May 13 8:11 PM Gases Chapter
Chapter 10: Properties of Gases: The Air We Breathe
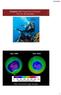 Chapter 10: Properties of Gases: The Air We Breathe Sept, 2006 Sept, 2016 http://ozonewatch.gsfc.nasa.gov 1 Chapter Outline 10.1 The Properties of Gases 10.2 The Kinetic Molecular Theory of Gases* 10.3
Chapter 10: Properties of Gases: The Air We Breathe Sept, 2006 Sept, 2016 http://ozonewatch.gsfc.nasa.gov 1 Chapter Outline 10.1 The Properties of Gases 10.2 The Kinetic Molecular Theory of Gases* 10.3
Physical Chemistry of Gases: Gas Exchange Linda Costanzo, Ph.D.
 Physical Chemistry of Gases: Gas Exchange Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. Application of the gas laws to pulmonary physiology. 2. How to
Physical Chemistry of Gases: Gas Exchange Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. Application of the gas laws to pulmonary physiology. 2. How to
Kinetic-Molecular Theory of Matter
 Gases Properties of Gases Gas Pressure Gases What gases are important for each of the following: O 2, CO 2 and/or He? A. B. C. D. 1 2 Gases What gases are important for each of the following: O 2, CO 2
Gases Properties of Gases Gas Pressure Gases What gases are important for each of the following: O 2, CO 2 and/or He? A. B. C. D. 1 2 Gases What gases are important for each of the following: O 2, CO 2
Chapter 10 Gases. Characteristics of Gases. Pressure. The Gas Laws. The Ideal-Gas Equation. Applications of the Ideal-Gas Equation
 Characteristics of Gases Chapter 10 Gases Pressure The Gas Laws The Ideal-Gas Equation Applications of the Ideal-Gas Equation Gas mixtures and partial pressures Kinetic-Molecular Theory Real Gases: Deviations
Characteristics of Gases Chapter 10 Gases Pressure The Gas Laws The Ideal-Gas Equation Applications of the Ideal-Gas Equation Gas mixtures and partial pressures Kinetic-Molecular Theory Real Gases: Deviations
Chem 110 General Principles of Chemistry
 CHEM110 Worksheet - Gases Chem 110 General Principles of Chemistry Chapter 9 Gases (pages 337-373) In this chapter we - first contrast gases with liquids and solids and then discuss gas pressure. - review
CHEM110 Worksheet - Gases Chem 110 General Principles of Chemistry Chapter 9 Gases (pages 337-373) In this chapter we - first contrast gases with liquids and solids and then discuss gas pressure. - review
Ch. 11 Mass transfer principles
 Transport of chemical species in solid, liquid, or gas mixture Transport driven by composition gradient, similar to temperature gradients driving heat transport We will look at two mass transport mechanisms,
Transport of chemical species in solid, liquid, or gas mixture Transport driven by composition gradient, similar to temperature gradients driving heat transport We will look at two mass transport mechanisms,
Under pressure pushing down
 Under pressure pushing down on me When Dalton was conducting his studies, which led him to the atomic-molecular theory of matter, he also included studies of the behaviour of gases. These led him to propose,
Under pressure pushing down on me When Dalton was conducting his studies, which led him to the atomic-molecular theory of matter, he also included studies of the behaviour of gases. These led him to propose,
More Practice with Gas Laws KEY
 More Practice with Gas Laws KEY Chemistry Directions: For each question, identify the applicable law and solve for the correct answer using dimensional analysis. Express your answer to the correct number
More Practice with Gas Laws KEY Chemistry Directions: For each question, identify the applicable law and solve for the correct answer using dimensional analysis. Express your answer to the correct number
Topic 6: Gases and Colligative Properties
 Topic 6: Gases and Colligative Properties Ideal Gas Equation Boyle noticed an inverse relationship between volume and pressure. Pressure x volume = constant P = a P 1/P Charles found the volume of a gas,
Topic 6: Gases and Colligative Properties Ideal Gas Equation Boyle noticed an inverse relationship between volume and pressure. Pressure x volume = constant P = a P 1/P Charles found the volume of a gas,
Applied Physics Topics 2
 Applied Physics Topics 2 Dr Andrey Varvinskiy Consultant Anaesthetist Torbay Hospital, UK EDAIC Paper B Lead and Examiner TOPICS 2 Gas Laws Other Laws: Dalton, Avogadro Critical temperature Critical pressure
Applied Physics Topics 2 Dr Andrey Varvinskiy Consultant Anaesthetist Torbay Hospital, UK EDAIC Paper B Lead and Examiner TOPICS 2 Gas Laws Other Laws: Dalton, Avogadro Critical temperature Critical pressure
4. Using the kinetic molecular theory, explain why a gas can be easily compressed, while a liquid and a solid cannot?
 Name Period HW 1 Worksheet (Goals 1-4) - Kinetic Molecular Theory 1. Describe how gases, liquids, and solids compare using the following table. Solids Liquids Gases Volume (definite or indefinite) Molecular
Name Period HW 1 Worksheet (Goals 1-4) - Kinetic Molecular Theory 1. Describe how gases, liquids, and solids compare using the following table. Solids Liquids Gases Volume (definite or indefinite) Molecular
temperature and pressure unchanging
 Gas Laws Review I. Variables Used to Describe a Gas A. Pressure (P) kpa, atm, mmhg (torr) -Pressure=force exerted per unit area (force/area) -Generated by collisions within container walls (more collisions=more
Gas Laws Review I. Variables Used to Describe a Gas A. Pressure (P) kpa, atm, mmhg (torr) -Pressure=force exerted per unit area (force/area) -Generated by collisions within container walls (more collisions=more
PROPERTIES OF GASES. [MH5; Ch 5, (only)]
![PROPERTIES OF GASES. [MH5; Ch 5, (only)] PROPERTIES OF GASES. [MH5; Ch 5, (only)]](/thumbs/76/74446950.jpg) PROPERTIES OF GASES [MH5; Ch 5, 5.1-5.5 (only)] FEATURES OF A GAS Molecules in a gas are a long way apart (under normal conditions). Molecules in a gas are in rapid motion in all directions. The forces
PROPERTIES OF GASES [MH5; Ch 5, 5.1-5.5 (only)] FEATURES OF A GAS Molecules in a gas are a long way apart (under normal conditions). Molecules in a gas are in rapid motion in all directions. The forces
Lecture Handout 5: Gases (Online Text Chapter 6)
 Lecture Handout 5: Gases (Online Text Chapter 6) I. The Structure of a Gas A. Gases are composed of particles that are flying around very fast in their container(s). 1. The particles travel in straight
Lecture Handout 5: Gases (Online Text Chapter 6) I. The Structure of a Gas A. Gases are composed of particles that are flying around very fast in their container(s). 1. The particles travel in straight
Chapter 10: Gases. Characteristics of Gases
 Chapter 10: Gases Learning Outcomes: Calculate pressure and convert between pressure units with an emphasis on torr and atmospheres. Calculate P, V, n, or T using the ideal-gas equation. Explain how the
Chapter 10: Gases Learning Outcomes: Calculate pressure and convert between pressure units with an emphasis on torr and atmospheres. Calculate P, V, n, or T using the ideal-gas equation. Explain how the
Kinetic Molecular Theory imaginary Assumptions of Kinetic Molecular Theory: Problems with KMT:
 AP Chemistry Ms. Ye Name Date Block Kinetic Molecular Theory Explains properties of gases, liquids, and solids in terms of energy using an ideal gas, an imaginary which fits all the assumptions of kinetic
AP Chemistry Ms. Ye Name Date Block Kinetic Molecular Theory Explains properties of gases, liquids, and solids in terms of energy using an ideal gas, an imaginary which fits all the assumptions of kinetic
Gas Laws V 1 V 2 T 1. Gas Laws.notebook. May 05, T = k P 1 V 1 = P 2 V 2. = 70 kpa. P. V = k. k = 1 atm = kpa
 Gas Laws At constant temperature, all gases behave the same when compressed As increasing pressure is applied to a gas in a closed container, the volume of the gas decreases he product of pressure and
Gas Laws At constant temperature, all gases behave the same when compressed As increasing pressure is applied to a gas in a closed container, the volume of the gas decreases he product of pressure and
2. Calculate the ratio of diffusion rates for carbon monoxide (CO) and carbon dioxide (CO2). υa = MB = 44 = 1.25
 Gas laws worksheet (2-08) (modified 3/17) Answer key Graham s Law 1. Calculate the ratio of effusion rates for nitrogen (N2) and neon (Ne). υa = MB = 20 = 0.845 υb MA 28 2. Calculate the ratio of diffusion
Gas laws worksheet (2-08) (modified 3/17) Answer key Graham s Law 1. Calculate the ratio of effusion rates for nitrogen (N2) and neon (Ne). υa = MB = 20 = 0.845 υb MA 28 2. Calculate the ratio of diffusion
Chapter 11. Recall: States of Matter. Properties of Gases. Gases
 Chapter 11 Gases Recall: States of Matter Solids and Liquids: are closely related because in each case the particles are interacting with each other Gases: Properties of Gases Gases can be compressed Gases
Chapter 11 Gases Recall: States of Matter Solids and Liquids: are closely related because in each case the particles are interacting with each other Gases: Properties of Gases Gases can be compressed Gases
Chemistry Chapter 12. Characteristics of Gases. Characteristics of Gases 1/31/2012. Gases and Liquids
 Importance of Gases Chemistry Chapter 12 Gases and Liquids Airbags fill with N 2 gas in an accident. Gas is generated by the decomposition of sodium azide, NaN 3. 2 NaN 3 ---> 2 Na + 3 N 2 THREE STATES
Importance of Gases Chemistry Chapter 12 Gases and Liquids Airbags fill with N 2 gas in an accident. Gas is generated by the decomposition of sodium azide, NaN 3. 2 NaN 3 ---> 2 Na + 3 N 2 THREE STATES
Chapter 5: Gases 5.1 Pressure Why study gases? An understanding of real world phenomena. An understanding of how science works.
 Chapter 5: Gases 5.1 Pressure Why study gases? An understanding of real world phenomena. An understanding of how science works. A Gas Uniformly fills any container. Easily compressed. Mixes completely
Chapter 5: Gases 5.1 Pressure Why study gases? An understanding of real world phenomena. An understanding of how science works. A Gas Uniformly fills any container. Easily compressed. Mixes completely
Chapter 13. Gases. Copyright Cengage Learning. All rights reserved 1
 Chapter 13 Gases Copyright Cengage Learning. All rights reserved 1 Section 13.1 Pressure Why study gases? An understanding of real world phenomena. An understanding of how science works. Copyright Cengage
Chapter 13 Gases Copyright Cengage Learning. All rights reserved 1 Section 13.1 Pressure Why study gases? An understanding of real world phenomena. An understanding of how science works. Copyright Cengage
Name Unit 9 Notes: Gas Laws Period. Complete throughout unit. Due on test day!
 Name Unit 9 Notes: Gas Laws Period Skills: 1. Gases and Entropy 2. Distinguish between Ideal and Real gases 3. Understand KMT and Avogadro s Law 4. Identify and Solve Boyle s Law Problems 5. Identify and
Name Unit 9 Notes: Gas Laws Period Skills: 1. Gases and Entropy 2. Distinguish between Ideal and Real gases 3. Understand KMT and Avogadro s Law 4. Identify and Solve Boyle s Law Problems 5. Identify and
Gases and Respiration. Respiration Overview I
 Respiration Overview I Respiration Overview II Gas Laws Equation of State: PV = nrt Same volumes of different gases have same # of molecules BTPS: body temp, atmospheric pressure, saturated ATPS: ambient
Respiration Overview I Respiration Overview II Gas Laws Equation of State: PV = nrt Same volumes of different gases have same # of molecules BTPS: body temp, atmospheric pressure, saturated ATPS: ambient
Unit 8: Gases and States of Matter
 Unit 8: Gases and States of Matter Gases Particles that have no definite shape or volume. They adapt to the shape and volume of their container. Ideal gases are imaginary gases that comply with all the
Unit 8: Gases and States of Matter Gases Particles that have no definite shape or volume. They adapt to the shape and volume of their container. Ideal gases are imaginary gases that comply with all the
AN OVERVIEW OF RESPIRATION AND AN INTRODUCTION TO DIFFUSION AND SOLUBILITY OF GASES 1
 AN OVERVIEW OF RESPIRATION AND AN INTRODUCTION TO DIFFUSION AND SOLUBILITY OF GASES 1 Summary: This set of notes gives an overview of respiration and then follows the overview with a detailed discussion
AN OVERVIEW OF RESPIRATION AND AN INTRODUCTION TO DIFFUSION AND SOLUBILITY OF GASES 1 Summary: This set of notes gives an overview of respiration and then follows the overview with a detailed discussion
1. A pure substance has a specific volume of 0.08 L/mol at a pressure of 3 atm and 298 K. The substance is most likely:
 Name: September 19, 2014 EXAM 1 P a g e 1 1. A pure substance has a specific volume of 0.08 L/mol at a pressure of 3 atm and 298 K. The substance is most likely: a. Liquid b. Gas c. Supercritical Fluid
Name: September 19, 2014 EXAM 1 P a g e 1 1. A pure substance has a specific volume of 0.08 L/mol at a pressure of 3 atm and 298 K. The substance is most likely: a. Liquid b. Gas c. Supercritical Fluid
PHYSICS. Light: Sound:
 PHYSICS Light: The speed of light changes as it passes through different things such as air, glass and water. This affects the way we see things underwater with a diving mask. As the light passes through
PHYSICS Light: The speed of light changes as it passes through different things such as air, glass and water. This affects the way we see things underwater with a diving mask. As the light passes through
Chapter 17 The Respiratory System: Gas Exchange and Regulation of Breathing
 Chapter 17 The Respiratory System: Gas Exchange and Regulation of Breathing Overview of Pulmonary Circulation o Diffusion of Gases o Exchange of Oxygen and Carbon Dioxide o Transport of Gases in the Blood
Chapter 17 The Respiratory System: Gas Exchange and Regulation of Breathing Overview of Pulmonary Circulation o Diffusion of Gases o Exchange of Oxygen and Carbon Dioxide o Transport of Gases in the Blood
Practice Packet Unit 8: Gases
 Name: Regents Chemistry: Practice Packet Unit 8: Gases www.chempride.weebly.com Vocabulary: Absolute Zero: Avogadro s Hypothesis: (Normal) Boiling Point: Direct Relationship: Evaporating: Gas: Ideal Gas:
Name: Regents Chemistry: Practice Packet Unit 8: Gases www.chempride.weebly.com Vocabulary: Absolute Zero: Avogadro s Hypothesis: (Normal) Boiling Point: Direct Relationship: Evaporating: Gas: Ideal Gas:
Lecture Presentation. Chapter 10. Gases. John D. Bookstaver St. Charles Community College Cottleville, MO Pearson Education, Inc.
 Lecture Presentation Chapter 10 John D. Bookstaver St. Charles Community College Cottleville, MO Characteristics of Unlike liquids and solids, gases Expand to fill their containers. Are highly compressible.
Lecture Presentation Chapter 10 John D. Bookstaver St. Charles Community College Cottleville, MO Characteristics of Unlike liquids and solids, gases Expand to fill their containers. Are highly compressible.
Chemistry Chapter 11 Test Review
 Chemistry Chapter 11 Test Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Pressure is the force per unit a. volume. c. length. b. surface area.
Chemistry Chapter 11 Test Review Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Pressure is the force per unit a. volume. c. length. b. surface area.
Chemistry 12 Notes on Graphs Involving LeChatelier s Principle
 Chemistry 12 Notes on Graphs Involving LeChatelier s Principle 1. Temperature Changes When a system adjusts due to a temperature change, there are no sudden changes in concentration of any species, so
Chemistry 12 Notes on Graphs Involving LeChatelier s Principle 1. Temperature Changes When a system adjusts due to a temperature change, there are no sudden changes in concentration of any species, so
Chemistry 51 Chapter 7 PROPERTIES OF GASES. Gases are the least dense and most mobile of the three phases of matter.
 ROERIES OF GASES Gases are the least dense and most mobile of the three phases of matter. articles of matter in the gas phase are spaced far apart from one another and move rapidly and collide with each
ROERIES OF GASES Gases are the least dense and most mobile of the three phases of matter. articles of matter in the gas phase are spaced far apart from one another and move rapidly and collide with each
Kinetic Molecular Theory
 Kinetic Molecular Theory Name Period Unit 7 HW 1 Worksheet (Goals 1 & 2) 1. Describe how gases, liquids, and solids compare using the following table. Volume (definite or indefinite) Molecular Motion (high,
Kinetic Molecular Theory Name Period Unit 7 HW 1 Worksheet (Goals 1 & 2) 1. Describe how gases, liquids, and solids compare using the following table. Volume (definite or indefinite) Molecular Motion (high,
B. As the gas particles move and strike a surface, they push on that surface 1. If we could measure the total amount of force exerted by gas
 Chapter 5: Gases I. The Structure of a Gas A. Gases are composed of particles that are flying around very fast in their container(s). 1. The particles travel in straight lines until they encounter either
Chapter 5: Gases I. The Structure of a Gas A. Gases are composed of particles that are flying around very fast in their container(s). 1. The particles travel in straight lines until they encounter either
PURE SUBSTANCE. Nitrogen and gaseous air are pure substances.
 CLASS Third Units PURE SUBSTANCE Pure substance: A substance that has a fixed chemical composition throughout. Air is a mixture of several gases, but it is considered to be a pure substance. Nitrogen and
CLASS Third Units PURE SUBSTANCE Pure substance: A substance that has a fixed chemical composition throughout. Air is a mixture of several gases, but it is considered to be a pure substance. Nitrogen and
GASES. Unit #8. AP Chemistry
 GASES Unit #8 AP Chemistry I. Characteristics of Gases A. Gas Characteristics: 1. Fills its container a. no definite shape b. no definite vol. 2. Easily mixes w/ other gases 3. Exerts pressure on its surroundings
GASES Unit #8 AP Chemistry I. Characteristics of Gases A. Gas Characteristics: 1. Fills its container a. no definite shape b. no definite vol. 2. Easily mixes w/ other gases 3. Exerts pressure on its surroundings
Example 5.1 Converting between Pressure Units
 Example 5.1 Converting between Pressure Units For Practice 5.1 Your local weather report announces that the barometric pressure is 30.44 in Hg. Convert this pressure to psi. For More Practice 5.1 Convert
Example 5.1 Converting between Pressure Units For Practice 5.1 Your local weather report announces that the barometric pressure is 30.44 in Hg. Convert this pressure to psi. For More Practice 5.1 Convert
Chapter 5. Nov 6 1:02 PM
 Chapter 5 Nov 6 1:02 PM Expand to fill their containers Fluid motion (they flow) Have low densities (1/1000 the density of equivalent liquids or solids) Compressible Can Effuse and Diffuse Effuse: The
Chapter 5 Nov 6 1:02 PM Expand to fill their containers Fluid motion (they flow) Have low densities (1/1000 the density of equivalent liquids or solids) Compressible Can Effuse and Diffuse Effuse: The
Dissolved Oxygen and measurement possibilities. Berno Lüpkes, 15 th March 2017
 Dissolved Oxygen and measurement possibilities Berno Lüpkes, 15 th March 2017 Content 1. Introduction to Dissolved Oxygen 2. Amperometric measurement principle 3. Optical measurement principle 4. Optical
Dissolved Oxygen and measurement possibilities Berno Lüpkes, 15 th March 2017 Content 1. Introduction to Dissolved Oxygen 2. Amperometric measurement principle 3. Optical measurement principle 4. Optical
Vapor Pressure of Liquids
 Experiment 10 In this experiment, you will investigate the relationship between the vapor pressure of a liquid and its temperature. When a liquid is added to the Erlenmeyer flask shown in Figure 1, it
Experiment 10 In this experiment, you will investigate the relationship between the vapor pressure of a liquid and its temperature. When a liquid is added to the Erlenmeyer flask shown in Figure 1, it
Chemistry A Molecular Approach. Fourth Edition. Chapter 5. Gases. Copyright 2017, 2014, 2011 Pearson Education, Inc. All Rights Reserved
 Chemistry A Molecular Approach Fourth Edition Chapter 5 Gases Supersonic Skydiving and the Risk of Decompression Gas Gases are composed of particles that are moving around very fast in their container(s).
Chemistry A Molecular Approach Fourth Edition Chapter 5 Gases Supersonic Skydiving and the Risk of Decompression Gas Gases are composed of particles that are moving around very fast in their container(s).
Unit 9 Packet: Gas Laws Introduction to Gas Laws Notes:
 Name: Unit 9 Packet: Gas Laws Introduction to Gas Laws Notes: Block: In chemistry, the relationships between gas physical properties are described as gas laws. Some of these properties are pressure, volume,
Name: Unit 9 Packet: Gas Laws Introduction to Gas Laws Notes: Block: In chemistry, the relationships between gas physical properties are described as gas laws. Some of these properties are pressure, volume,
Multiple Choice (40%)
 AP Chemistry Test (Chapter 5) Please do not write on this test thank you! Multiple Choice (40%) 1) A sealed rigid container is filled with three ideal gases: A, B and C. The partial pressure of each gas
AP Chemistry Test (Chapter 5) Please do not write on this test thank you! Multiple Choice (40%) 1) A sealed rigid container is filled with three ideal gases: A, B and C. The partial pressure of each gas
Chapter 13: The Behavior of Gases
 Chapter 13: The Behavior of Gases I. First Concepts a. The 3 states of matter most important to us: solids, liquids, and gases. b. Real Gases and Ideal Gases i. Real gases exist, ideal gases do not ii.
Chapter 13: The Behavior of Gases I. First Concepts a. The 3 states of matter most important to us: solids, liquids, and gases. b. Real Gases and Ideal Gases i. Real gases exist, ideal gases do not ii.
Pressure of the atmosphere varies with elevation and weather conditions. Barometer- device used to measure atmospheric pressure.
 Chapter 12 Section 1 Pressure A gas exerts pressure on its surroundings. Blow up a balloon. The gas we are most familiar with is the atmosphere, a mixture of mostly elemental nitrogen and oxygen. Pressure
Chapter 12 Section 1 Pressure A gas exerts pressure on its surroundings. Blow up a balloon. The gas we are most familiar with is the atmosphere, a mixture of mostly elemental nitrogen and oxygen. Pressure
Determination of Dissolved Gases in Ground Waters. By Ray Martrano Laboratory Director Seewald Laboratories Inc.
 Determination of Dissolved Gases in Ground Waters By Ray Martrano Laboratory Director Seewald Laboratories Inc. Overview of Presentation What is a dissolved gas? Why do we want to study dissolved gases?
Determination of Dissolved Gases in Ground Waters By Ray Martrano Laboratory Director Seewald Laboratories Inc. Overview of Presentation What is a dissolved gas? Why do we want to study dissolved gases?
ENVE 301 Environmental Engineering Unit Operations
 ENVE 301 Environmental Engineering Unit Operations Lecture 5 Gas Transfer, Aeration II SPRING 2014 Assist. Prof. A. Evren Tugtas Gas Transfer Examples 2 Example 1: Gas Transfer - Problems Henry s law constant
ENVE 301 Environmental Engineering Unit Operations Lecture 5 Gas Transfer, Aeration II SPRING 2014 Assist. Prof. A. Evren Tugtas Gas Transfer Examples 2 Example 1: Gas Transfer - Problems Henry s law constant
General Chemistry Mr. MacGillivray Test: States of Matter
 General Chemistry Mr. MacGillivray Test: States of Matter I. Label each of the 5 phase changes in the diagram below with the letter of the correct response. Not all of the letters get used. a) Vaporization
General Chemistry Mr. MacGillivray Test: States of Matter I. Label each of the 5 phase changes in the diagram below with the letter of the correct response. Not all of the letters get used. a) Vaporization
Practice Packet Unit 8: Gases
 Regents Chemistry: Mr. Palermo Practice Packet Unit 8: Gases Vocabulary: Lesson 1: Lesson 2: Lesson 3: Study Guide: 1 Vocabulary For each word, provide a short but specific definition from YOUR OWN BRAIN!
Regents Chemistry: Mr. Palermo Practice Packet Unit 8: Gases Vocabulary: Lesson 1: Lesson 2: Lesson 3: Study Guide: 1 Vocabulary For each word, provide a short but specific definition from YOUR OWN BRAIN!
CP Chapter 13/14 Notes The Property of Gases Kinetic Molecular Theory
 CP Chapter 13/14 Notes The Property of Gases Kinetic Molecular Theory Kinetic Molecular Theory of Gases The word kinetic refers to. Kinetic energy is the an object has because of its motion. Kinetic Molecular
CP Chapter 13/14 Notes The Property of Gases Kinetic Molecular Theory Kinetic Molecular Theory of Gases The word kinetic refers to. Kinetic energy is the an object has because of its motion. Kinetic Molecular
Classes at: - Topic: Gaseous State
 PHYSICAL CHEMISTRY by: SHAILENDRA KR. Classes at: - SCIENCE TUTORIALS; Opp. Khuda Baksh Library, Ashok Rajpath, Patna PIN POINT STUDY CIRCLE; House No. 5A/65, Opp. Mahual Kothi, Alpana Market, Patna Topic:
PHYSICAL CHEMISTRY by: SHAILENDRA KR. Classes at: - SCIENCE TUTORIALS; Opp. Khuda Baksh Library, Ashok Rajpath, Patna PIN POINT STUDY CIRCLE; House No. 5A/65, Opp. Mahual Kothi, Alpana Market, Patna Topic:
Dissolved Oxygen Guide
 Educat i onser i es Di ssol vedoxygengui de Dissolved Oxygen Guide Introduction Dissolved oxygen probes provide a convenient approach to essentially direct measurement of molecular oxygen. The membrane
Educat i onser i es Di ssol vedoxygengui de Dissolved Oxygen Guide Introduction Dissolved oxygen probes provide a convenient approach to essentially direct measurement of molecular oxygen. The membrane
Chapter 10: Properties of Gases: The Air We Breathe
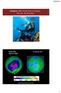 Chapter 10: Properties of Gases: The Air We Breathe South Pole Sept 24, 2006 15 February 2017 http://ozonewatch.gsfc.nasa.gov 1 Chapter Outline 10.1 10.2 10.3 The Properties of Gases Effusion and the Kinetic
Chapter 10: Properties of Gases: The Air We Breathe South Pole Sept 24, 2006 15 February 2017 http://ozonewatch.gsfc.nasa.gov 1 Chapter Outline 10.1 10.2 10.3 The Properties of Gases Effusion and the Kinetic
Chapter 12. The Gaseous State of Matter
 Chapter 12 The Gaseous State of Matter The air in a hot air balloon expands When it is heated. Some of the air escapes from the top of the balloon, lowering the air density inside the balloon, making the
Chapter 12 The Gaseous State of Matter The air in a hot air balloon expands When it is heated. Some of the air escapes from the top of the balloon, lowering the air density inside the balloon, making the
4.) There are no forces of attraction or repulsion between gas particles. This means that
 KINETIC MOLECULAR (K-M) THEORY OF MATTER NOTES - based on the idea that particles of matter are always in motion - assumptions of the K-M Theory 1.) Gases consist of large numbers of tiny particles that
KINETIC MOLECULAR (K-M) THEORY OF MATTER NOTES - based on the idea that particles of matter are always in motion - assumptions of the K-M Theory 1.) Gases consist of large numbers of tiny particles that
Accelerated Chemistry Study Guide Chapter 13: Gases
 Accelerated Chemistry Study Guide Chapter 13: Gases Terms, definitions, topics Diffusion Kinetic Molecular Theory Atmospheric pressure Barometer Manometer STP Absolute zero Page 1 of 42 Molar volume Partial
Accelerated Chemistry Study Guide Chapter 13: Gases Terms, definitions, topics Diffusion Kinetic Molecular Theory Atmospheric pressure Barometer Manometer STP Absolute zero Page 1 of 42 Molar volume Partial
Chemistry 1B Chapter 10 Worksheet - Daley. Name
 Name 1) The National Weather Service routinely supplies atmospheric pressure data to help pilots set their altimeters. The units the NWS uses for atmospheric pressure are inches of mercury. A barometric
Name 1) The National Weather Service routinely supplies atmospheric pressure data to help pilots set their altimeters. The units the NWS uses for atmospheric pressure are inches of mercury. A barometric
3/30/2013. Vapor Pressure and Changes of State Phase Diagrams. Chapter 10 Sections 8, 9
 Vapor Pressure and Changes of State Chapter 10 Sections 8, 9 Vapor Pressure In a closed container, liquid evaporates. The vapor starts to condensate. Condensation and evaporation happens simultaneously.
Vapor Pressure and Changes of State Chapter 10 Sections 8, 9 Vapor Pressure In a closed container, liquid evaporates. The vapor starts to condensate. Condensation and evaporation happens simultaneously.
Gas Pressure. Pressure is the force exerted per unit area by gas molecules as they strike the surfaces around them.
 Chapter 5 Gases Gas Gases are composed of particles that are moving around very fast in their container(s). These particles moves in straight lines until they collides with either the container wall or
Chapter 5 Gases Gas Gases are composed of particles that are moving around very fast in their container(s). These particles moves in straight lines until they collides with either the container wall or
Gases Day 12. Phases of Matter
 Phases of Matter Gases Day 12 Kinetic Molecular Theory ( Ideal Gases ) 1) The molecules of a gas are in continual, and random, motion of varying speeds. 2) The average kinetic energy of the gas molecules
Phases of Matter Gases Day 12 Kinetic Molecular Theory ( Ideal Gases ) 1) The molecules of a gas are in continual, and random, motion of varying speeds. 2) The average kinetic energy of the gas molecules
Dissolved Oxygen Meter. ino2. Instruction Manual. Serial No..
 Innovative Instruments, Inc. http://www.2in.com Dissolved Oxygen Meter ino2 Instruction Manual Serial No.. Innovative Instruments, Inc. 8533 Queen Brooks Ct. Tampa, FL 33637. USA Phone: 813-727-0676. Fax:
Innovative Instruments, Inc. http://www.2in.com Dissolved Oxygen Meter ino2 Instruction Manual Serial No.. Innovative Instruments, Inc. 8533 Queen Brooks Ct. Tampa, FL 33637. USA Phone: 813-727-0676. Fax:
KATEDRA MATERIÁLOVÉHO INŽENÝRSTVÍ A CHEMIE. 123MEAN water vapor transport
 KATEDRA MATERIÁLOVÉHO INŽENÝRSTVÍ A CHEMIE 123MEAN water vapor transport Water vapor transport : Thermal insulation properties of porous building materials dramatically decrease because of water vapor
KATEDRA MATERIÁLOVÉHO INŽENÝRSTVÍ A CHEMIE 123MEAN water vapor transport Water vapor transport : Thermal insulation properties of porous building materials dramatically decrease because of water vapor
SCH3U7 Quantitative Chemistry
 SCH3U7 Quantitative Chemistry So far, we have looked at solids and liquids (solutions) Today we will look at gases and the laws that govern their behaviour in chemical reactions 4 Factors Affecting Gases
SCH3U7 Quantitative Chemistry So far, we have looked at solids and liquids (solutions) Today we will look at gases and the laws that govern their behaviour in chemical reactions 4 Factors Affecting Gases
PRE LABORATORY ASSIGNMENT: Lab Section Score: /10 READ THE LAB TEXT BEFORE ATTEMPTING THESE PROBLEMS!
 EXPERIMENT # 6 Name: PRE LABORATORY ASSIGNMENT: Lab Section Score: /10 READ THE LAB TEXT BEFORE ATTEMPTING THESE PROBLEMS! 1. Calculate the height of a corresponding column of mercury (in mm) that is at
EXPERIMENT # 6 Name: PRE LABORATORY ASSIGNMENT: Lab Section Score: /10 READ THE LAB TEXT BEFORE ATTEMPTING THESE PROBLEMS! 1. Calculate the height of a corresponding column of mercury (in mm) that is at
NOTES: Behavior of Gases
 NOTES: Behavior of Gases Properties of Gases Gases have weight Gases take up space Gases exert pressure Gases fill their containers Gases are mostly empty space The molecules in a gas are separate, very
NOTES: Behavior of Gases Properties of Gases Gases have weight Gases take up space Gases exert pressure Gases fill their containers Gases are mostly empty space The molecules in a gas are separate, very
11/22/ (4) Harmonization: <846> SPECIFIC SURFACE AREA
 BRIEFING 846 Specific Surface Area, USP 27 page 2385. The European Pharmacopoeia is the coordinating pharmacopeia for the international harmonization of the Specific Surface Area General Chapter, as part
BRIEFING 846 Specific Surface Area, USP 27 page 2385. The European Pharmacopoeia is the coordinating pharmacopeia for the international harmonization of the Specific Surface Area General Chapter, as part
1. Quantity of a gas (moles) 2. Temperature of the gas. 3. Volume occupied by the gas. 4. Pressure exerted by the gas. PV = nrt
 Experiment 5 Stoichiometry : Gases Determining the Ideal Gas Constant Lab Owl Announcement: Upon completion of this lab log onto OWL. Your fourth Lab Owl assignment, Lab Owl: Exp 5 should appear there.
Experiment 5 Stoichiometry : Gases Determining the Ideal Gas Constant Lab Owl Announcement: Upon completion of this lab log onto OWL. Your fourth Lab Owl assignment, Lab Owl: Exp 5 should appear there.
Chapter 14 Practice Problems
 Chapter 14 Practice Problems In problems that require the atomic masses (atomic weights) of atomic hydrogen, oxygen, nitrogen, and carbon, we will use the rounded values, 1, 16, 14, and 12, respectively.
Chapter 14 Practice Problems In problems that require the atomic masses (atomic weights) of atomic hydrogen, oxygen, nitrogen, and carbon, we will use the rounded values, 1, 16, 14, and 12, respectively.
Kinetic-Molecular Theory
 GASES Chapter Eleven Kinetic-Molecular Theory! Recall that our only previous description of gases stated that gases completely fill and take the shape of their containers.! The Kinetic-Molecular Theory
GASES Chapter Eleven Kinetic-Molecular Theory! Recall that our only previous description of gases stated that gases completely fill and take the shape of their containers.! The Kinetic-Molecular Theory
Expand to fill their containers, are highly compressible, have extremely low densities.
 Chem150 week6 Handout 1 Gases Characteristics of Gases: Unlike liquids and solids, they Expand to fill their containers, are highly compressible, have extremely low densities. Pressure is the amount of
Chem150 week6 Handout 1 Gases Characteristics of Gases: Unlike liquids and solids, they Expand to fill their containers, are highly compressible, have extremely low densities. Pressure is the amount of
Chemistry Chapter 10 Test
 Chemistry Chapter 10 Test True/False Indicate whether the sentence or statement is true or false. 1. KMT stands for Kinetic Mole Theory. 2. One of the assumptions in the KMT is that the particles are spread
Chemistry Chapter 10 Test True/False Indicate whether the sentence or statement is true or false. 1. KMT stands for Kinetic Mole Theory. 2. One of the assumptions in the KMT is that the particles are spread
CP Chapter 13/14 Notes The Property of Gases Kinetic Molecular Theory
 CP Chapter 13/14 Notes The Property of Gases Kinetic Molecular Theory Kinetic Molecular Theory of Gases The word kinetic refers to. Kinetic energy is the an object has because of its motion. Kinetic Molecular
CP Chapter 13/14 Notes The Property of Gases Kinetic Molecular Theory Kinetic Molecular Theory of Gases The word kinetic refers to. Kinetic energy is the an object has because of its motion. Kinetic Molecular
Chapter 11 The Behavior of Gases
 Chapter 11 The Behavior of Gases 1 Section 11.1 The Properties of Gases Objectives: Explain why gases are easier to compress than solids or liquids are. Describe the three factors that affect gas pressure
Chapter 11 The Behavior of Gases 1 Section 11.1 The Properties of Gases Objectives: Explain why gases are easier to compress than solids or liquids are. Describe the three factors that affect gas pressure
PHY131H1S - Class 23. Today: Fluids Pressure Pascal s Law Gauge Pressure Buoyancy, Archimedes Principle. A little pre-class reading quiz
 PHY131H1S - Class 23 Today: Fluids Pressure Pascal s Law Gauge Pressure Buoyancy, Archimedes Principle Archimedes (287-212 BC) was asked to check the amount of silver alloy in the king s crown. The answer
PHY131H1S - Class 23 Today: Fluids Pressure Pascal s Law Gauge Pressure Buoyancy, Archimedes Principle Archimedes (287-212 BC) was asked to check the amount of silver alloy in the king s crown. The answer
Chemistry Honors - Gases
 Name: Class: Date: ID: A Chemistry Honors - Gases Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Why does a can collapse when a vacuum pump removes air
Name: Class: Date: ID: A Chemistry Honors - Gases Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Why does a can collapse when a vacuum pump removes air
A) It cannot be predicted. B) It is squared. C) It is doubled. D) It is halved. E) It does not change.
 AP Chemistry Test (Chapter 5) Class Set Multiple Choice (50%) 1) A sample of argon gas is sealed in a container. The volume of the container is doubled at a constant temperature. What happens to the pressure
AP Chemistry Test (Chapter 5) Class Set Multiple Choice (50%) 1) A sample of argon gas is sealed in a container. The volume of the container is doubled at a constant temperature. What happens to the pressure
THE GAS STATE. Unit 4. CHAPTER KEY TERMS HOME WORK 9.1 Kinetic Molecular Theory States of Matter Solid, Liquid, gas.
 Unit 4 THE GAS STATE CHAPTER KEY TERMS HOME WORK 9. Kinetic Molecular Theory States of Matter Solid, Liquid, gas Page 4 # to 4 9. Boyles Law P α /V PV = Constant P V = P V Pressure Atmospheric Pressure
Unit 4 THE GAS STATE CHAPTER KEY TERMS HOME WORK 9. Kinetic Molecular Theory States of Matter Solid, Liquid, gas Page 4 # to 4 9. Boyles Law P α /V PV = Constant P V = P V Pressure Atmospheric Pressure
Completed ALL 2 Warm-up IC Kinetic Molecular Theory Notes. Kinetic Molecular Theory and Pressure Worksheet
 Name: Unit 10- Gas Laws Day Page # Description IC/HW Due Date Completed ALL 2 Warm-up IC 1 3 5 Kinetic Molecular Theory Notes IC 1 6 8 Kinetic Molecular Theory and Pressure Worksheet IC 2 9 10 Gas Law
Name: Unit 10- Gas Laws Day Page # Description IC/HW Due Date Completed ALL 2 Warm-up IC 1 3 5 Kinetic Molecular Theory Notes IC 1 6 8 Kinetic Molecular Theory and Pressure Worksheet IC 2 9 10 Gas Law
Section 5.1 Pressure. Why study gases? An understanding of real world phenomena. An understanding of how science works.
 Chapter 5 Gases Section 5.1 Pressure Why study gases? An understanding of real world phenomena. An understanding of how science works. Copyright Cengage Learning. All rights reserved 2 Section 5.1 Pressure
Chapter 5 Gases Section 5.1 Pressure Why study gases? An understanding of real world phenomena. An understanding of how science works. Copyright Cengage Learning. All rights reserved 2 Section 5.1 Pressure
