Mixing Methacholine Dilutions for Bronchoprovocation Challenge Testing
|
|
|
- Clarence Cain
- 6 years ago
- Views:
Transcription
1 powder for solution, for inhalation Mixing Methacholine Dilutions for Bronchoprovocation Challenge Testing Provocholine is a registered trademark of Methapharm Inc. Copyright Methapharm Inc All rights reserved. Healthcare professionals should consult the Provocholine package insert for the complete product safety information. A sterile 0.22 μm bacterial retentive filter (e.g., Millex GV ) should be used when transferring a solution to the nebulizer. Warning: Provocholine is a bronchoconstrictor agent for diagnostic purposes only and should not be used as a therapeutic agent. Provocholine inhalation challenge should be performed only under the supervision of a physician trained in and thoroughly familiar with all aspects of the technique of methacholine challenge, all contraindications, warnings and precautions, and the management of respiratory distress. Emergency equipment and medication should be immediately available to treat acute respiratory distress. For complete prescribing information, please consult the Product Insert which is available for download at or on request by calling Methapharm Medical Information at ext 7804 or faxing us at This information is provided as a professional courtesy, and it is intended to provide data available to us that may assist you in deriving your own conclusions and opinions. This information is not intended to advocate any indications, dosage, or other claim that is not described in the package insert. 1 PROVO-MIX-R1-0516
2 Overview 1. Gathering Necessary Equipment 2. Special Precautions 3. Reconstituting Provocholine 2
3 1 Supplies 100 mg vial of Provocholine 3
4 1 Supplies Diluent options 0.9% Saline 0.9% Saline with 0.4% Phenol (ph 7.0)* Sterile empty vials* Syringes and needles * Available for purchase from Methapharm, Inc. 4
5 1 Supplies 0.22 μm sterile bacterial-retentive filter* Alcohol preparation pads Green labels (included) * Available for purchase from Methapharm, Inc. 5
6 2 Reconstituting Precautions Please refer to the Provocholine package insert for full instructions and safety precautions 6
7 2 Reconstituting Precautions Accurate sterile technique is essential to obtain appropriate test results while maintaining patient safety 7
8 2 Precautions Green Labels Fill in all information on the labels Include 14 day expiration on Vials A-D Vial E should be prepared the day of testing 8
9 2 Precautions Green Labels Dilution Strength Type of Diluent Used Lot number Preparation Date Expiration Date Initials of person performing dilution 9
10 3 Dosing Protocols US Package Insert (mg/ml) ATS Short (mg/ml) Diluent Diluent 10
11 Step 1 Attach the completed green labels to the sterile empty vials. 11
12 Step 2 Wipe the top of the Provocholine vial, the diluent vial and the sterile empty vials with alcohol preparation pads. 12
13 Step 3 Label two syringes, one for the Provocholine and one for the diluent. 13
14 Step 4 Package Insert Dilutions Using the Diluent syringe, withdraw 4 ml of diluent and inject it into the Provocholine vial creating a stock solution. Shake well. This solution contains 25 mg/ml of methacholine chloride. 14
15 Step 5 Package Insert Dilutions Using the Provocholine vial as your stock solution, withdraw 1 ml of the 25 mg/ml solution with the Provocholine syringe and add it to the vial labeled Vial B. 15
16 Step 5 Package Insert Dilutions Using the Diluent syringe, add 1.5 ml of diluent to the same vial. Shake well. Vial B will contain a 10 mg/ml solution of methacholine chloride. 16
17 Step 6 Package Insert Dilutions Using the Provocholine vial as your stock solution, withdraw 1 ml of the 25 mg/ml solution with the Provocholine syringe and add it to the vial labeled Vial C. 17
18 Step 6 Package Insert Dilutions Using the Diluent syringe, add 9 ml of diluent to the same vial. Shake well. Vial C will contain a 2.5 mg/ml solution of methacholine chloride. 18
19 Step 7 Package Insert Dilutions From here, we will perform a serial dilution to make the remaining two concentrations. Using the Provocholine syringe, withdraw 1 ml from Vial C (2.5 mg/ml) and add it to the vial labeled Vial D. 19
20 Step 7 Package Insert Dilutions Using the Diluent syringe, add 9 ml of diluent to the same vial. Shake well. Vial D will contain a 0.25 mg/ml solution of methacholine chloride. 20
21 Step 8 Package Insert Dilutions Repeat the same process for Vial E the day of the test, withdrawing 1 ml from Vial D (0.25 mg/ml) and adding 9 ml of diluent. Vial E will contain a mg/ml solution of methacholine chloride. 21
22 Step 4 ATS Short Dilutions Using the Diluent syringe, withdraw 6.25 ml of diluent and inject it into the Provocholine vial. Shake well. This solution contains 16 mg/ml of methacholine chloride. 22
23 Step 5 ATS Short Dilutions For the ATS Short Dilutions you will perform a serial dilution using the previous dilution as the stock solution at each step. Using the Provocholine vial, withdraw 3 ml of the 16 mg/ml solution using the Provocholine syringe and add it to the vial labeled Vial B. 23
24 Step 5 ATS Short Dilutions Using the Diluent syringe, add 9 ml of diluent to the same vial. Shake well. Vial B will contain a 4 mg/ml solution of methacholine chloride. 24
25 Step 6 ATS Short Dilutions Using the previous vial (Vial B), withdraw 3 ml of the 4 mg/ml solution using the Provocholine syringe and add it to the vial labeled Vial C. 25
26 Step 6 ATS Short Dilutions Using the Diluent syringe, add 9 ml of diluent to the same vial. Shake well. Vial C will contain a 1 mg/ml solution of methacholine chloride. 26
27 Step 7 ATS Short Dilutions Using the Provocholine syringe, withdraw 3 ml from Vial C (1 mg/ml) and add it to the vial labeled Vial D. Add 9 ml of diluent to the same vial. Shake well. Vial D will contain a 0.25 mg/ml solution of methacholine chloride. 27
28 Step 8 ATS Short Dilutions Repeat the same process for vial E the day of the test, withdrawing 3 ml from Vial D (0.25 mg/ml) and adding 9 ml of diluent. Vial E will contain a mg/ml solution of methacholine chloride. 28
29 3 Storage of Dilutions Refrigerate reconstituted solutions at 36 to 46 F (2 to 8 C) for up to 2 weeks for vials A-D Vial E must be prepared on the day of testing Freezing does not affect stability of the dilutions 29
30 Summary US Package Insert (mg/ml) ATS Short (mg/ml) Diluent Diluent 30
31 Questions or Concerns Contact Methapharm at
Dilution Sequence Protocol
 Dilution Sequence Protocol ATS Short Quadrupling Doses 0.0625 0.25 1 4 16 Provocholine 1600 mg/vial (US NDC 64281-016-01) 1 Provocholine (methacholine chloride USP) powder for solution, for inhalation,
Dilution Sequence Protocol ATS Short Quadrupling Doses 0.0625 0.25 1 4 16 Provocholine 1600 mg/vial (US NDC 64281-016-01) 1 Provocholine (methacholine chloride USP) powder for solution, for inhalation,
Dilution Sequence Protocol
 Dilution Sequence Protocol ATS Short Quadrupling Doses 0.0625 0.25 1 4 16 Provocholine 100 mg/vial (US NDC 64281-100-06) 1 Provocholine (methacholine chloride USP) powder for solution, for inhalation,
Dilution Sequence Protocol ATS Short Quadrupling Doses 0.0625 0.25 1 4 16 Provocholine 100 mg/vial (US NDC 64281-100-06) 1 Provocholine (methacholine chloride USP) powder for solution, for inhalation,
Dilution Sequence Protocol
 Dilution Sequence Protocol US Package Insert Multi-Patient (2-5 patients; requires 2 vials of Provocholine) 0.025 0.25 2.5 10 25 Provocholine 100 mg/vial (US NDC 64281-100-06) 1 Provocholine (methacholine
Dilution Sequence Protocol US Package Insert Multi-Patient (2-5 patients; requires 2 vials of Provocholine) 0.025 0.25 2.5 10 25 Provocholine 100 mg/vial (US NDC 64281-100-06) 1 Provocholine (methacholine
CHAPTER 10 Reconstitution of Powdered Drugs
 CHAPTER 10 RECONSTITUTION OF POWDERED DRUGS 89 CHAPTER 10 Reconstitution of Powdered Drugs Objectives The learner will: 1. prepare solutions from powdered drugs using directions printed on vial labels.
CHAPTER 10 RECONSTITUTION OF POWDERED DRUGS 89 CHAPTER 10 Reconstitution of Powdered Drugs Objectives The learner will: 1. prepare solutions from powdered drugs using directions printed on vial labels.
3. This solution retains potency for 2 8 hours at room temperature after reconstitution.
 1 CHAPTER 10 Reconstitution of Powdered Drugs Problems 10.1 (page 124) 1. The drug should be stored at or below 86 F (30 C). 2. This drug is administered IM or IV. 3. This solution retains potency for
1 CHAPTER 10 Reconstitution of Powdered Drugs Problems 10.1 (page 124) 1. The drug should be stored at or below 86 F (30 C). 2. This drug is administered IM or IV. 3. This solution retains potency for
TEV-TROPIN [somatropin (rdna origin) for injection] 5 mg & 10 mg
![TEV-TROPIN [somatropin (rdna origin) for injection] 5 mg & 10 mg TEV-TROPIN [somatropin (rdna origin) for injection] 5 mg & 10 mg](/thumbs/74/70163471.jpg) TEV-TROPIN [somatropin (rdna origin) for injection] 5 mg & 10 mg TEV-TROPIN (somatropin for injection) Information for the Patient or Parent Read all instructions before proceeding. Do not mix (reconstitute)
TEV-TROPIN [somatropin (rdna origin) for injection] 5 mg & 10 mg TEV-TROPIN (somatropin for injection) Information for the Patient or Parent Read all instructions before proceeding. Do not mix (reconstitute)
RECONSTITUTION AND ADMINISTRATION INSTRUCTIONS FOR HEALTHCARE PROFESSIONALS
 RECONSTITUTION AND ADMINISTRATION INSTRUCTIONS FOR HEALTHCARE PROFESSIONALS TRUST THE POWER OF PREVENTION WITH CINRYZE Whether your patients are new to treatment or are continuing their journey, remind
RECONSTITUTION AND ADMINISTRATION INSTRUCTIONS FOR HEALTHCARE PROFESSIONALS TRUST THE POWER OF PREVENTION WITH CINRYZE Whether your patients are new to treatment or are continuing their journey, remind
Aseptic Techniques. Techniques for Sterile Compounding. Pharmacy Technician Training Systems Passassured, LLC
 Aseptic Techniques Techniques for Sterile Compounding Pharmacy Technician Training Systems Passassured, LLC Aseptic Techniques, Tech for Sterile Compounding PassAssured's Pharmacy Technician Training Program
Aseptic Techniques Techniques for Sterile Compounding Pharmacy Technician Training Systems Passassured, LLC Aseptic Techniques, Tech for Sterile Compounding PassAssured's Pharmacy Technician Training Program
RECONSTITUTION AND ADMINISTRATION INSTRUCTIONS FOR HEALTHCARE PROFESSIONALS. Hereditary Angioedema
 RECONSTITUTION AND ADMINISTRATION INSTRUCTIONS FOR HEALTHCARE PROFESSIONALS Hereditary Angioedema TRUST PROVEN PREVENTION WITH CINRYZE Whether your patients are new to treatment or are continuing their
RECONSTITUTION AND ADMINISTRATION INSTRUCTIONS FOR HEALTHCARE PROFESSIONALS Hereditary Angioedema TRUST PROVEN PREVENTION WITH CINRYZE Whether your patients are new to treatment or are continuing their
helping patients thrive Clotting Factor Concentrate Information
 helping patients thrive Clotting Factor Concentrate Information LEGEND: VWF Von Willebrand factor Plasma derived IU International unit RCo Ristocetin cofactor ml Milliliter mg Milligram clotting factor
helping patients thrive Clotting Factor Concentrate Information LEGEND: VWF Von Willebrand factor Plasma derived IU International unit RCo Ristocetin cofactor ml Milliliter mg Milligram clotting factor
Note: Does have indication for Prophy- suggest 25 units/kg every other day
 Recombinant Factor VIII Helixate (CSL): Vial Sizes: 250,500, 1000, 2000, 3000 2.5ml 5ml 2000 s & 3,000 s Administer within 3 hours of reconstitution. A dose of Helixate may be administered over a period
Recombinant Factor VIII Helixate (CSL): Vial Sizes: 250,500, 1000, 2000, 3000 2.5ml 5ml 2000 s & 3,000 s Administer within 3 hours of reconstitution. A dose of Helixate may be administered over a period
PREPARATION AND SELF-ADMINISTRATION INSTRUCTIONS FOR PATIENTS
 PREPARATION AND SELF-ADMINISTRATION INSTRUCTIONS FOR PATIENTS Hereditary Angioedema Jenny, a real HAE patient TAKE CONTROL OF YOUR HEREDITARY ANGIOEDEMA (HAE) TREATMENT WITH SELF-ADMINISTRATION Choosing
PREPARATION AND SELF-ADMINISTRATION INSTRUCTIONS FOR PATIENTS Hereditary Angioedema Jenny, a real HAE patient TAKE CONTROL OF YOUR HEREDITARY ANGIOEDEMA (HAE) TREATMENT WITH SELF-ADMINISTRATION Choosing
RECOMBINATE. [Antihemophilic Factor (Recombinant)] (For intravenous use only)
![RECOMBINATE. [Antihemophilic Factor (Recombinant)] (For intravenous use only) RECOMBINATE. [Antihemophilic Factor (Recombinant)] (For intravenous use only)](/thumbs/79/80305643.jpg) Website for more information: http://www.recombinate.com/ INSTRUCTIONS FOR USE RECOMBINATE [Antihemophilic Factor (Recombinant)] (For intravenous use only) Do not attempt to do an infusion to yourself
Website for more information: http://www.recombinate.com/ INSTRUCTIONS FOR USE RECOMBINATE [Antihemophilic Factor (Recombinant)] (For intravenous use only) Do not attempt to do an infusion to yourself
PREPARATION AND SELF-ADMINISTRATION INSTRUCTIONS FOR PATIENTS
 PREPARATION AND SELF-ADMINISTRATION INSTRUCTIONS FOR PATIENTS Hereditary Angioedema TAKE CONTROL OF HEREDITARY ANGIOEDEMA (HAE) TREATMENT WITH FLEXIBLE ADMINISTRATION Choosing to treat with CINRYZE (C1
PREPARATION AND SELF-ADMINISTRATION INSTRUCTIONS FOR PATIENTS Hereditary Angioedema TAKE CONTROL OF HEREDITARY ANGIOEDEMA (HAE) TREATMENT WITH FLEXIBLE ADMINISTRATION Choosing to treat with CINRYZE (C1
APPENDIX 5. Donor Heart Perfusion & Preservation Protocol for NORS Teams in the UK
 APPENDIX 5 Donor Heart Perfusion & Preservation Protocol for NORS Teams in the UK 1. Systemically anticoagulate donors with 30,000 units of IV heparin 2. Venting of the IVC (chest or abdomen) as agreed
APPENDIX 5 Donor Heart Perfusion & Preservation Protocol for NORS Teams in the UK 1. Systemically anticoagulate donors with 30,000 units of IV heparin 2. Venting of the IVC (chest or abdomen) as agreed
MSD 96-Well MULTI-ARRAY CRP Assay
 MSD 96-Well MULTI-ARRAY CRP Assay The following assay protocol has been optimized for analysis of C-reactive protein (CRP) in human serum and plasma samples. MSD Materials Storage Read Buffer T (4X), with
MSD 96-Well MULTI-ARRAY CRP Assay The following assay protocol has been optimized for analysis of C-reactive protein (CRP) in human serum and plasma samples. MSD Materials Storage Read Buffer T (4X), with
Varithena Delivery System Instructions for Use INSTRUCTIONS FOR USE Please read the Product Monograph before using the product.
 Varithena Delivery System Instructions for Use INSTRUCTIONS FOR USE Please read the Product Monograph before using the product. The Instructions for Use are for the entire Varithena TM system. There are
Varithena Delivery System Instructions for Use INSTRUCTIONS FOR USE Please read the Product Monograph before using the product. The Instructions for Use are for the entire Varithena TM system. There are
Health Products Regulatory Authority
 1 NAME OF THE VETERINARY MEDICINAL PRODUCT Myodine 25 mg/ml solution for injection for dogs and cats 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains: Active substance: Nandrodine laurate 25
1 NAME OF THE VETERINARY MEDICINAL PRODUCT Myodine 25 mg/ml solution for injection for dogs and cats 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml contains: Active substance: Nandrodine laurate 25
C O L O G Y. New. g u O N. l o t a C a. The high security connection
 O N g u e C O L O G Y New l o t a C a The high security connection Preparation devices for medications/solutions Bag qimospike B 7218.01 qimospike B is an IV fluid bag access spike fitted with qimo. Aspiration
O N g u e C O L O G Y New l o t a C a The high security connection Preparation devices for medications/solutions Bag qimospike B 7218.01 qimospike B is an IV fluid bag access spike fitted with qimo. Aspiration
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Human, Recombinant) Reference Number: CP.CPA.76 Effective Date: 11.16.16 Last Review Date: 08.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of
Clinical Policy: (Human, Recombinant) Reference Number: CP.CPA.76 Effective Date: 11.16.16 Last Review Date: 08.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of
Varithena Delivery System Instructions for Use
 Varithena Delivery System Instructions for Use Please read all prescribing information before using the product. The Instructions for Use are for the entire Varithena system. There are 2 packaging configurations:
Varithena Delivery System Instructions for Use Please read all prescribing information before using the product. The Instructions for Use are for the entire Varithena system. There are 2 packaging configurations:
MSD 96-Well MULTI-ARRAY Human (6E10) Abeta 42 Ultra-Sensitive Kit
 MSD 96-Well MULTI-ARRAY Human (6E10) Abeta 42 Ultra-Sensitive Kit The first protocol has been optimized for quantifying Aβ 1-42 peptide in human cerebrospinal fluid (CSF). An additional general protocol
MSD 96-Well MULTI-ARRAY Human (6E10) Abeta 42 Ultra-Sensitive Kit The first protocol has been optimized for quantifying Aβ 1-42 peptide in human cerebrospinal fluid (CSF). An additional general protocol
Safety Data Sheet MasterSet DELVO ESC also DELVO ESC Revision date : 2014/12/16 Page: 1/6
 Revision date : 2014/12/16 Page: 1/6 1. Product and Company Identification Company BASF Canada Inc. 100 Milverton Drive Mississauga, ON L5R 4H1, CANADA 24 Hour Emergency Response Information CANUTEC (reverse
Revision date : 2014/12/16 Page: 1/6 1. Product and Company Identification Company BASF Canada Inc. 100 Milverton Drive Mississauga, ON L5R 4H1, CANADA 24 Hour Emergency Response Information CANUTEC (reverse
MSD 96-Well MULTI-ARRAY Human (6E10) Abeta 40 Ultra-Sensitive Kit
 MSD 96-Well MULTI-ARRAY Human (6E10) Abeta 40 Ultra-Sensitive Kit The first protocol has been optimized for quantifying Aβ 1-40 peptide in human cerebrospinal fluid (CSF). The second protocol has been
MSD 96-Well MULTI-ARRAY Human (6E10) Abeta 40 Ultra-Sensitive Kit The first protocol has been optimized for quantifying Aβ 1-40 peptide in human cerebrospinal fluid (CSF). The second protocol has been
International Journal of Generic Drugs ANALYTICAL METHOD PROCEDURES THIS SOP IS 'SWITCHED : OFF : ON'
 DISSOLUTION ASSAY Release & Stability Studies Ed. 04. General Index: 1. PRODUCT SPECIFICATIONS (USP MONOGRAPH) 2. IDENTIFICATION BY HPLC 3. ASSAY - HPLC SETUP 4. STANDARD PREPARATION 5. SYSTEM SUITABILITY
DISSOLUTION ASSAY Release & Stability Studies Ed. 04. General Index: 1. PRODUCT SPECIFICATIONS (USP MONOGRAPH) 2. IDENTIFICATION BY HPLC 3. ASSAY - HPLC SETUP 4. STANDARD PREPARATION 5. SYSTEM SUITABILITY
CSL Behring PACKAGE LEAFLET: INFORMATION FOR THE USER
 CSL Behring PACKAGE LEAFLET: INFORMATION FOR THE USER Berinert 500 IU Powder and solvent for solution for injection / infusion Berinert 1500 IU Powder and solvent for solution for injection Human C1-esterase
CSL Behring PACKAGE LEAFLET: INFORMATION FOR THE USER Berinert 500 IU Powder and solvent for solution for injection / infusion Berinert 1500 IU Powder and solvent for solution for injection Human C1-esterase
Voncento SDS Revision Date: 06/04/2015
 1. Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product Identity Voncento Alternate Names Human Coagulation Factor VIII/Von Willebrand Factor complex concentrate
1. Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product Identity Voncento Alternate Names Human Coagulation Factor VIII/Von Willebrand Factor complex concentrate
HUMAN IL6 KITS PROTOCOL
 HUMAN IL6 KITS PROTOCOL Part # 62HIL06PEG & 62HIL06PEH Test size: 500 tests (62HIL06PEG), 10,000 tests (62HIL06PEH) - assay volume: 20 µl Revision: 04 (Jan. 2018) Store at: -60 C or below This product
HUMAN IL6 KITS PROTOCOL Part # 62HIL06PEG & 62HIL06PEH Test size: 500 tests (62HIL06PEG), 10,000 tests (62HIL06PEH) - assay volume: 20 µl Revision: 04 (Jan. 2018) Store at: -60 C or below This product
ab IgG1 Human ELISA Kit
 ab100548 IgG1 Human ELISA Kit Instructions for Use For the quantitative measurement of Human IgG1 in serum and plasma This product is for research use only and is not intended for diagnostic use. Version
ab100548 IgG1 Human ELISA Kit Instructions for Use For the quantitative measurement of Human IgG1 in serum and plasma This product is for research use only and is not intended for diagnostic use. Version
Stimate Nasal Spray SDS Revision Date: 10/22/2015
 1. Identification 1.1. Product identifier Product Identity Alternate Names Stimate Nasal Spray Stimate Nasal Spray 1.2. Relevant identified uses of the substance or mixture and uses advised against Intended
1. Identification 1.1. Product identifier Product Identity Alternate Names Stimate Nasal Spray Stimate Nasal Spray 1.2. Relevant identified uses of the substance or mixture and uses advised against Intended
Safety Data Sheet. allergylabs.com. 1. Product & Company Identification. 2. Hazards Identification
 L Safety Data Sheet allergylabs.com Version 1.0 Revision Date: 09/15/2014 1. Product & Company Identification 1.1 Product identifiers Product Name : Sterile Diluent - 10% Glycero-Saline Product Code(s)
L Safety Data Sheet allergylabs.com Version 1.0 Revision Date: 09/15/2014 1. Product & Company Identification 1.1 Product identifiers Product Name : Sterile Diluent - 10% Glycero-Saline Product Code(s)
Understanding Tracheostomy Care for your Child
 Understanding Tracheostomy Care for your Child 2 Inside this guide: This guide will help you learn how to take care of your child s tracheostomy (trach). It is important to ask questions. You will be given
Understanding Tracheostomy Care for your Child 2 Inside this guide: This guide will help you learn how to take care of your child s tracheostomy (trach). It is important to ask questions. You will be given
Quantos Automated Dosing Solution Preparation Precise concentrations Process compliance Minimize out-of-specs
 Quantos Automated Dosing Solution Preparation Precise concentrations Process compliance Minimize out-of-specs Gravimetric Sample Preparation The Alternative to Volumetric Flasks From Volumetric to Gravimetric
Quantos Automated Dosing Solution Preparation Precise concentrations Process compliance Minimize out-of-specs Gravimetric Sample Preparation The Alternative to Volumetric Flasks From Volumetric to Gravimetric
Bt-Cry1Ac & Bt-Cry2A Multi-trait ELISA Kit DAS ELISA for the detection of Bt-Cry1Ac & Bt-Cry2A proteins Catalog number: PSM 14900
 List of contents Lot number Item 480 wells 4800 wells Antibody-coated 96-well microtiter plates 5 strip 50 solid Alkaline phosphatase enzyme conjugate, concentrated 0.550 ml 5 x 1.1 ml Peroxidase enzyme
List of contents Lot number Item 480 wells 4800 wells Antibody-coated 96-well microtiter plates 5 strip 50 solid Alkaline phosphatase enzyme conjugate, concentrated 0.550 ml 5 x 1.1 ml Peroxidase enzyme
MATERIAL SAFETY DATA SHEET
 GE Healthcare OMNIPAQUE (Iohexal) Injection MATERIAL SAFETY DATA SHEET 1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION Product Name: OMNIPAQUE (140, 180, 240, 300 & 350 mgi/ml) Synonyms: None Manufacturer/Distributor:
GE Healthcare OMNIPAQUE (Iohexal) Injection MATERIAL SAFETY DATA SHEET 1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION Product Name: OMNIPAQUE (140, 180, 240, 300 & 350 mgi/ml) Synonyms: None Manufacturer/Distributor:
Understanding Tracheostomy Care
 Understanding Tracheostomy Care Inside this guide: This guide will help you learn how to take care of your tracheostomy (trach). It is important to ask questions. You will be given time to learn. Working
Understanding Tracheostomy Care Inside this guide: This guide will help you learn how to take care of your tracheostomy (trach). It is important to ask questions. You will be given time to learn. Working
Making Life Easier Easypump II
 Making Life Easier Easypump II The elastomeric pump system for short- and long-term infusion therapy Elastomeric Infusion Systems Technical Guide Easypump II Disposable Elastomeric Infusion Pump System
Making Life Easier Easypump II The elastomeric pump system for short- and long-term infusion therapy Elastomeric Infusion Systems Technical Guide Easypump II Disposable Elastomeric Infusion Pump System
CspB ELISA Kit DAS ELISA for the detection of CspB protein Catalog number: PSP 35500
 List of contents Lot number Item 480 wells 4800 wells Antibody-coated 96-well microtiter plates 5 strip 50 solid Peroxidase enzyme conjugate, concentrated 0.550 ml 5 x 1.1 ml RUB6 enzyme conjugate diluent
List of contents Lot number Item 480 wells 4800 wells Antibody-coated 96-well microtiter plates 5 strip 50 solid Peroxidase enzyme conjugate, concentrated 0.550 ml 5 x 1.1 ml RUB6 enzyme conjugate diluent
CGMP KITS PROTOCOL ASSAY PRINCIPLE
 CGMP KITS PROTOCOL Part # 62GM2PEG & 62GM2PEH Test size#: 500 tests (62GM2PEG), 10,000 tests (62GM2PEH) - assay volume: 20 µl Revision: 02-Jan.2018 Store at: 2-8 C (62GM2PEG); 2-8 C (62GM2PEH) For research
CGMP KITS PROTOCOL Part # 62GM2PEG & 62GM2PEH Test size#: 500 tests (62GM2PEG), 10,000 tests (62GM2PEH) - assay volume: 20 µl Revision: 02-Jan.2018 Store at: 2-8 C (62GM2PEG); 2-8 C (62GM2PEH) For research
Proficiency Testing Corrective Action Checklist: PT PROVIDER: PT EVENT: TEST:
 Proficiency Testing Corrective Action Checklist: PT PROVIDER: PT EVENT: TEST: PT Process Package Received Who received it? Handling Upon arrival Was the kit cold? Was the kit damaged? Was the kit complete?
Proficiency Testing Corrective Action Checklist: PT PROVIDER: PT EVENT: TEST: PT Process Package Received Who received it? Handling Upon arrival Was the kit cold? Was the kit damaged? Was the kit complete?
ab VEGF Human ELISA Kit
 ab100663 VEGF Human ELISA Kit Instructions for Use For the quantitative measurement of Human VEGF in cell lysates and tissue lysates. This product is for research use only and is not intended for diagnostic
ab100663 VEGF Human ELISA Kit Instructions for Use For the quantitative measurement of Human VEGF in cell lysates and tissue lysates. This product is for research use only and is not intended for diagnostic
McGill University Health Center Pediatric Drug Infusion Preparation Chart
 McGill University Health Center Pediatric Drug Preparation Chart Tableau de préparation d infusion en Pédiatrie au Centre Universitaire de Santé McGill The purpose of the current chart is to: 1. Establish
McGill University Health Center Pediatric Drug Preparation Chart Tableau de préparation d infusion en Pédiatrie au Centre Universitaire de Santé McGill The purpose of the current chart is to: 1. Establish
Assisting with Insertion. Care of Intraspinal Catheters
 Guidelines included: Assisting with an Insertion Care of Various types of Intraspinal s Care of the Intraspinal Infusion Monitoring Removal of the Short Term Non Assisting with Insertion INR should be
Guidelines included: Assisting with an Insertion Care of Various types of Intraspinal s Care of the Intraspinal Infusion Monitoring Removal of the Short Term Non Assisting with Insertion INR should be
PRODUCT SAFETY DATA SHEET
 PRODUCT SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE / PREPARATION Name: Address: Foscavir (foscarnet sodium) Injection Clinigen Healthcare Limited Pitcairn House Crown Square First Avenue Burton-on-Trent
PRODUCT SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE / PREPARATION Name: Address: Foscavir (foscarnet sodium) Injection Clinigen Healthcare Limited Pitcairn House Crown Square First Avenue Burton-on-Trent
Contacts. Quick Start Guide
 Contacts Clinical Support Specialist: Phone: Cell Phone: Email: Fresenius Renal Technologies A division of Fresenius Medical Care North America 920 Winter Street Waltham, MA 02451 Technical Service Customer
Contacts Clinical Support Specialist: Phone: Cell Phone: Email: Fresenius Renal Technologies A division of Fresenius Medical Care North America 920 Winter Street Waltham, MA 02451 Technical Service Customer
Kcentra SDS Revision Date: 06/03/2015
 1. Identification 1.1. Product identifier Product Identity Alternate Names Kcentra Kcentra 1.2. Relevant identified uses of the substance or mixture and uses advised against Intended use Application Method
1. Identification 1.1. Product identifier Product Identity Alternate Names Kcentra Kcentra 1.2. Relevant identified uses of the substance or mixture and uses advised against Intended use Application Method
STANDARD OPERATING PROCEDURE ADVANCED INTRAVENOUS TRAINING ARM (S )
 Page 1 of 27 1. Scope This Standard Operating Procedure (SOP) applies to the staff and students using the Advanced Intravenous Training Arm in the Pharmacy Practice Resource Unit (PPRU) at the Pharmacy
Page 1 of 27 1. Scope This Standard Operating Procedure (SOP) applies to the staff and students using the Advanced Intravenous Training Arm in the Pharmacy Practice Resource Unit (PPRU) at the Pharmacy
Independent Health Facilities
 Independent Health Facilities Assessment Protocol for Pulmonary Function Studies - Technologist Assessor INSTRUCTIONS: Please complete ( ) the attached protocol during the assessment. Ensure that all the
Independent Health Facilities Assessment Protocol for Pulmonary Function Studies - Technologist Assessor INSTRUCTIONS: Please complete ( ) the attached protocol during the assessment. Ensure that all the
MSD 96-Well MULTI-ARRAY and MULTI-SPOT Human Granulocyte Colony Stimulating Factor (hg-csf) Ultrasensitive Assay
 MSD 96-Well MULTI-ARRAY and MULTI-SPOT Human Granulocyte Colony Stimulating Factor (hg-csf) Ultrasensitive Assay Summary This assay measures Human Granulocyte Colony Stimulating Factor (G-CSF) in a 96-well
MSD 96-Well MULTI-ARRAY and MULTI-SPOT Human Granulocyte Colony Stimulating Factor (hg-csf) Ultrasensitive Assay Summary This assay measures Human Granulocyte Colony Stimulating Factor (G-CSF) in a 96-well
Corifact SDS Revision Date: 06/04/2015
 1. Identification 1.1. Product identifier Product Identity Alternate Names Corifact Factor XIII Concentrate (Human) 1.2. Relevant identified uses of the substance or mixture and uses advised against Intended
1. Identification 1.1. Product identifier Product Identity Alternate Names Corifact Factor XIII Concentrate (Human) 1.2. Relevant identified uses of the substance or mixture and uses advised against Intended
RayBio Human vwf ELISA Kit
 RayBio Human vwf ELISA Kit Catalog #: ELH-vWF User Manual Last revised April 15, 2016 Caution: Extraordinarily useful information enclosed ISO 13485 Certified 3607 Parkway Lane, Suite 100 Norcross, GA
RayBio Human vwf ELISA Kit Catalog #: ELH-vWF User Manual Last revised April 15, 2016 Caution: Extraordinarily useful information enclosed ISO 13485 Certified 3607 Parkway Lane, Suite 100 Norcross, GA
Big Wipes 4x4 Health & Safety Data Sheet
 November 2015 Big Wipes 4x4 Health & Safety Data Sheet Product names: Heavy-Duty, Multi-Surface, Multi-Purpose ALL SIZES T: +44 (0)1628 567 700 F: +44 (0)1628 567 701 SALES@BIGWIPES.COM Page 1 of 6 M SAFETY
November 2015 Big Wipes 4x4 Health & Safety Data Sheet Product names: Heavy-Duty, Multi-Surface, Multi-Purpose ALL SIZES T: +44 (0)1628 567 700 F: +44 (0)1628 567 701 SALES@BIGWIPES.COM Page 1 of 6 M SAFETY
Multiplate Platelet Function Analyzer: An Overview
 Multiplate Platelet Function Analyzer: An Overview The Multiplate is for research use only in the US and Canada Scope: This is a summary of most aspects of platelet function testing using the Multiplate
Multiplate Platelet Function Analyzer: An Overview The Multiplate is for research use only in the US and Canada Scope: This is a summary of most aspects of platelet function testing using the Multiplate
SAFETY DATA SHEET. In accordance with 29 CFR Section 1. Identification of the substance or the mixture and of the supplier
 Page ( 6 (/4/206 Section. Identification of the substance or the mixture and of the supplier. Product Identifier Product identifier: Other identifiers:.2 Relevant identified uses of the substance of mixture
Page ( 6 (/4/206 Section. Identification of the substance or the mixture and of the supplier. Product Identifier Product identifier: Other identifiers:.2 Relevant identified uses of the substance of mixture
96-well Serum/Plasma Fatty Acid Kit Non-Esterified Fatty Acids Detection 100 point kit Cat# SFA-1
 96-well Serum/Plasma Fatty Acid Kit on-esterified Fatty Acids Detection 100 point kit Cat# SFA-1 ISTRUCTIO MAUAL ZBM0021.04 STORAGE CODITIOS Reagents & Buffers: 4 C Blank assay plates (96-well): Room Temperature
96-well Serum/Plasma Fatty Acid Kit on-esterified Fatty Acids Detection 100 point kit Cat# SFA-1 ISTRUCTIO MAUAL ZBM0021.04 STORAGE CODITIOS Reagents & Buffers: 4 C Blank assay plates (96-well): Room Temperature
ab FGF basic (FGF2) Human ELISA Kit
 ab99979 - FGF basic (FGF2) Human ELISA Kit Instructions for Use For the quantitative measurement of Human FGF basic in serum, plasma, and cell culture supernatants. This product is for research use only
ab99979 - FGF basic (FGF2) Human ELISA Kit Instructions for Use For the quantitative measurement of Human FGF basic in serum, plasma, and cell culture supernatants. This product is for research use only
SAFETY DATA SHEET. In accordance with 29 CFR Section 1. Identification of the substance or the mixture and of the supplier
 Page ( 6 Issue date: 02/02/207 (02/02/207 In accordance with 29 CFR 90.200 Section. Identification of the substance or the mixture and of the supplier. Product Identifier Product identifier: Other identifiers:.2
Page ( 6 Issue date: 02/02/207 (02/02/207 In accordance with 29 CFR 90.200 Section. Identification of the substance or the mixture and of the supplier. Product Identifier Product identifier: Other identifiers:.2
Patients Guide for Self Administering Intravenous Antibiotics General points
 Patients Guide for Self Administering Intravenous Antibiotics General points An intravenous line is an open route for infection therefore you need to prevent this by washing your hands before any procedure
Patients Guide for Self Administering Intravenous Antibiotics General points An intravenous line is an open route for infection therefore you need to prevent this by washing your hands before any procedure
ab99968 Adiponectin Human ELISA Kit
 ab99968 Adiponectin Human ELISA Kit Instructions for Use For the quantitative measurement of Human Adiponectin in serum, plasma and cell culture supernatants. This product is for research use only and
ab99968 Adiponectin Human ELISA Kit Instructions for Use For the quantitative measurement of Human Adiponectin in serum, plasma and cell culture supernatants. This product is for research use only and
ab Sonic Hedgehog Human ELISA Kit
 ab100639 Sonic Hedgehog Human ELISA Kit Instructions for Use For the quantitative measurement of Human Sonic Hedgehog in serum, plasma and cell culture supernatants. This product is for research use only
ab100639 Sonic Hedgehog Human ELISA Kit Instructions for Use For the quantitative measurement of Human Sonic Hedgehog in serum, plasma and cell culture supernatants. This product is for research use only
extraction of EG and DEG from the matrix. However, the addition of all diluent at once resulted in poor recoveries.
 Informal Commentary Limit of Diethylene Glycol (DEG) and Ethylene Glycol (EG) in Sorbitol Solution, Sorbitol Sorbitan Solution and Noncrystallizing Sorbitol Solution December 2009 Monograph/Section(s):
Informal Commentary Limit of Diethylene Glycol (DEG) and Ethylene Glycol (EG) in Sorbitol Solution, Sorbitol Sorbitan Solution and Noncrystallizing Sorbitol Solution December 2009 Monograph/Section(s):
ab HB EGF Human ELISA Kit
 ab100531 HB EGF Human ELISA Kit Instructions for Use For the quantitative measurement of Human HB EGF in serum, plasma, and cell culture supernatants. (Human HB EGF concentration is pretty low in normal
ab100531 HB EGF Human ELISA Kit Instructions for Use For the quantitative measurement of Human HB EGF in serum, plasma, and cell culture supernatants. (Human HB EGF concentration is pretty low in normal
Bt-Cry2Ab and Bt-Cry3Bb1 Multi-trait ELISA Kit DAS ELISA for the detection of Bt-Cry2Ab and Bt-Cry3Bb1 proteins Catalog number: PSM 26800
 List of contents Lot number Item 480 wells 4800 wells Antibody-coated 96-well microtiter plates 5 strip 50 solid Alkaline phosphatase enzyme conjugate, concentrated 0.550 ml 5 x 1.1 ml Peroxidase enzyme
List of contents Lot number Item 480 wells 4800 wells Antibody-coated 96-well microtiter plates 5 strip 50 solid Alkaline phosphatase enzyme conjugate, concentrated 0.550 ml 5 x 1.1 ml Peroxidase enzyme
Ocular Irritation: Dilution Method
 Protocol Ocular Irritation: Dilution Method For Use with EpiOcular Tissue Model (OCL-200) Overview: This protocol utilizes the MTT tissue viability assay to determine exposure time for a test article to
Protocol Ocular Irritation: Dilution Method For Use with EpiOcular Tissue Model (OCL-200) Overview: This protocol utilizes the MTT tissue viability assay to determine exposure time for a test article to
BD Cytopeia Fluidic Kit User s Guide
 BD Cytopeia Fluidic Kit User s Guide For Research Use Only 23-17618-00 9/2015 Becton, Dickinson and Company BD Biosciences 2350 Qume Drive San Jose, CA 95131 USA BD Biosciences European Customer Support
BD Cytopeia Fluidic Kit User s Guide For Research Use Only 23-17618-00 9/2015 Becton, Dickinson and Company BD Biosciences 2350 Qume Drive San Jose, CA 95131 USA BD Biosciences European Customer Support
1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY/UNDERTAKING
 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY/UNDERTAKING Product Name: STERILE ULTRASOUND GEL Use/description of product: ULTRASOUND GEL Company Name : TURKUAZ MEDICAL CO. Telephone:
1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY/UNDERTAKING Product Name: STERILE ULTRASOUND GEL Use/description of product: ULTRASOUND GEL Company Name : TURKUAZ MEDICAL CO. Telephone:
New. The high security connection
 L ON Cata C lo gu e O O G Y New The high security connection Preparation devices for medications/solutions Bag qimospike B 7218.01 qimospike B is an IV fluid bag access spike fitted with qimo. Aspiration
L ON Cata C lo gu e O O G Y New The high security connection Preparation devices for medications/solutions Bag qimospike B 7218.01 qimospike B is an IV fluid bag access spike fitted with qimo. Aspiration
Reconstitution and Administration Instructions for Healthcare Professionals.
 Reconstitution and Administration Instructions for Healthcare Professionals. Indication CINRYZE (C1 esterase inhibitor [human]) is indicated for routine prophylaxis against angioedema attacks in adolescent
Reconstitution and Administration Instructions for Healthcare Professionals. Indication CINRYZE (C1 esterase inhibitor [human]) is indicated for routine prophylaxis against angioedema attacks in adolescent
ab IL-4 (Interleukin-4) Mouse ELISA Kit
 ab100710 IL-4 (Interleukin-4) Mouse ELISA Kit Instructions for Use For the quantitative measurement of mouse IL-4 in cell lysatse and tissue lysates. This product is for research use only and is not intended
ab100710 IL-4 (Interleukin-4) Mouse ELISA Kit Instructions for Use For the quantitative measurement of mouse IL-4 in cell lysatse and tissue lysates. This product is for research use only and is not intended
Safety Data Sheet. Section 1: Product and Company Information. Section 2: Hazards Identification
 Product Information Safety Data Sheet Section 1: Product and Company Information Product Name Reishi Mushroom P.E. Product Code GF-REM4R1 Botanical Name Ganoderma Lucidium Part Used Fruit tops Shelf Life
Product Information Safety Data Sheet Section 1: Product and Company Information Product Name Reishi Mushroom P.E. Product Code GF-REM4R1 Botanical Name Ganoderma Lucidium Part Used Fruit tops Shelf Life
Material Safety Data Sheet acc. to ISO/DIS 11014
 Page 1 of 6 1 Identification of substance: Product Details: MSDS Number: VS60328-01 Issue Date: September 29, 2005 Supersedes: VS60328-00 ECO No.: ECO36501 Catalog Numbers: 365830 Manufacturer/Supplier:
Page 1 of 6 1 Identification of substance: Product Details: MSDS Number: VS60328-01 Issue Date: September 29, 2005 Supersedes: VS60328-00 ECO No.: ECO36501 Catalog Numbers: 365830 Manufacturer/Supplier:
SAFETY DATA SHEET. In accordance with 29 CFR Section 1. Identification of the substance or the mixture and of the supplier
 Page ( 5 (/29/206 Section. Identification of the substance or the mixture and of the supplier. Product Identifier Product identifier: Other identifiers: Glycerine Glycerol; CAS: 56-8-5.2 Relevant identified
Page ( 5 (/29/206 Section. Identification of the substance or the mixture and of the supplier. Product Identifier Product identifier: Other identifiers: Glycerine Glycerol; CAS: 56-8-5.2 Relevant identified
SAFETY DATA SHEET. In accordance with 29 CFR Section 1. Identification of the substance or the mixture and of the supplier
 Page ( 6 (02/03/207 Kiwi (Double Flavor 34674 Section. Identification of the substance or the mixture and of the supplier. Product Identifier Product identifier: Other identifiers: Kiwi (Double Flavor
Page ( 6 (02/03/207 Kiwi (Double Flavor 34674 Section. Identification of the substance or the mixture and of the supplier. Product Identifier Product identifier: Other identifiers: Kiwi (Double Flavor
Corning Step-R. Repeating Pipettor. Instruction Manual
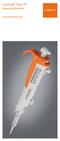 Corning Step-R Repeating Pipettor Instruction Manual Table of Contents 1. Introduction... 1 2. Using the Step-R Repeating Pipettor... 1 3. Corning Syringe Tips... 4 4. Recommendations... 5 5. Recalibration...
Corning Step-R Repeating Pipettor Instruction Manual Table of Contents 1. Introduction... 1 2. Using the Step-R Repeating Pipettor... 1 3. Corning Syringe Tips... 4 4. Recommendations... 5 5. Recalibration...
Material Safety Data Sheet
 Material Safety Data Sheet Chlamydia (IgG) -Ab ELISA Catalog Number: 20-CGGHU-E01 Page 2/13 Kit Components: Sample Diluent Pos. Control, Neg. Control, Cut-off Control, Standard Enzyme Conjugate Wash Solution
Material Safety Data Sheet Chlamydia (IgG) -Ab ELISA Catalog Number: 20-CGGHU-E01 Page 2/13 Kit Components: Sample Diluent Pos. Control, Neg. Control, Cut-off Control, Standard Enzyme Conjugate Wash Solution
MATERIAL SAFETY DATA SHEET SECTION 1. IDENTIFICATION OF SUBSTANCE AND CONTACT INFORMATION NOBIVAC LEPTO4
 Merck Animal Health One Merck Dr. Whitehouse Station, NJ 08889 MATERIAL SAFETY DATA SHEET Merck Animal Health urges each user or recipient of this MSDS to read the entire data sheet to become aware of
Merck Animal Health One Merck Dr. Whitehouse Station, NJ 08889 MATERIAL SAFETY DATA SHEET Merck Animal Health urges each user or recipient of this MSDS to read the entire data sheet to become aware of
USP <797> LABELING ESTABLISHING BEYOND-USE DATES AND IN-USE TIMES
 USP LABELING ESTABLISHING BEYOND-USE DATES AND IN-USE TIMES Each CSP label must state the date beyond which the preparation cannot be used and must be discarded. A CSP may also be labeled with an
USP LABELING ESTABLISHING BEYOND-USE DATES AND IN-USE TIMES Each CSP label must state the date beyond which the preparation cannot be used and must be discarded. A CSP may also be labeled with an
Berirab P SDS Revision Date: 06/03/2015
 1. Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product Identity Alternate Names Berirab P Human rabies immunoglobulin 1.2. Relevant identified uses of
1. Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product Identity Alternate Names Berirab P Human rabies immunoglobulin 1.2. Relevant identified uses of
96-well Serum/Plasma Fatty Acid Kit Non-Esterified Fatty Acids Detection 500 Point Kit Cat# SFA-5
 96-well Serum/Plasma Fatty Acid Kit on-esterified Fatty Acids Detection 500 Point Kit Cat# SFA-5 ISTRUCTIO MAUAL ZBM0030.03 STORAGE CODITIOS Reagents & Buffers: 4 C All Zen-Bio Inc products are for research
96-well Serum/Plasma Fatty Acid Kit on-esterified Fatty Acids Detection 500 Point Kit Cat# SFA-5 ISTRUCTIO MAUAL ZBM0030.03 STORAGE CODITIOS Reagents & Buffers: 4 C All Zen-Bio Inc products are for research
MATERIAL SAFETY DATA SHEET. Known Hazardous Components CAS Number Percent. Stop Solution 99% Known Hazardous Components CAS Number Percent
 MATERIAL SAFETY DATA SHEET Product name: Tnf (Rat) ELISA Kit Catalog number: KA0280 1. Composition/Information on Ingredients Component Antibody coated microwells Size 96 wells Biotin-Conjugate antibody
MATERIAL SAFETY DATA SHEET Product name: Tnf (Rat) ELISA Kit Catalog number: KA0280 1. Composition/Information on Ingredients Component Antibody coated microwells Size 96 wells Biotin-Conjugate antibody
US - OSHA SAFETY DATA SHEET
 US - OSHA SAFETY DATA SHEET Issue Date 17-Apr-2015 Version 1 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY/UNDERTAKING Product identifier Product Name ActHIB Other means of identification
US - OSHA SAFETY DATA SHEET Issue Date 17-Apr-2015 Version 1 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND OF THE COMPANY/UNDERTAKING Product identifier Product Name ActHIB Other means of identification
MATERIAL SAFETY DATA SHEET
 MATERIAL SAFETY DATA SHEET BAYER HEALTHCARE LLC Biological Products Division 8368 US 70 West Clayton, NC 27520 TRANSPORTATION EMERGENCY NON-TRANSPORTATION CALL CHEMTREC...: (800) 424-9300 BAYER EMERGENCY
MATERIAL SAFETY DATA SHEET BAYER HEALTHCARE LLC Biological Products Division 8368 US 70 West Clayton, NC 27520 TRANSPORTATION EMERGENCY NON-TRANSPORTATION CALL CHEMTREC...: (800) 424-9300 BAYER EMERGENCY
1461BR Lindell Rd Las Vegas, NV (Customer Service) (800) USA (813) International (Chemtel 24 hrs)
 Safety Data Sheet 1461BR 1 Material and Supplier Identification Product Name: Description: Recommended use of the chemical and restrictions: Turbo DC Disinfectant/Cleaner/Sanitizer Use only for the purpose
Safety Data Sheet 1461BR 1 Material and Supplier Identification Product Name: Description: Recommended use of the chemical and restrictions: Turbo DC Disinfectant/Cleaner/Sanitizer Use only for the purpose
1 Designation of the substance or mixture, respectively, and of the company
 Metrohm AG CH-9100 Herisau Switzerland info@metrohm.com www.metrohm.com Material Safety Data Sheet Pursuant to Regulation (EC) No. 1272/2008 Storage solution, 250 ml June 19, 2013 8.106.7575EN fpe/ts Storage
Metrohm AG CH-9100 Herisau Switzerland info@metrohm.com www.metrohm.com Material Safety Data Sheet Pursuant to Regulation (EC) No. 1272/2008 Storage solution, 250 ml June 19, 2013 8.106.7575EN fpe/ts Storage
AQUADEX FLEXFLOW SYSTEM QUICK REFERENCE GUIDE
 AQUADEX FLEXFLOW SYSTEM QUICK REFERENCE GUIDE AQUADEX FLEXFLOW SYSTEM DISCLAIMER The quick reference guide is not intended to replace CHF Solutions, Inc. Aquadex FlexFlow Direction For Use (DFU). Always
AQUADEX FLEXFLOW SYSTEM QUICK REFERENCE GUIDE AQUADEX FLEXFLOW SYSTEM DISCLAIMER The quick reference guide is not intended to replace CHF Solutions, Inc. Aquadex FlexFlow Direction For Use (DFU). Always
ISO INTERNATIONAL STANDARD. Sleep apnoea breathing therapy Part 2: Masks and application accessories
 INTERNATIONAL STANDARD ISO 17510-2 Second edition 2007-10-01 Sleep apnoea breathing therapy Part 2: Masks and application accessories Thérapie respiratoire de l'apnée du sommeil Partie 2: Masques et accessoires
INTERNATIONAL STANDARD ISO 17510-2 Second edition 2007-10-01 Sleep apnoea breathing therapy Part 2: Masks and application accessories Thérapie respiratoire de l'apnée du sommeil Partie 2: Masques et accessoires
biosensis Mouse CXCL10/IP-10 ELISA Kit Protocol
 biosensis Mouse CXCL10/IP-10 ELISA Kit Protocol Catalogue No: BEK-2124-2P TABLE OF CONTENTS I Materials provided...2 II Equipment required but not supplied...2 III Technical hints....2 IV Storage of kit
biosensis Mouse CXCL10/IP-10 ELISA Kit Protocol Catalogue No: BEK-2124-2P TABLE OF CONTENTS I Materials provided...2 II Equipment required but not supplied...2 III Technical hints....2 IV Storage of kit
TJF-Q180V Cleaning and Disinfection Checklist
 TJF-Q180V Cleaning and Disinfection Checklist TJF-Q180V Cleaning and Disinfection Checklist This checklist is used to evaluate and confirm if cleaning and disinfection of the TJF-Q180V has been performed
TJF-Q180V Cleaning and Disinfection Checklist TJF-Q180V Cleaning and Disinfection Checklist This checklist is used to evaluate and confirm if cleaning and disinfection of the TJF-Q180V has been performed
1. IDENTIFICATION OF THE SUBSTANCE/MIXTURE AND OF THE COMPANY/UNDERTAKING
 SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE/MIXTURE AND OF THE COMPANY/UNDERTAKING Product identifier NEOSTIGMINE SOLUTION FOR Details of the supplier of the safety data sheet : ASTRAZENECA PTY
SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE/MIXTURE AND OF THE COMPANY/UNDERTAKING Product identifier NEOSTIGMINE SOLUTION FOR Details of the supplier of the safety data sheet : ASTRAZENECA PTY
SECTION III COMPOSITION/INFORMATION ON INGREDIENTS
 SECTION I PRODUCT & COMPANY IDENTIFICATION Product Name: Product Number: Manufacturer: Telephone: (877) 771 1093 Fax: (303) 357 9001 DNA Fragmentation, End Repair/dA- tailing SA0039 ArcherDX, Inc. 2477
SECTION I PRODUCT & COMPANY IDENTIFICATION Product Name: Product Number: Manufacturer: Telephone: (877) 771 1093 Fax: (303) 357 9001 DNA Fragmentation, End Repair/dA- tailing SA0039 ArcherDX, Inc. 2477
Monosed ESR Non-Vacuum Tube USER S MANUAL
 Monosed ESR Non-Vacuum Tube USER S MANUAL Manual code MAN-134 Revision 02 Revised date: November 2015 Instructions for use for in vitro diagnostic instruments for professional use. PRODUCT NAME PRODUCT
Monosed ESR Non-Vacuum Tube USER S MANUAL Manual code MAN-134 Revision 02 Revised date: November 2015 Instructions for use for in vitro diagnostic instruments for professional use. PRODUCT NAME PRODUCT
TF (Human) Chromogenic Activity Assay Kit
 TF (Human) Chromogenic Activity Assay Kit Catalog Number KA0975 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the
TF (Human) Chromogenic Activity Assay Kit Catalog Number KA0975 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the
DEXMEDETOMIDINE HYDROCHLORIDE INJ
 1. PRODUCT AND COMPANY INFORMATION Distributed By: 120 Route 17 North Suite 115 Paramus, NJ 07652 USA Product Name: Primary Component/ Active Ingredient: DEXMEDETOMIDINE HYDROCHLORIDE INJ Dexmedetomidine
1. PRODUCT AND COMPANY INFORMATION Distributed By: 120 Route 17 North Suite 115 Paramus, NJ 07652 USA Product Name: Primary Component/ Active Ingredient: DEXMEDETOMIDINE HYDROCHLORIDE INJ Dexmedetomidine
Material safety data sheet According to the Regulation (EC) No: 1907/2006
 Material safety data sheet According to the Regulation (EC) No: 1907/2006 Date: February 9 th 2016 Former Date: June 1 st 2015 SECTION 1: IDENTIFICATION OF THE SUBSTANCE / MIXTURE AND OF THE COMPANY /
Material safety data sheet According to the Regulation (EC) No: 1907/2006 Date: February 9 th 2016 Former Date: June 1 st 2015 SECTION 1: IDENTIFICATION OF THE SUBSTANCE / MIXTURE AND OF THE COMPANY /
Aseptic processing. the starting material to the finished product, should be controlled by Validation Control of the aseptic process
 10 Aseptic processing When products are prepared aseptically in unlicensed units there is usually no possibility of quality control of the product before release and use. Consequently quality assurance
10 Aseptic processing When products are prepared aseptically in unlicensed units there is usually no possibility of quality control of the product before release and use. Consequently quality assurance
Material Safety Data Sheet PROFORCE NO RINSE FLOOR CLEANER
 Material Safety Data Sheet PROFORCE NO RINSE FLOOR CLEANER 1. Product and company identification Trade name of product : PROFORCE NO RINSE FLOOR CLEANER Product use : Product dilution information : Up
Material Safety Data Sheet PROFORCE NO RINSE FLOOR CLEANER 1. Product and company identification Trade name of product : PROFORCE NO RINSE FLOOR CLEANER Product use : Product dilution information : Up
A high flying simplicity DUAL SOFT BAG INJECTION SYSTEM
 A high flying simplicity DUAL SOFT BAG INJECTION SYSTEM THE INNOVATIVE SOLUTION for contrast media and saline injection in CT medical imaging Designed for MULTI-USE Dual soft bag injector for MONO & BI-LIQUID
A high flying simplicity DUAL SOFT BAG INJECTION SYSTEM THE INNOVATIVE SOLUTION for contrast media and saline injection in CT medical imaging Designed for MULTI-USE Dual soft bag injector for MONO & BI-LIQUID
Ceftriaxone for Injection, USP 500mg Vial: 1g Vial: 2g Vial: 10g Vial:
 1. PRODUCT AND COMPANY INFORMATION Distributed By: 120 Route 17 North Suite 115 Paramus, NJ 07652 USA Product Name: Product Code: Common / Trade Name: 500mg Vial: 1g Vial: 2g Vial: 10g Vial: 44567-700-25
1. PRODUCT AND COMPANY INFORMATION Distributed By: 120 Route 17 North Suite 115 Paramus, NJ 07652 USA Product Name: Product Code: Common / Trade Name: 500mg Vial: 1g Vial: 2g Vial: 10g Vial: 44567-700-25
Safety Data Sheet 1. PRODUCT AND COMPANY IDENTIFICATION. Telephone: or Fax: Emergency Phone #:
 Page 1 of 9 Safety Data Sheet 1121 5 th Street NW Washington, DC 20001 www.edvotek.com Version 2 Revision Date 10/24/2014 1. PRODUCT AND COMPANY IDENTIFICATION Product name: Cadmium chloride Product Number:
Page 1 of 9 Safety Data Sheet 1121 5 th Street NW Washington, DC 20001 www.edvotek.com Version 2 Revision Date 10/24/2014 1. PRODUCT AND COMPANY IDENTIFICATION Product name: Cadmium chloride Product Number:
TECHNICAL BULLETIN. TriControls. Control Materials for Liquid QC and Calibration Verification
 i-stat TECHNICAL BULLETIN Tris Materials for Liquid QC and OVERVIEW As part of the READi initiative (Responds, Enhances, And Delivers innovations), Abbott Point of Care (APOC) has released a new set of
i-stat TECHNICAL BULLETIN Tris Materials for Liquid QC and OVERVIEW As part of the READi initiative (Responds, Enhances, And Delivers innovations), Abbott Point of Care (APOC) has released a new set of
