Layered SnS2 -Reduced Graphene Oxide Composite A High-Capacity, High-Rate, and Long-Cycle Life Sodium-Ion Battery Anode Material
|
|
|
- Horatio Allison
- 6 years ago
- Views:
Transcription
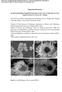
1 Layered SnS2 -Reduced Graphene Oxide Composite A High-Capacity, High-Rate, and Long-Cycle Life Sodium-Ion Battery Anode Material Baihua Qu, Chuze Ma, Ge Ji, Chaohe Xu, Jing Xu, Ying Shirley Meng, Taihong Wang, * and Jim Yang Lee * The idea of sodium-ion batteries (NIBs) as a substitute of lithium-ion batteries (LIBs) for grid-scale energy storage was initially driven by cost considerations. [1 ] Research in the last several years has shown that NIBs are not necessarily deficient in performance. [2 ] Hence there is a strong current interest in developing high-performance NIB anode [3 ] and cathode [4 ] materials. Thus far tin and tin-based compounds have shown good prospect as high-capacity NIB anodes based on the theoretical stoichiometry of Na 15 Sn 4 (847 mah g 1 ). [5 15 ] For example, Liu et al. [9 ] reported initial capacity of 722 mah g 1 and capacity of 405 mah g 1 after 150 cycles at 50 ma g 1 for a composite Sn anode consisting of an array of nanorods with a carbon outer shell, a Sn intermediate layer and a metal inner core (3D Sn nanoforests). Anodes which were based on SnO 2 (432 mah g 1 after 150 cycles at 20 ma g 1 ) [10 ] and SnO nanocomposite (638 mah g 1 after 100 cycles at 20 ma g 1 ) [11 ] have also been demonstrated. Wu and co-workers [12 ] examined Sn SnS C nanocomposites as NIB anodes and reported reversible capacities of 664 mah g 1 at 20 ma g 1 and 350 mah g 1 at 800 ma g 1. These are clearly encouraging developments but a high-capacity tin-based anode with good rate performance and long cycle life has yet to be identified. The lack of cycle stability is often attributed to the deleterious volume expansion in tin-sodium alloy formation (a 420% volume expansion upon the formation of Na 15 Sn 4 ) resulting in electrode disintegration and gradual loss of electrical contact with the current collector. [13,16 ] The situation is similar to the early days of the development of alloy anodes for B. Qu, Prof. T. Wang Key Laboratory for Micro-Nano Optoelectronic Devices of Ministry of Education and State Key Laboratory of Chemo/Biosensing and Chemometrics Hunan University Changsha , P. R. China thwang@hnu.edu.cn B. Qu, Dr. G. Ji, Dr. C. Xu, Prof. J. Y. Lee Department of Chemical and Biomolecular Engineering National University of Singapore 10 Kent Ridge Crescent , Singapore cheleejy@nus.edu.sg C. Ma, J. Xu, Dr. Y. S. Meng Department of NanoEngineering University of California San Diego, La Jolla, CA 92037, USA DOI: /adma the LIBs. Considerable improvements in the design and optimization of anode composition and structure are still required. This note reports our design and implementation of a SnS 2 - based nanocomposite anode for the NIBs. SnS 2 has a CdI 2 -type of layered structure (a = nm, c = nm, space group P3m1) consisting of a layer of tin atoms sandwiched between two layers of hexagonally close packed sulfur atoms. This layered structure with a large interlayer spacing (c = nm) should easy the insertion and extraction of guest species and adapt more easily to the volume changes in the host during cycling. This has been confirmed by the performance of SnS 2 as a reversible lithium storage host in several studies. [17 ] The electrochemical properties of layered sulfides (SnS 2, MoS2, WS2 ) were further improved by integration with graphene. The structural compatibility between the two layered compounds and the good electronic properties of graphene led to very stable composites (i.e. long cycle-life) with high reversible capacity and good rate performance in LIB applications. [18 ] The SnS 2 layer structure should also be viable for reversible Na + storage since, in comparison with tin and other tin-based materials, it has the largest buffer for the volume changes in Na-Sn reactions. The LIB developmental efforts also suggest layer-structured SnS 2 - reduced graphene oxide (SnS 2 -RGO) nanocomposites as an improved version of the SnS 2 anode. The design of the SnS 2 -RGO hybrid structure for reversible storage of Na + was based on the following materials principles: 1) a large interlayer spacing in the SnS 2 structure benefiting Na + intercalation and diffusion, and more buffering space for beneficial adjustment the volume changes in the host during cycling; 2) fast collection and conduction of electrons through a highly conductive RGO network; and 3) inhibition of Sn (Na x Sn) aggregation during cycling by RGO after material hybridization. The experimental results validated the expectations: the SnS 2 -RGO anode delivered a high charge (desodiation) specific capacity of 630 mah g 1 at 0.2 A g 1, and more impressively, 544 mah g 1 after a ten-fold increase in current density to 2 A g 1. The electrode was also very stable to cycling; providing a nearly unvarying capacity of 500 mah g 1 at 1 A g 1 even after 400 charge-discharge cycles. The SnS2 -RGO nanocomposite was produced by a facile hydrothermal route from a mixture of tin (IV) chloride, thioacetamide (TAA) and graphene oxide (GO) (details in the Experimental Section). In the comparison of the X-ray diffraction (XRD) patterns of the SnS 2 -RGO composite, SnS 2 and GO in Figure 1 a, GO only displayed a single diffraction peak at 10.9 from the (002) planes. [19 ] The powder XRD patterns of SnS 2 and 3854
2 Figure 1. a) XRD patterns. (b) Raman spectra of SnS2, GO, and the SnS2-RGO composite. c) A representative FESEM image of the SnS2-RGO composite. d) A HRTEM image of the SnS2-RGO composite. SnS2-RGO index well with the 2T-type layered structure (space group: P3m1, JCPDS card no );[17] and there was no significant impurity in the composite. The absence of the GO peak at 10.9 in the XRD pattern of SnS2-RGO confirms the formation of RGO during the hydrothermal treatment.[18b] The GO, SnS2 and SnS2-RGO samples were also characterized by Raman spectroscopy (Figure 1b). GO could be characterized by the graphitic-g band at 1350 cm 1 and the disorder-induced D band at 1580 cm 1. SnS2 could be characterized by a peak at 311 cm 1 corresponding to the A1g mode of SnS2,[18a,b,20] which was present in the spectra of SnS2 and SnS2-RGO. The intensity ratio of the D-band to G-band of GO (ID/IG) was higher in SnS2-RGO; indicating a reduction in the average size of the sp2 domains after GO reduction.[21] The field-emission scanning electron microscopy (FESEM) image of the SnS2-RGO composite (Figure 1c) shows a homogeneous structure with many large but thin sheets (several hundred nanometers in size). High resolution transmission electron microscopy (HRTEM) revealed the composite nature of the sheets consisting of a distributed of the SnS2 nanosheets ( 10 layers) among the RGO sheets. Figure 1d shows the interplanar spacing of 0.59 nm corresponding well with the (001) plane of 2T-type layered structure SnS2. Cyclic voltammetry (CV) was performed in the 0 to 2.5 V potential region at 0.1 mv s 1 (Figure 2a). Although the storage mechanism has yet to be exactly known, there are some consensuses about the common voltammetric features: The very broad peak at V in the first cathodic process (reduction) is commonly assigned to sodium intercalation of the SnS2 layers without phase decomposition (similar to the lithium intercalation of the SnS2 layers).[17d] The sharp and intense cathodic peak at 0.55 V could be attributed to conversion and alloying reactions. As well as the formation of irreversible solid electrolyte interphase (SEI) in the 1st cycle. Indeed the cathodic peak at 0.55 V was replaced by two peaks at 0.62 and 0.9 V in subsequent cycles, an indication of the good reversibility and predominance of the storage reactions after a stable SEI had been formed in the 1st cycle. In the anodic scan, the inconspicuous oxidation peaks at 0.3 and 0.75 V in all cycles may be attributed to the desodiation reaction of NaxSn. The distinct oxidation peak at 1.2 V may correspond to the restitution of the original SnS2-RGO. The voltammograms were superimposable after the first cycle; indicating very good reversibility of SnS2-RGO for the sodiation and desodiation reactions. On deep galvanostatic cycling between 0.01 and 2.5 V, the discharge (sodiation) capacity was 839 mah g 1 and the charge (desodiation) capacity was 630 mah g 1 in the first cycle at a current density of 0.2 A g 1.The first cycle irreversible capacity loss of 209 mah g 1 (25%) could be attributed mainly to SEI formation (Figure 2b). The electrode showed good cycle stability from the 2nd cycle onwards, delivering discharge capacities of 645, 654, 628 mah g 1 in the 2nd, 20th and 100th cycle respectively. The overlapping voltage profiles in Figure 2b demonstrate the excellent electrochemical reversibility during cycling sustaining a coulombic efficiency of >99% from the 2nd cycle onwards (Figure S3). For comparison the performance of SnS2 and RGO was also measured under identical test conditions as shown in Figure 2c. It was confirmed that RGO contributed negligibly to Na+ storage ( 30 mah g 1). For the SnS2 electrode, not only was its specific capacity significantly lower than the SnS2-RGO electrode, there was also severe capacity fading resulting in a 3855
3 Figure 2. a) The fi rst fi ve cycle voltammograms of the SnS 2 -RGO electrode at 0.1 mv s 1. b) Voltage profi les of a SnS 2 -RGO electrode in the 1 st, 2nd, 20 th and 100 th cycles. c) Cycling performance of SnS 2 -RGO, SnS 2 and graphene electrodes at 0.2 A g 1 for 100 cycles, respectively. d) Rate performance of SnS 2 -RGO and SnS 2 electrodes. discharge capacity of only 113 mah g 1 after 100 cycles. Severe capacity fading could be due to the low electronic conductivity of unsupported SnS 2 and un-restrained aggregations of Sn (or Na x Sn) during cycling. The voltage profiles of the SnS 2 and RGO electrodes in the selected number of cycles can be found in Figure S1 and Figure S2. The rate capability of the anode material is an important performance indicator for grid-scale electricity storage and electric vehicle applications. Figure 2 d shows the rate capability of SnS 2 and SnS 2 -RGO from 0.1 to 2 A g 1. The SnS 2 - RGO electrode was clearly superior by comparison. The test began with the current density of 0.1 A g 1, where the discharge capacity of the SnS 2 -RGO electrode was consistently 670 mah g 1 for the first ten cycle. When the current density was increased in steps to 0.2, 0.5, and 1.0 A g 1, the capacity decrease was very small for a battery material. Even after a 20 fold increase in current density to 2.0 A g 1, a discharge capacity of 544 mah g 1 or 81% of the capacity at 0.1 A g 1 (671 mah g 1 ), could still be retained. The discharge capacities when the current density was returned to 0.5 and 0.2 A g 1 were 620 and 650 mah g 1, which were only marginally lower than the values before the current density increase. The negligible changes in discharge capacity with current density changes indicate the resilience of the SnS 2 - RGO hybrid structure. Evidently the large interlayer spacing allowed Na + transport to be carried out at high rates (high current densities) without causing irreversible changes to the hybrid structure. The typical voltage profiles of the SnS 2 - RGO electrode at 0.1, 0.2, 0.5, 1.0, and 2.0 A g 1 rates could be found in Figure S4. For the evaluation of the long-term cycle stability of the SnS 2 - RGO electrode, the electrode was galvanostatically discharged and charged at 0.2 A g 1 for the first five cycles; and then at 1 A g 1 for 400 cycles ( Figure 3 a). The discharge capacity of 594 mah g 1 in the 6 th cycle at 1 A g 1 was used as the baseline. The capacities at the end of 50, 100, 200 and 400 cycles were 566 mah g 1 (95%), 565 mah g 1 (95%), 552 mah g 1 (93%) and 500 mah g 1 (84%) respectively. The performance surpasses any other NIB anode reported in the literature to date (Table S1). There was practically no capacity decrease in the first two hundred cycles. The superior cycle stability of the SnS 2 - RGO electrode for reversible Na + storage was highly repeatable. We have tested three different batches of SnS 2 -RGO electrodes and the results are shown in Figure 3 b and 3 c. Figure 3 b shows the overlapping voltage profiles of these electrodes in the 200 th cycle. The cycling performance of the three different SnS 2 -RGO electrodes was also perfectly superimposable under the same test conditions (Figure 3c). To further confirmations of the practically of the SnS 2 -RGO composite for NIBs applications, the SnS 2 -RGO anode was paired up with P 2 phase Na 0.80 Li 0.12 Ni 0.22 Mn 0.66 O 2 cathode, which has a theoretical capacity of 118 mah g 1, to form into a Na + full cell. [22 ] The active materials were loaded on the cathode and anode in the ratio of 5.4:1 (mass loading of anode was 1.67 mg). The 1 st, 2nd, 5 th, 10th, and 15 th cycle chargedischarge curves of the full cell are shown in Figure 4a. The first charge capacity was mah and the initial capacity loss was mah (36%). The full cell displayed relatively good stability, retaining 74% of the initial capacity after 50 cycles Figure 4 b. The good coulombic efficiency was also replicated 3856
4 Figure 3. a) Long-term cycle stability of the SnS 2 -RGO electrode (0.2 A g 1 for the fi rst fi ve cycles and 1 A g 1 for the next 400 cycles. b) Voltage profi les of the three different SnS 2 -G electrodes in the 200 th cycle. c) The cycle stability of three different batches of SnS 2 -RGO electrodes. in full cell studies (Figure S5). This preliminary data also represents the best of sodium-ion full cells reported to date. [3e,23 ] The greater capacity fading in the full cell was most likely caused by an un-optimized anode to cathode mass ratio; and the lack of match the of the C-rates between the two electrode materials. These parameters are difficult to optimize at the current stage of research. In conclusion, we have developed a SnS 2 -RGO composite with excellent electrochemical performance as the anode of NIBs. The SnS 2 -RGO electrode demonstrated a high charge specific capacity (630 mah g 1 at 0.2 A g 1 ), good rate performance (544 mah g 1 at 2 A g 1 ) and long cycle-life (500 mah g 1 at 1 A g 1 for 400 cycles). The excellent electrochemical performance could categorically be attributed to the SnS 2 layered structure where the increased interlayer spacing could better accommodate the volume change in Na-Sn insertion and deinsertions; and the good conductivity and mechanical resilience of RGO nanocomposites. Figure 4. a) Voltage profi les of a full Na + cell consisting of Na 0.80 Li 0.12 Ni 0.22 Mn 0.66 O 2 cathode and SnS 2 -RGO anode between 1.0 and 4.2 V at ma. b) Cycling performance of the Na + full cell consisting of Na 0.80 Li 0.12 Ni0.22Mn 0.66 O 2 cathode and SnS 2 -RGO anode. 3857
5 Experimental Section Materials Synthesis and Characterization : The graphite powder from Alfa Aesar, tin (IV) chloride pentahydrate (SnCl 4 5H 2 O), thioacetamide (TAA) and carboxymethyl cellulose (CMC) from Sigma-Aldrich; and other analytical grade chemicals from various sources, were used as received without further purification. Deionized water was processed by Millipore Milli-Q to a resistivity of 18 MΩ cm 1 and higher. GO was prepared from the graphite powder by a modified Hummers method, [24 ] as reported previously. [25 ] Exfoliation was carried out by sonicating a GO dispersion (1.5 mg ml 1 ) under ambient conditions for 2 h. A typical synthesis of SnS 2 -RGO composite is given as follows: g SnCl 4 5H 2 O (2 mmol) and g thioacetamide (TAA) (8 mmol) were added to 40 ml of 1.5 mg ml 1 GO suspension under stirring. The mixture was sonicated for 30 min before it was transferred to a 50ml Teflon-lined stainless steel autoclave, and heated at 160 C for 12 h. After cooling to room temperature, the solid product was recovered by centrifugation, washed with deionized water and absolute alcohol thrice, and vacuum dried at 60 C overnight. The dried product was then heated in Ar at the rate of 5 C min 1 to 400 C and kept at this temperature for 4 h. Pure SnS 2 was synthesized under the same conditions but without the presence of GO. RGO was obtained by treating GO in an Ar/H 2 (5%) mixture at 800 C for 20 min. All solid products prepared above were characterized by fieldemission scanning electron microscopy (FESEM) on a JEOL JSM-6700F operating at 5 kv, by transmission electron microscopy (TEM) and Highresolution TEM (HRTEM) on a JEOL JEM-2010F operating at 200 kv, respectively. Powder X-ray diffractions (XRD) were recorded on a Shimadu XRD-6000 using Cu Kα radiation. Raman spectra were taken on a LabRAM HR 800 spectrometer at 633 nm. Electrochemical Tests : For electrochemical measurements, 80wt% SnS 2 -RGO (or SnS 2 or RGO), 10 wt% of conducting additive (acetylene black), and 10 wt% CMC were added to a distilled water-absolute alcohol mixture (3:2 by volume), and stirred for 8h to form a homogeneous slurry. The slurry was then applied to a copper foil and dried in vacuum at 80 C overnight to form the working electrode. The test cells were coin cells (CR2032) which were assembled in an Ar-fi lled glove box (H 2 O and O < 1ppm). Each cell typically contained mg cm 1 of the active material. For half-cell studies the counter electrode was a sodium metal foil (Sigma-Aldrich) and a glass fi ber GF/D (Whatman) fi lter was used as the separator. A 1 M solution of NaClO 4 (Sigma- Aldrich, 98%) in ethylene carbonate (EC)/diethylene carbonate (1:1 by volume) was prepared and used as the electrolyte. The cells were aged for 12 h before the measurements to ensure complete electrode wetting by the electrolyte. Galvanostatic discharge and charge at various current densities were performed on a battery tester (NEWARE BTS-5V, Neware Technology Co., Ltd.) with cut off potentials of 0.01 V for discharge and 2.5 V for charge. Cyclic voltammetry (CV) was performed on an AutoLab FRA2 type III electrochemical system at the scan rate of 0.1 mv s 1 at room temperature. Full Cell Fabrication : A Swagelok cell was used as the full cell. The cathode was a mixture of Na 0.80 Li 0.12 Ni 0.22 Mn 0.66 O 2, acetylene black and poly(tetrafluoroethylene) (PTFE) in the weight ratio of 85:10:5. Anode preparation followed the same procedures as those used in the half-cell studies. The cathode to anode weight ratio was 5.4:1. The electrolyte was 1 M NaPF 6 in a 1:1 (v/v) mixture of ethylene carbonate (EC) and diethylene carbonate (DEC). The separator was a Whatman GF/D glass fi ber fi lter paper. Battery assembly was carried out in an MBraun glovebox (H 2 O and O < 1 ppm). Galvanostatic discharge and charge were performed on an Arbin BT2000 battery cycler between V. Supporting Information Supporting Information is available from the Wiley Online Library or from the author. Acknowledgements This work was supported by a Scholarship Award for Excellent Doctoral Students from the Chinese Ministry of Education (2012). We thank the fi nancial support of National Natural Science Foundation of China (Grant No ). C. Ma and Y. S. Meng acknowledge the funding support by USA National Science Foundation under Award Number B. H. Qu would also like to acknowledge the China Scholarship Council (CSC) for providing his exchange scholarship for Ph. D. study at the National University of Singapore (NUS). Received: December 30, 2013 Revised: February 25, 2014 Published online: March 27, 2014 [1] a) B. Dunn, H. Kamath, J. M. Tarascon, Science 2011, 334, 928 ; b) Z. G. Yang, J. L. Zhang, M. C. W. K. Meyer, X. C. Lu, D. W. Choi, J. P. Lemmon, J. Liu, Chem. Rev. 2011, 111, [2] a) S. W. Kim, D. H. Seo, X. H. Ma, G. Ceder, K. Kang, Adv. Energy Mater. 2012, 2, 710 ; b) V. Palomares, P. Serras, I. Villaluenga, K. B. Hueso, J. C. Gonzalez, T. Rojo, Energy Environ. Sci. 2012, 5, 5884 ; c) M. D. Slater, D. Kim, E. Lee, C. S. Johnson, Adv. Funct. Mater. 2013, 23, 947 ; d) H. L. Pan, Y. S. Hu, L. Q. Chen, Energy Environ. Sci. 2013, 6, [3] a) V. L. Chevrier, G. Ceder, J. Electrochem. Soc. 2011, 158, A1011 ; b) K. Tang, L. J. Fu, R. J. White, L. H. Yu, M. M. Titirici, M. Antonietti, J. Maier, Adv. Energy Mater. 2012, 2, 873 ; c) A. Darwiche, C. Marino, M. T. Sougrati, B. Fraisse, L. Stievano, L. Monconduit, J. Am. Chem. Soc. 2012, 134, ; d) Y. J. Kim, Y. W. Park, A. Choi, N. S. Choi, J. Kim, J. Lee, J. H. Ryu, S. M. Oh, K. T. Lee, Adv. Mater. 2013, 25, 3045 ; e) Y. S. Wang, X. Q. Yu, S. Y. Xu, J. M. Bai, R. J. Xiao, Y. S. Hu, H. Li, X. Q. Yang, L. Q. Chen, X. J. Huang, Nat. Comm. 2013, 4, [4] a) Y. L. Cao, L. F. Xia, W. Wang, D. W. Choi, Z. M. Nie, J. G. Yu, L. V. Saraf, Z. G. Yang, J. Liu, Adv. Mater. 2011, 23, 3155 ; b) N. Yabuuchi, M. Kajiyama, J. Iwatate, H. Nishikawa, S. Hitomi, R. Okuyama, R. Usui, Y. Yamada, S. Komaba, Nat. Mater. 2012, 11, 512 ; c) Z. L. Jian, W. Z. Han, X. Lu, H. X. Yang, Y. S. Hu, J. Zhou, Z. B. Zhou, J. Q. Li, W. Chen, D. F. Chen, L. Q. Chen, Adv. Energy Mater. 2013, 3, 156 ; d) H. L. Chen, Q. Hao, O. Zivkovic, G. Hautier, L. S. Du, Y. Z. Tang, Y. Y. Hu, X. H. Ma, C. P. Grey, G. Ceder, Chem. Mater. 2013, 25, 2777 ; e) D. H. Lee, J. Xu, Y. S. Meng, Phys. Chem. Chem. Phys. 2013, 15, 3304 ; f) M. Guignard, C. Didier, J. Darriet, P. Bordet, E. Elkaïm, C. Delmas, Nat. Mater. 2013, 12, 74 ; g) L. Wang, Y. H. Lu, J. Liu, M. W. Xu, J. G. Cheng, D. W. Zhang, J. B. Goodenough, Angew. Chem. Int. Ed. 2013, 52, [5] S. Komaba, Y. Matsuura, T. Ishikawa, N. Yabuuchi, W. Murata, S. Kuze, Electrochem. Commun. 2012, 21, 65. [6] Y. H. Xu, Y. J. Zhu, Y. H. Liu, C. S. Wang, Adv. Energy Mater. 2013, 3, 128. [7] M. K. Datta, R. Epur, P. Saha, K. Kadakia, S. K. Park, P. N. Kumt, J. Power Sources 2013, 225, 316. [8] Y. Wang, D. W. Su, C. Y. Wang, G. X. Wang, Electrochem. Commun. 2013, 29, 8. [9] Y. H. Liu, Y. H. Xu, Y. J. Zhu, J. N. Culver, C. A. Lundgren, K. Xu, C. S. Wang, ACS Nano 2013, 7, [10] D. W. Su, C. Y. Wang, H. J. Ahn, G. X. Wang, Phys. Chem. Chem. Phys. 2013, 15, [11] D. W. Su, H. J. Ahn, G. X. Wang, Chem. Commun. 2013, 49, [12] L. Wu, X. H. Hu, J. F. Qian, F. Pei, F. Y. Wu, R. J. Mao, X. P. Ai, H. X. Yang, Y. L. Cao, J. Mater. Chem. A. 2013, 1, [13] H. L. Zhu, Z. Jia, Y. C. Chen, N. Weadock, J. Y. Wan, O. Vaaland, X. G. Han, T. Li, L. B. Hu, Nano Lett. 2013, 13,
6 [14] Y. M. Lin, P. R. Abel, A. Gupta, J. B. Goodenough, A. Heller, C. B. Mullins, ACS Appl. Mater. Inter. 2013, 5, [15] Y. X. Wang, Y. G. Lim, M. S. Park, S. L. Chou, J. H. Kim, H. K. Liu, S. X. Dou, Y. J. Kim, J. Mater. Chem. A. 2014, 2, 529. [16] J. W. Wang, X. H. Liu, S. X. Mao, J. Y. Huang, Nano Lett. 2012, 12, [17] a) J. W. Seo, J. T. Jang, S. W. Park, C. J. Kim, B. W. Park, J. Cheon, Adv. Mater. 2008, 20, 4269 ; b) C. X. Zhai, N. Du, H. Zhang, D. R. Yang, Chem. Commun. 2011, 47, 1270 ; c) Y. P. Du, Z. Y. Yin, X. H. Rui, Z. Y. Zeng, X. J. Wu, J. Q. Liu, Y. Y. Zhu, J. X. Zhu, X. Huang, Q. Y. Yan, H. Zhang, Nanoscale 2013, 5, 1456 ; d) Q. Wu, L. F. Jiao, J. Du, J. Q. Yang, L. J. Guo, Y. C. Liu, Y. J. Wang, H. T. Yuan, J. Power Sources 2013, 239, 89. [18] a) B. Luo, Y. Fang, B. Wang, J. S. Zhou, H. H. Song, L. J. Zhi, Energy Environ. Sci. 2012, 5, 5226 ; b) Z. F. Jiang, C. Wang, G. H. Du, Y. J. Zhong, J. Z. Jiang, J. Mater. Chem. 2012, 22, 9494 ; c) K. Chang, Z. Wang, G. C. Huang, H. Li, W. X. Chen, J. Y. Lee, J. Power Sources 2012, 201, 259 ; d) L. Mei, C. Xu, T. Yang, J. M. Ma, L. B. Chen, Q. H. Li, T. H. Wang, J. Mater. Chem. A. 2013, 1, 8658 ; e) G. C. Huang, T. Chen, W. X. Chen, Z. Wang, K. Chang, L. Ma, F. H. Huang, D. Y. Chen, J. Y. Lee, Small 2013, 9, 3693 ; f) D. Y. Chen, G. Ji, B. Ding, Y. Ma, B. H. Qu, W. X. Chen, J. Y. Lee, Nanoscale 2013, 5, [19] H. K. Jeong, Y. P. Lee, R. J. W. E. Lahaye, M. H. Park, K. H. An, I. J. Kim, C. W. Yang, C. Y. Park, R. S. Ruoff, Y. H. Lee, J. Am. Chem. Soc. 2008, 130, [20] a) C. R. Wang, K. B. Tang, Q. Yang, Y. T. Qian, Chem. Phy. Lett. 2002, 357, 371 ; b) H. S. Kim, Y. H. Chung, S. H. Kang, Y. E. Sung, Electrochim. Acta 2009, 54, [21] S. Stankovich, D. A. Dikin, R. D. Piner, K. A. Kohlhaas, A. Kleinhammes, Y. Y. Jia, Y. Wu, S. T. Nguyen, R. S. Ruoff, Carbon 2007, 45, [22] J. Xu, D. H. Lee, R. J. Clément, X. Yu, M. Leskes, A. J. Pell, G. Pintacuda, X. Q. Yang, C. P. Grey, Y. S. Meng, Chem. Mater. 2014, 26, [23] a) S. Komaba, W. Murata, T. Ishikawa, N. Yabuuchi, T. Ozeki, T. Nakayama, A. Ogata, K. Gotoh, K. Fujiwara, Adv. Funct. Mater. 2011, 21, 3859 ; b) A. Abouimrane, W. Weng, H. Eltayeb, Y. Cui, J. Niklas, O. Poluektov, K. Amine, Energy Environ. Sci. 2012, 5, 9632 ; c) S. M. Oh, S. T. Myung, M. W. Jang, B. Scrosati, J. Hassoun, Y. K. Sun ; d) S. Hariharan, K. Saravanan, P. Balaya, Electrochem. Commun. 2013, 31, 5 ; e) D. Y. W. Yu, P. V. Prikhodchenko, C. W. Mason, S. K. Batabyal, J. Gun, S. Sladkevich, A. G. Medvedev, O. Lev, Nat. Comm. 2013, 4, [24] W. S. Hummers, R. E. Offeman, J. Am. Chem. Soc. 1958, 80, [25] C. H. Xu, J. Sun, L. Gao, J. Mater. Chem. 2012, 22,
Supporting Information. Mitigating the P2 O2 phase transition of high-voltage
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2019 Supporting Information Mitigating the P2 O2 phase transition of high-voltage
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2019 Supporting Information Mitigating the P2 O2 phase transition of high-voltage
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information Fe 3 O 4 Quantum Dots Decorated MoS 2 Nanosheet
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information Fe 3 O 4 Quantum Dots Decorated MoS 2 Nanosheet
Electronic Supplementary Information (ESI)
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information (ESI) Nanomat Li-S batteries based on
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information (ESI) Nanomat Li-S batteries based on
Fabrication of Novel Lamellar Alternating Nitrogen-Doped
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Fabrication of Novel Lamellar Alternating Nitrogen-Doped Microporous Carbon
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Fabrication of Novel Lamellar Alternating Nitrogen-Doped Microporous Carbon
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information Metal-organic framework-derived CoSe2/(NiCo)Se2
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information Metal-organic framework-derived CoSe2/(NiCo)Se2
Supporting Information for
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supporting Information for Core-shell ZnO/ZnFe 2 O 4 @C Mesoporous Nanospheres
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supporting Information for Core-shell ZnO/ZnFe 2 O 4 @C Mesoporous Nanospheres
Hierachical Nickel-Carbon Structure Templated by Metal-Organic Frameworks for Efficient Overall Water Splitting
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Hierachical Nickel-Carbon Structure
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Hierachical Nickel-Carbon Structure
Supporting information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry Please do 2018 not adjust margins Supporting information Self-assembled 3D flower-like
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry Please do 2018 not adjust margins Supporting information Self-assembled 3D flower-like
Nickel Hexacyanoferrate/Carbon Composite as a High-Rate and. Long-Life Cathode Material for Aqueous Hybrid Energy
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Nickel Hexacyanoferrate/Carbon Composite as a High-Rate and Long-Life Cathode Material for Aqueous
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Nickel Hexacyanoferrate/Carbon Composite as a High-Rate and Long-Life Cathode Material for Aqueous
Interconnected hierarchical NiCo 2 O 4 microspheres as high performance. electrode material for supercapacitor
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Interconnected hierarchical microspheres as high performance electrode material for
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2017 Interconnected hierarchical microspheres as high performance electrode material for
Supplementary Information. O-vacancy Enriched NiO Hexagonal Platelets Fabricated on Ni. Foam as Self-supported Electrode for Extraordinary
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supplementary Information O-vacancy Enriched NiO Hexagonal Platelets Fabricated
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supplementary Information O-vacancy Enriched NiO Hexagonal Platelets Fabricated
Supporting Information
 Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2016. Supporting Information for Adv. Energy Mater., DOI: 10.1002/aenm.201600322 Integrated Intercalation-Based and Interfacial Sodium
Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2016. Supporting Information for Adv. Energy Mater., DOI: 10.1002/aenm.201600322 Integrated Intercalation-Based and Interfacial Sodium
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information 3D-composite structure of FeP nanorods supported
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information 3D-composite structure of FeP nanorods supported
Tunable CoFe-Based Active Sites on 3D Heteroatom Doped. Graphene Aerogel Electrocatalysts via Annealing Gas Regulation for
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Tunable CoFe-Based Active Sites on 3D Heteroatom Doped Graphene Aerogel
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Tunable CoFe-Based Active Sites on 3D Heteroatom Doped Graphene Aerogel
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2019 Supporting Information Bioinspired Pomegranate-like Microflowers Confining
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2019 Supporting Information Bioinspired Pomegranate-like Microflowers Confining
Electronic Supplementary Information. Hierarchically porous Fe-N-C nanospindles derived from. porphyrinic coordination network for Oxygen Reduction
 Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Hierarchically porous Fe-N-C nanospindles
Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Hierarchically porous Fe-N-C nanospindles
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information High-performance LiTi 2 (PO 4 ) 3
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information High-performance LiTi 2 (PO 4 ) 3
Supplementary Information
 Supplementary Information Fig S1 a top-down SEM image of PS template. b. cross-sectional SEM of PS template (with diameter of 440 nm). Fig. S2 The cross-sectional SEM of the dense Ge film(a) and 3DOM Ge
Supplementary Information Fig S1 a top-down SEM image of PS template. b. cross-sectional SEM of PS template (with diameter of 440 nm). Fig. S2 The cross-sectional SEM of the dense Ge film(a) and 3DOM Ge
Facile synthesis of N-rich carbon quantum dots by spontaneous. polymerization and incision of solvents as efficient bioimaging probes
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supporting Information Facile synthesis of N-rich carbon quantum dots by spontaneous polymerization
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supporting Information Facile synthesis of N-rich carbon quantum dots by spontaneous polymerization
Supporting Information
 Copyright WILEY VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2016. Supporting Information for Adv. Energy Mater., DOI: 10.1002/aenm.201502539 From Hollow Carbon Spheres to N-Doped Hollow Porous
Copyright WILEY VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2016. Supporting Information for Adv. Energy Mater., DOI: 10.1002/aenm.201502539 From Hollow Carbon Spheres to N-Doped Hollow Porous
Supporting Information. In-Situ Facile Bubble-Templated Fabrication of New-Type Urchin-Like (Li, Mo)- Doped Lix(Mo0.3V0.7)2O5 for Zn 2+ Storage
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information In-Situ Facile Bubble-Templated Fabrication of New-Type
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information In-Situ Facile Bubble-Templated Fabrication of New-Type
Supplementary Material
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supplementary Material Biomass Chitosan Derived Cobalt/Nitrogen Doped Carbon
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supplementary Material Biomass Chitosan Derived Cobalt/Nitrogen Doped Carbon
Supporting Information. High cycling stable supercapacitor through electrochemical. deposition of metal-organic frameworks/polypyrrole positive
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2018 Supporting Information High cycling stable supercapacitor through electrochemical deposition
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2018 Supporting Information High cycling stable supercapacitor through electrochemical deposition
Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supplementary Information Scalable and Ascendant Synthesis of Coated Carbon
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supplementary Information Scalable and Ascendant Synthesis of Coated Carbon
Layered polyaniline/graphene film from sandwich-structured polyaniline/graphene/polyaniline nanosheets for high-performance pseudosupercapacitors
 Supporting Information for: Layered polyaniline/graphene film from sandwich-structured polyaniline/graphene/polyaniline nanosheets for high-performance pseudosupercapacitors Zhongqiu Tong a, Yongning Yang
Supporting Information for: Layered polyaniline/graphene film from sandwich-structured polyaniline/graphene/polyaniline nanosheets for high-performance pseudosupercapacitors Zhongqiu Tong a, Yongning Yang
Electronic Supplementary Information (ESI)
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2019 Electronic Supplementary Information (ESI) FeP@C Nanoarray Vertically Grown on Graphene Nanosheets:
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2019 Electronic Supplementary Information (ESI) FeP@C Nanoarray Vertically Grown on Graphene Nanosheets:
A reformative oxidation strategy using high concentration nitric acid for. enhancing emission performance of graphene quantum dots
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 A reformative oxidation strategy using high concentration nitric acid for enhancing emission
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 A reformative oxidation strategy using high concentration nitric acid for enhancing emission
Supplementary Information. High areal capacity lithium sulfur battery cathode by. site-selective vapor infiltration of hierarchical
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2017 Supplementary Information High areal capacity lithium sulfur battery cathode by site-selective
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2017 Supplementary Information High areal capacity lithium sulfur battery cathode by site-selective
state asymmetric supercapacitors
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 3D hierarchical CoO@MnO 2 core-shell nanohybrid for high-energy solid state
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 3D hierarchical CoO@MnO 2 core-shell nanohybrid for high-energy solid state
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information Paragenesis BN/CNTs Hybrid as a Monoclinic Sulfur
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information Paragenesis BN/CNTs Hybrid as a Monoclinic Sulfur
High performance carbon nanotube based fiber-shaped. supercapacitors using redox additives of polypyrrole and. hydroquinone
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supporting information High performance carbon nanotube based fiber-shaped
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supporting information High performance carbon nanotube based fiber-shaped
Supplementary Information
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2018 Supplementary Information Core@Shell Structured Co-CoO@NC Nanoparticles Supported on Nitrogen
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2018 Supplementary Information Core@Shell Structured Co-CoO@NC Nanoparticles Supported on Nitrogen
Supporting Information for
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 Supporting Information for 3D Hierarchical Ni(PO 3 ) 2 Nanosheet Arrays
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 Supporting Information for 3D Hierarchical Ni(PO 3 ) 2 Nanosheet Arrays
Electronic Supplementary Information (ESI)
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information (ESI) Hierarchically porous Mo-doped
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information (ESI) Hierarchically porous Mo-doped
catalytically deposited Cu current collector patterns for high-performance flexible in-plane micro-size energy
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 018 Electronic Supplementary Information (ESI) In-situ growth of Cu(OH) @FeOOH
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 018 Electronic Supplementary Information (ESI) In-situ growth of Cu(OH) @FeOOH
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supporting Information Template-Preparation of Three-Dimensional Molybdenum
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supporting Information Template-Preparation of Three-Dimensional Molybdenum
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information Electronic coupling-tunable of cobalt sulfide/carbon
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information Electronic coupling-tunable of cobalt sulfide/carbon
Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2019 Supplementary Information Mo modulation effect on the hydrogen binding
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2019 Supplementary Information Mo modulation effect on the hydrogen binding
Pingping zhao, Xing Hua, Wei Xu, Wei Luo,* Shengli Chen,* and Gongzhen Cheng
 Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information Metal-Organic Framework-Derived Hybrid
Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information Metal-Organic Framework-Derived Hybrid
Facile fabrication of well-defined polyaniline microtubes derived. from natural kapok fiber for supercapacitor with long-term.
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Facile fabrication of well-defined polyaniline microtubes derived from natural kapok fiber
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Facile fabrication of well-defined polyaniline microtubes derived from natural kapok fiber
Ultrathin Co-Fe Hydroxide Nanosheet Arrays for Improved
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2017 Supplementary Information Ultrathin Co-Fe Hydroxide Nanosheet Arrays for Improved Oxygen Evolution
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2017 Supplementary Information Ultrathin Co-Fe Hydroxide Nanosheet Arrays for Improved Oxygen Evolution
Supporting information
 Electronic Supplementary Material (ESI) for Inorganic Chemistry Frontiers. This journal is the Partner Organisations 2018 Supporting information Cube-Like CuCoO Nanostructures on Reduced Graphene Oxide
Electronic Supplementary Material (ESI) for Inorganic Chemistry Frontiers. This journal is the Partner Organisations 2018 Supporting information Cube-Like CuCoO Nanostructures on Reduced Graphene Oxide
Supplementary Information. Indole-Based Conjugated Macromolecule as Redox- Mediated Electrolyte for Ultrahigh Power Supercapacitor
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2017 Supplementary Information Indole-Based Conjugated Macromolecule as Redox-
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2017 Supplementary Information Indole-Based Conjugated Macromolecule as Redox-
Octahedral Pd Nanocages with Porous Shells Converted by Co(OH) 2 Nanocages with Nanosheet surface as Robust Electrocatalysts for Ethanol Oxidation
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Octahedral Pd Nanocages with Porous Shells Converted by Co(OH) 2 Nanocages
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Octahedral Pd Nanocages with Porous Shells Converted by Co(OH) 2 Nanocages
Supporting Information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2017 Supporting Information Dual active nitrogen doped hierarchical porous hollow carbon nanospheres
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2017 Supporting Information Dual active nitrogen doped hierarchical porous hollow carbon nanospheres
Electronic Supporting Information (ESI)
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Electronic Supporting Information (ESI) Designing a Carbon Nanotubes Interconnected
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Electronic Supporting Information (ESI) Designing a Carbon Nanotubes Interconnected
high-performance intercalation pseudocapacitive anode for lithiumion capacitors
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2019 A novel calendula-like MnNb 2 O 6 anchored on graphene sheet as high-performance
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2019 A novel calendula-like MnNb 2 O 6 anchored on graphene sheet as high-performance
noble-metal-free hetero-structural photocatalyst for efficient H 2
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Ni 12 P 5 nanoparticles embedded into porous g-c 3 N 4 nanosheets as a
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Ni 12 P 5 nanoparticles embedded into porous g-c 3 N 4 nanosheets as a
Key Laboratory of Material Chemistry for Energy Conversion and Storage (Ministry
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2018 Supporting Information for Dramatically enhanced visible-light driven H 2 evolution
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2018 Supporting Information for Dramatically enhanced visible-light driven H 2 evolution
Supporting Information
 Electronic Supplementary Material (ESI) for Materials Horizons. This journal is The Royal Society of Chemistry 2017 Supporting Information Co/CoP Embedded in Hairy Nitrogen-Doped Carbon Polyhedron as an
Electronic Supplementary Material (ESI) for Materials Horizons. This journal is The Royal Society of Chemistry 2017 Supporting Information Co/CoP Embedded in Hairy Nitrogen-Doped Carbon Polyhedron as an
Supporting Information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2017 Supporting Information Phytic acid-derivative transition metal phosphides encapsulated in N,P-codoped
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2017 Supporting Information Phytic acid-derivative transition metal phosphides encapsulated in N,P-codoped
Supporting Information
 Electronic Supplementary Material (ESI) for Sustainable Energy & Fuels. This journal is The Royal Society of Chemistry 2018 Supporting Information Electrospray Synthesis of Si Encapsulated in Graphite/carbon
Electronic Supplementary Material (ESI) for Sustainable Energy & Fuels. This journal is The Royal Society of Chemistry 2018 Supporting Information Electrospray Synthesis of Si Encapsulated in Graphite/carbon
A Ni 3 N-Co 3 N hybrid nanowire array electrode for high-performance nonenzymatic glucose detection
 Electronic Supplementary Material (ESI) for Analytical Methods. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information A Ni 3 N-Co 3 N hybrid nanowire array electrode
Electronic Supplementary Material (ESI) for Analytical Methods. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information A Ni 3 N-Co 3 N hybrid nanowire array electrode
Porous and High-strength Graphitic Carbon/SiC Three-Dimensional Electrode for Capacitive Deionization and Fuel Cell Applications
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information Porous and High-strength Graphitic Carbon/SiC Three-Dimensional
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information Porous and High-strength Graphitic Carbon/SiC Three-Dimensional
MXene/Graphene Hybrid Fibers for High. Performance Flexible Supercapacitors
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information for MXene/Graphene Hybrid Fibers for High Performance
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information for MXene/Graphene Hybrid Fibers for High Performance
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 018 Electronic Supplementary Information Superhydrophilic amorphous Co-B-P nanosheet
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 018 Electronic Supplementary Information Superhydrophilic amorphous Co-B-P nanosheet
Supporting Information. Metal organic framework-derived Fe/C Nanocubes toward efficient microwave absorption
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supporting Information Metal organic framework-derived Fe/C Nanocubes toward
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supporting Information Metal organic framework-derived Fe/C Nanocubes toward
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information Experimental Section Materials: Carbon
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information Experimental Section Materials: Carbon
3D CNTs/Graphene-S-Al 3 Ni 2 Cathodes for High-Sulfur-Loading and Long-Life Lithium Sulfur Batteries
 FULL PAPER Lithium-Sulfur Batteries 3D CNTs/Graphene-S-Al 3 Ni 2 Cathodes for High-Sulfur-Loading and Long-Life Lithium Sulfur Batteries Zeqing Guo, Huagui Nie,* Zhi Yang,* Wuxing Hua, Chunping Ruan, Dan
FULL PAPER Lithium-Sulfur Batteries 3D CNTs/Graphene-S-Al 3 Ni 2 Cathodes for High-Sulfur-Loading and Long-Life Lithium Sulfur Batteries Zeqing Guo, Huagui Nie,* Zhi Yang,* Wuxing Hua, Chunping Ruan, Dan
Journal of Materials Chemistry A. Supporting Information. Cobalt Nickel Boride as an Active Electrocatalyst for Water Splitting
 Electronic Supplementary Material (ESI) for. This journal is The Royal Society of Chemistry Please do 2017 not adjust margins Supporting Information Cobalt Nickel Boride as an Active Electrocatalyst for
Electronic Supplementary Material (ESI) for. This journal is The Royal Society of Chemistry Please do 2017 not adjust margins Supporting Information Cobalt Nickel Boride as an Active Electrocatalyst for
Supplementary Information. A synergistic interaction between isolated Au nanoparticles with oxygen vacancies in
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supplementary Information A synergistic interaction between isolated Au
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supplementary Information A synergistic interaction between isolated Au
Supporting Information. Outstanding hydrogen evolution reaction catalyzed by porous nickel diselenide
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2017 Supporting Information Outstanding hydrogen evolution reaction catalyzed
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2017 Supporting Information Outstanding hydrogen evolution reaction catalyzed
3D Yolk-Shelled NiGa 2 S 4 Microspheres Confined with Nanosheets for High Performance Supercapacitors
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information (ESI) 3D Yolk-Shelled NiGa 2 S 4 Microspheres
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information (ESI) 3D Yolk-Shelled NiGa 2 S 4 Microspheres
Electronic Supplementary Information (ESI) for Analyst. A Facile Graphene Oxide-Based Fluorescent Nanosensor for in Situ
 Electronic Supplementary Material (ESI) for Analyst. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information (ESI) for Analyst A Facile Graphene Oxide-Based Fluorescent
Electronic Supplementary Material (ESI) for Analyst. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information (ESI) for Analyst A Facile Graphene Oxide-Based Fluorescent
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Flexible 3D Porous CuO Nanowire Arrays for Enzymeless Glucose
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Flexible 3D Porous CuO Nanowire Arrays for Enzymeless Glucose
Supporting Information
 Supporting Information A General Strategy to Fabricate P as Highly Efficient Cocatalyst via Photo-Reduction Deposition for Hydrogen Evolution Yuming Dong a, *, Linggang Kong a, Pingping Jiang a, Guangli
Supporting Information A General Strategy to Fabricate P as Highly Efficient Cocatalyst via Photo-Reduction Deposition for Hydrogen Evolution Yuming Dong a, *, Linggang Kong a, Pingping Jiang a, Guangli
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 Supporting Information Branched polyethylenimine grafted electrospun polyacrylonitrile
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 Supporting Information Branched polyethylenimine grafted electrospun polyacrylonitrile
Zhaohuai Li, Qiu He, Xu Xu,* Yan Zhao, Xiaowei Liu, Cheng Zhou, Dong Ai, Lixue Xia, and Liqiang Mai*
 Communication Lithium Sulfur Batteries A 3D Nitrogen-Doped Graphene/TiN Nanowires Composite as a Strong Polysulfide Anchor for Lithium Sulfur Batteries with Enhanced Rate Performance and High Areal Capacity
Communication Lithium Sulfur Batteries A 3D Nitrogen-Doped Graphene/TiN Nanowires Composite as a Strong Polysulfide Anchor for Lithium Sulfur Batteries with Enhanced Rate Performance and High Areal Capacity
Silver Nanowires Coated on Cotton for Flexible Pressure Sensors. College of Materials Science and Engineering, Key Lab of Guangdong Province for
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry C. This journal is The Royal Society of Chemistry 2015 Supporting Information Silver Nanowires Coated on Cotton for Flexible Pressure
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry C. This journal is The Royal Society of Chemistry 2015 Supporting Information Silver Nanowires Coated on Cotton for Flexible Pressure
Recent advances in energy transfer in bulk and nanoscale. luminescent materials: From spectroscopy to applications
 Electronic Supplementary Material (ESI) for Chemical Society Reviews. This journal is The Royal Society of Chemistry 215 Electronic supplementary information Recent advances in energy transfer in ulk and
Electronic Supplementary Material (ESI) for Chemical Society Reviews. This journal is The Royal Society of Chemistry 215 Electronic supplementary information Recent advances in energy transfer in ulk and
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry B. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Experimental 2.1 Chemicals and Materials
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry B. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Experimental 2.1 Chemicals and Materials
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information From ZIF-8 Polyhedron to Three-Dimensional Nitrogen
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information From ZIF-8 Polyhedron to Three-Dimensional Nitrogen
High performing AgNWs transparent conducting electrodes with 2.5Ω/Sq based upon Roll-to- Roll compatible post processing technique
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2019 High performing AgNWs transparent conducting electrodes with 2.5Ω/Sq based upon Roll-to- Roll
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2019 High performing AgNWs transparent conducting electrodes with 2.5Ω/Sq based upon Roll-to- Roll
Highly enhanced performance of spongy graphene as oil sorbent
 Supporting Information Highly enhanced performance of spongy graphene as oil sorbent Hengchang Bi, a Xiao Xie, a Kuibo Yin, a Yilong Zhou, a Shu Wan, a Rodney S. Ruoff b and Litao Sun* a a SEU-FEI Nano-Pico
Supporting Information Highly enhanced performance of spongy graphene as oil sorbent Hengchang Bi, a Xiao Xie, a Kuibo Yin, a Yilong Zhou, a Shu Wan, a Rodney S. Ruoff b and Litao Sun* a a SEU-FEI Nano-Pico
Nitrogen-Doped Core-Sheath Carbon Nanotube Array for Highly Stretchable Supercapacitor
 www.advancedsciencenews.com Nitrogen-Doped Core-Sheath Carbon Nanotube Array for Highly Stretchable Supercapacitor Zhitao Zhang, Lie Wang, Yiming Li, Yuhang Wang, Jing Zhang, Guozhen Guan, Zhiyong Pan,
www.advancedsciencenews.com Nitrogen-Doped Core-Sheath Carbon Nanotube Array for Highly Stretchable Supercapacitor Zhitao Zhang, Lie Wang, Yiming Li, Yuhang Wang, Jing Zhang, Guozhen Guan, Zhiyong Pan,
Electronic Supplementary Information for. Highly Stable Mesoporous Silica Nanospheres Embedded with
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 17 Electronic Supplementary Information for Highly Stable Mesoporous Silica Nanospheres Embedded
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 17 Electronic Supplementary Information for Highly Stable Mesoporous Silica Nanospheres Embedded
Sodium Borohydride Stabilizes Very Active Gold Nanoparticle Catalyst ELECTRONIC SUPPLEMENTARY INFORMATIONS. I. General data..p2
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Sodium Borohydride Stabilizes Very Active Gold Nanoparticle Catalyst Christophe Deraedt, Lionel
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Sodium Borohydride Stabilizes Very Active Gold Nanoparticle Catalyst Christophe Deraedt, Lionel
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information Continuous fabrication of graphene-confined polypyrrole
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information Continuous fabrication of graphene-confined polypyrrole
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information Cobalt nanoparticles encapsulated in carbon nanotube-grafted
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information Cobalt nanoparticles encapsulated in carbon nanotube-grafted
Curriculum Vitae. Yu (Will) Wang
 Curriculum Vitae Yu (Will) Wang Assistant Research Professor yu.wang3@wsu.edu School of Mechanical and Materials Engineering Washington State University, Pullman, 99164, WA, USA EDUCATION B.S. Sichuan
Curriculum Vitae Yu (Will) Wang Assistant Research Professor yu.wang3@wsu.edu School of Mechanical and Materials Engineering Washington State University, Pullman, 99164, WA, USA EDUCATION B.S. Sichuan
Flexible and Printable Paper Based Strain Sensors for Wearable and Large Area Green Electronics
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2016 Supporting Information Flexible and Printable Paper Based Strain Sensors for Wearable and Large
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2016 Supporting Information Flexible and Printable Paper Based Strain Sensors for Wearable and Large
small
 Communication Lithium Sulfur Batteries Functional Differentiation of Three Pores for Effective Sulfur Confinement in Li S Battery Qian Wang, Minghui Yang,* Zhen-Bo Wang,* Chao Li, and Da-Ming Gu Shuttle
Communication Lithium Sulfur Batteries Functional Differentiation of Three Pores for Effective Sulfur Confinement in Li S Battery Qian Wang, Minghui Yang,* Zhen-Bo Wang,* Chao Li, and Da-Ming Gu Shuttle
Flexible Nb 4 N 5 /rgo Electrode for High-Performance Solid State Supercapacitors. Huazhong University of Science and Technology Wuhan , China
 Copyright 2018 American Scientific Publishers All rights reserved Printed in the United States of America Article Journal of Nanoscience and Nanotechnology Vol. 18, 30 38, 2018 www.aspbs.com/jnn Flexible
Copyright 2018 American Scientific Publishers All rights reserved Printed in the United States of America Article Journal of Nanoscience and Nanotechnology Vol. 18, 30 38, 2018 www.aspbs.com/jnn Flexible
Supplementary Information. Flexible crystalline silicon radial junction photovoltaics with vertically aligned tapered microwires
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2018 Supplementary Information Flexible crystalline silicon radial junction photovoltaics
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2018 Supplementary Information Flexible crystalline silicon radial junction photovoltaics
The study of 3-strand PP/PE composite monofilament Rope
 International Conference on Manufacturing Science and Engineering (ICMSE 2015) The study of 3-strand PP/PE composite monofilament Rope Jian-gao Shi1 *, Wenwen Yu1, Yongli Liu1, Lei Wang1, Xiaoxue Chen1,
International Conference on Manufacturing Science and Engineering (ICMSE 2015) The study of 3-strand PP/PE composite monofilament Rope Jian-gao Shi1 *, Wenwen Yu1, Yongli Liu1, Lei Wang1, Xiaoxue Chen1,
Supporting Information. Highly simple and rapid synthesis of ultrathin gold nanowires with
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information Highly simple and rapid synthesis of ultrathin gold
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information Highly simple and rapid synthesis of ultrathin gold
Roles of Nitrogen Functionalities in Enhancing the. Excitation-Independent Green-Color Photoluminescence of
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Roles of Nitrogen Functionalities in Enhancing the Excitation-Independent
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Roles of Nitrogen Functionalities in Enhancing the Excitation-Independent
A ligand conformation preorganization approach to construct a. copper-hexacarboxylate framework with a novel topology for
 Electronic Supplementary Material (ESI) for Inorganic Chemistry Frontiers. This journal is the Partner rganisations 2018 A ligand conformation preorganization approach to construct a copper-hexacarboxylate
Electronic Supplementary Material (ESI) for Inorganic Chemistry Frontiers. This journal is the Partner rganisations 2018 A ligand conformation preorganization approach to construct a copper-hexacarboxylate
Electronic Supplementary Information for
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information for Al-doping to Synchronously Improve
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information for Al-doping to Synchronously Improve
LC-MS-Guided Isolation of Insulin Secretion-promoting. Monoterpenoid Carbazole Alkaloids from Murraya
 Supporting Information LC-MS-Guided Isolation of Insulin Secretion-promoting Monoterpenoid Carbazole Alkaloids from Murraya microphylla Xiao-Li Ma, Jun Li, Jiao Zheng, Xiao-Pan Gu, Daneel Ferreira, Jordan
Supporting Information LC-MS-Guided Isolation of Insulin Secretion-promoting Monoterpenoid Carbazole Alkaloids from Murraya microphylla Xiao-Li Ma, Jun Li, Jiao Zheng, Xiao-Pan Gu, Daneel Ferreira, Jordan
A Flexible, Lightweight, and Wearable Triboelectric Nanogenerator for Energy Harvesting and Self- Powered Sensing
 FULL PAPER Triboelectric Nanogenerators A Flexible, Lightweight, and Wearable Triboelectric Nanogenerator for Energy Harvesting and Self- Powered Sensing Fan Wu, Congju Li,* Yingying Yin, Ran Cao, Hui
FULL PAPER Triboelectric Nanogenerators A Flexible, Lightweight, and Wearable Triboelectric Nanogenerator for Energy Harvesting and Self- Powered Sensing Fan Wu, Congju Li,* Yingying Yin, Ran Cao, Hui
An Ultrathin Flexible 2D Membrane Based on Single-Walled Nanotube MoS 2 Hybrid Film for High-Performance Solar Steam Generation
 FULL PAPER Solar Steam Generation An Ultrathin Flexible 2D Membrane Based on Single-Walled Nanotube MoS 2 Hybrid Film for High-Performance Solar Steam Generation Xiangdong Yang, Yanbing Yang, Linna Fu,
FULL PAPER Solar Steam Generation An Ultrathin Flexible 2D Membrane Based on Single-Walled Nanotube MoS 2 Hybrid Film for High-Performance Solar Steam Generation Xiangdong Yang, Yanbing Yang, Linna Fu,
Complex Systems and Applications
 Complex Systems and Applications Volume 1 TABLE OF CONTENTS State Feedback and Pole Assignment for A Class of the Second Order Coupled Generalized Distributed Parameter Systems 1 Z. Q. Ge, G. T. Zhu On
Complex Systems and Applications Volume 1 TABLE OF CONTENTS State Feedback and Pole Assignment for A Class of the Second Order Coupled Generalized Distributed Parameter Systems 1 Z. Q. Ge, G. T. Zhu On
Supporting Information. High-performance nonfullerene polymer solar cells based on fluorinated wide
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information High-performance nonfullerene polymer solar cells
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information High-performance nonfullerene polymer solar cells
Efficient ternary non-fullerene polymer solar cells with PCE of 11.92% and FF of 76.5%
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 218 Electronic Supplementary Information Efficient ternary non-fullerene polymer
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 218 Electronic Supplementary Information Efficient ternary non-fullerene polymer
Supporting Information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2018 Supporting Information Efficient and reliable surface charge transfer doping of black phosphorus
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2018 Supporting Information Efficient and reliable surface charge transfer doping of black phosphorus
Dehydrogenative Transformations of Imines Using a Heterogeneous Photocatalyst. Supporting Information
 S1 Dehydrogenative Transformations of Imines Using a Heterogeneous Photocatalyst Colby M. Adolph, Jacob Werth, Ramajeyam Selvaraj, Evan C. Wegener, and Christopher Uyeda* Department of Chemistry, Purdue
S1 Dehydrogenative Transformations of Imines Using a Heterogeneous Photocatalyst Colby M. Adolph, Jacob Werth, Ramajeyam Selvaraj, Evan C. Wegener, and Christopher Uyeda* Department of Chemistry, Purdue
Jun Yang, Hongcheng Gao, Shuang Men, Zhenqing Shi, Zhang Lin, Xiongwu Kang,* and Shaowei Chen* FULL PAPER. 1. Introduction
 FULL PAPER Batteries CoSe 2 Nanoparticles Encapsulated by N-Doped Carbon Framework Intertwined with Carbon Nanotubes: High- Performance Dual-Role Anode Materials for Both Li- and Na-Ion Batteries Jun Yang,
FULL PAPER Batteries CoSe 2 Nanoparticles Encapsulated by N-Doped Carbon Framework Intertwined with Carbon Nanotubes: High- Performance Dual-Role Anode Materials for Both Li- and Na-Ion Batteries Jun Yang,
Transformation Optics and Experiments
 Forum for Electromagnetic Research Methods and Application Technologies (FERMAT) Transformation Optics and Experiments Tie Jun Cui and Wei Xiang Jiang State Key Laboratory of Millimeter Waves Southeast
Forum for Electromagnetic Research Methods and Application Technologies (FERMAT) Transformation Optics and Experiments Tie Jun Cui and Wei Xiang Jiang State Key Laboratory of Millimeter Waves Southeast
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Wide-bandgap polymer based on alkylphenyl-substituted
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Wide-bandgap polymer based on alkylphenyl-substituted
