Analysis of Deuterium Enrichment by Fourier Transform Infrared Spectrometry (FTIR): Practice. Christine Slater PhD
|
|
|
- Margaret Dixon
- 5 years ago
- Views:
Transcription
1 Analysis of Deuterium Enrichment by Fourier Transform Infrared Spectrometry (FTIR): Practice Christine Slater PhD Nutrition Specialist International Atomic Energy Agency
2 Preparation of calibration standard Make a large volume of the calibrating standard A solution of ~1000 mg/kg (ppm) or 1 g/l should be prepared by weighing 99.8 atom % deuterium oxide (D 2 O) and diluting in normal drinking water from the region Do not used distilled water to make the calibration standard. Distilled water is subject to fractionation De-ionised (ultra-filtered) water can be used Note that the density of deuterium oxide is g/ml All glassware must be clean and dry before use
3 Preparation of calibration standard Use a volumetric flask (1 L or 250 ml) Transfer to a borosilicate bottle with a PTFE-lined screw cap for storage until required Also retain 1 L of the water used to make the dilution (0 standard)
4 Storage of the calibration standard It is a good idea to store the calibration standards in several smaller, tightly sealed bottles (e.g. 250 ml borosilicate bottles with PTFE-lined screw caps) Only one enriched and one natural abundance bottle should be in use at any time, as working standards The remainder should be sealed until required
5 Storage of calibration standard The calibration standards will last for several years if stored in a cool, dark place out of direct sunlight Wrapping bottles in aluminium foil helps to protect the contents from light The bottles must be well-sealed to prevent ingress of water from the atmosphere Do not store the calibration standard in the same place as the deuterium oxide
6 Preparation of the calibration standard The D 2 O should be weighed on an analytical balance accurate to g or preferably g The standard can be prepared in two stages, but it is important that the weight of deuterium oxide is known to g Balances must be levelled and calibrated before use
7 Stage 1 using analytical balance Weigh a clean, dry 50 ml volumetric flask with its stopper in place on an analytical balance to g Alternatively use a clean, dry glass bottle with a cap Add a small volume (20-30 ml) of drinking water to the flask, replace the cap and weigh again Add 1 g of D 2 O to the bottle 1 g D 2 O is approximately ~0.9 ml as the density of deuterium oxide is higher than water (1.105 g/ml and 1.0 g/ml respectively) Replace the stopper or cap to avoid losses by evaporation, and note the weight Calculate the weight of D 2 O in the bottle
8 Stage 2 Weigh a clean dry 1 L volumetric flask with its stopper. At this stage a balance weighing to 0.1 g can be used Quantitatively transfer the water from the 50 ml container into the 1 L volumetric flask using a funnel Add local drinking water (or de-ionised water) to the smaller container and pour it into the larger container Repeat this at least 3 times to ensure that all the D 2 O is transferred. Be careful not to spill any Add local drinking water to the 1 L volumetric flask up to the mark Replace the stopper and weigh again Calculate the enrichment of D 2 O in mg/kg
9 The FTIR cell Calcium fluoride cells with a cell thickness (path length) of 10-4 m (100 m) are recommended for analysis of deuterium in saliva samples These cells cannot be used for the analysis of urine samples because they are damaged by the ammonium and phosphate content of urine Sodium chloride cells, which are often supplied with the FTIR, are not suitable for analysis of samples containing water
10 The FTIR cell: demountable cell assembly
11 The FTIR cell When not in use, store the cells in their original packaging Wipe only with a lintfree cloth (lens tissue)
12 Filling cells Fill 1 ml syringe with standard water or saliva Firmly press folded absorbent paper over the exit port to absorb excess sample and prevent ingress of air Fill the cell by gently pushing the syringe plunger or using firm taps on the plunger with the index finger Remove excess/splashes from the outside of the cell window using absorbent paper
13 Check for bubbles by holding the cell up to a light
14 Avoid cross-contamination If there are visible bubbles in the cell, add more sample until all of the bubbles have been pushed out Measure the absorbance from cm -1 Remove the sample using the same syringe that was used for filling. Discard the syringe Use a new syringe for each sample to avoid crosscontamination When all the samples have been analysed, rinse the cell with drinking quality water before storing
15 Operation of the FTIR Switch on the FTIR minutes before use to allow the electronics to stabilise If there is a power cut, wait for 30 min after power has been resumed before analysing samples Check that both the interface and the mirror are working properly Ensure that the following are set: Measurement mode: Absorbance Apodization: SqrTriangular No of scans: 32 Resolution: 2.0 Range (cm -1 ): Minimum Maximum
16 Operation of the FTIR Perform a Background scan using the zero standard - the water used to make the calibration standard Calibrate the instrument using the 1000 mg/kg (ppm) standard The background for body water samples is the baseline (time 0) saliva sample
17 Post-installation checks: accuracy and precision Calibration curve Within-day reproducibility Between days reproducibility
18 Standard curve The accuracy of deuterium analysis over the range of enrichments likely to be encountered should be checked using gravimetrically prepared standards Smaller volumes (e.g. 100 ml) of these standards can be prepared by diluting D 2 O with local drinking water in a volumetric flask and weighing on an analytical balance The enrichment should range from 0 (natural abundance drinking water) to 2000 mg/kg; an enrichment above that likely to be encountered in saliva samples Make independently weighed standards, NOT a serial dilution of the calibration standard
19 Preparation of gravimetric standards Target enrichment (mg/kg) μl D 2 O Standards should be made (in 100 ml local drinking water or de-ionised water) according to the table The deuterium oxide can be pipetted into the volumetric flask but it must be accurately weighed
20 Preparation of gravimetric standards Tare the balance with the volumetric flask and its stopper Half-fill the flask with water. Note the weight (A) Add the deuterium oxide. Note the weight (B) Fill up to the mark with water. Note the weight (C) Calculate the weight of D 2 O (D=(B-A)*1000 mg) Calculate the total weight of water (W=(C-D)/1000 kg) The enrichment in (mg/kg) is D/W
21 Measured (mg/kg) Deuterium calibration curve measured by FTIR y = x R 2 = Calculated (m g/kg) Analyse the standards in triplicate (3 separate fills)
22 Within-day precision The cell was loaded once with each standard and scanned 10 times against the same calibration standard (1050 mg/kg) Use standards Make repeated scans of the calibration standards 5-10 times Fill the cell with the same sample 5-10 times Data from Mauro Valencia Each repetition was independently loaded into the cell n times and read using the same calibration standard (1050 mg/kg) Precision can be quoted as the standard deviation (SD) or coefficient of variation (CV) CV (%) = (SD/mean) x 100 Data from Mauro Valencia
23 Between days precision If the precision analysis is repeated on several days over a period of time, an estimate of between days precision is achieved Between days precision is sometimes slightly worse than within-day precision
24 Acknowledgements 1 This presentation and the accompanying presentation on FTIR Theory were originally prepared by Christine Slater for use in the Regional Training Course on deuterium dilution methods for assessment of body composition, and human milk intake by breastfed infants (Botswana, 2008). The is grateful to the late Dr Lesley Bluck from MRC-HNR, Cambridge, UK who originally developed and validated the analysis of deuterium enrichment by FTIR for generously sharing his expertise. Jennings, Bluck, Wright & Elia, Clin. Chem 45:7 (1999) Some of the information presented here does not apply to the new portable FTIRs.
25 Acknowledgements 2 The is grateful to staff from the chemistry department at the Botswana National Food Technology Research Centre (Juda Bogopa and Kabo Mosetlha), who are featured in the photos in this presentation, and made and analysed the standards shown in the standard curve. Data on within-day precision of analysis are from Mauro Valencia Juillerat, Mexico, who was also the photographer and fellow lecturer on the Regional Training Course.
26 Advantages of the new portable FTIRs Portable: supplied as standard with a toughened transport case Battery driven and free from interruptions in electricity supply No optical mirrors making them much less prone to damage while being moved No optical housing making measurements less prone to interference from carbon dioxide Software control runs under a contemporary 64 bit operating system
CHM250 Calibration and Measurement Lab. Balance Calibration
 CHM250 Calibration and Measurement Lab Green Profile Balance Calibration Introduction: Balances that are properly operated, calibrated and maintained are crucial for laboratory operations. The accuracy
CHM250 Calibration and Measurement Lab Green Profile Balance Calibration Introduction: Balances that are properly operated, calibrated and maintained are crucial for laboratory operations. The accuracy
Instruction Sheet. Calibration of Multiple 1720 and FT660 Turbidimeters
 Instruction Sheet 26596-89 Calibration of Multiple 1720 and FT660 Turbidimeters Overview This instruction sheet describes the calibration of up to eight 1720C, 1720D, or 1720E turbidimeters, or up to three
Instruction Sheet 26596-89 Calibration of Multiple 1720 and FT660 Turbidimeters Overview This instruction sheet describes the calibration of up to eight 1720C, 1720D, or 1720E turbidimeters, or up to three
CH2250: Techniques in Laboratory Chemistry. Outline Measuring Mass Measuring Volume Significant figures. Mass Measurement
 CH2250: Techniques in Laboratory Chemistry Outline Measuring Mass Measuring Volume Significant figures Mass Measurement Mass Measurement Measure mass not weight Mass is measured with a balance (a scale
CH2250: Techniques in Laboratory Chemistry Outline Measuring Mass Measuring Volume Significant figures Mass Measurement Mass Measurement Measure mass not weight Mass is measured with a balance (a scale
Transferpette -8/-12
 Transferpette -8/-12 Testing Instructions (SOP) May 2009 1. Introduction The standard DIN EN ISO 8655 describes both the design and the testing of the piston operated pipette Transferpette 8/ 12. The following
Transferpette -8/-12 Testing Instructions (SOP) May 2009 1. Introduction The standard DIN EN ISO 8655 describes both the design and the testing of the piston operated pipette Transferpette 8/ 12. The following
VOLUMETRIC TECHNIQUES
 REVISED 10/14 CHEMISTRY 1101L VOLUMETRIC TECHNIQUES Volume measurements are important in many experimental procedures. Sometimes volume measurements must be exact; other times they can be approximate.
REVISED 10/14 CHEMISTRY 1101L VOLUMETRIC TECHNIQUES Volume measurements are important in many experimental procedures. Sometimes volume measurements must be exact; other times they can be approximate.
Lab Equipment ANALYTICAL BALANCE
 Lab Equipment ANALYTICAL BALANCE Analytical balances are used for very accurate, quantitative measurements of mass to the nearest 0.001 g. (Some read to 0.0001 g.) These are delicate instruments, subject
Lab Equipment ANALYTICAL BALANCE Analytical balances are used for very accurate, quantitative measurements of mass to the nearest 0.001 g. (Some read to 0.0001 g.) These are delicate instruments, subject
Calibration of Volumetric Glassware
 Calibration of Volumetric Glassware Introduction This set of laboratory experiments is designed to introduce you to some of the apparatus and operations you will be using during the remainder of this course,
Calibration of Volumetric Glassware Introduction This set of laboratory experiments is designed to introduce you to some of the apparatus and operations you will be using during the remainder of this course,
Predicted Dispense Volume vs. Gravimetric Measurement for the MICROLAB 600. November 2010
 Predicted Dispense Volume vs. Gravimetric Measurement for the MICROLAB 600 November 2010 Table of Contents ``Abstract...3 ``Introduction...4 ``Methods & Results...6 ``Data Analysis...9 ``Conclusion...12
Predicted Dispense Volume vs. Gravimetric Measurement for the MICROLAB 600 November 2010 Table of Contents ``Abstract...3 ``Introduction...4 ``Methods & Results...6 ``Data Analysis...9 ``Conclusion...12
Transferpette. Testing Instructions (SOP) 1. Introduction. May 2009
 Transferpette Testing Instructions (SOP) May 2009 1. Introduction The standards DIN EN ISO 8655 describe both the design and the testing of the piston operated pipette Transferpette. The following Testing
Transferpette Testing Instructions (SOP) May 2009 1. Introduction The standards DIN EN ISO 8655 describe both the design and the testing of the piston operated pipette Transferpette. The following Testing
CHM 317H1S Winter Section P Procedures and Tables
 CHM 317H1S Winter 2018 Section P Procedures and Tables Procedures Page 1 Standard Operating Procedures Throughout the laboratory portion of this course, you will be required to perform a number of operations
CHM 317H1S Winter 2018 Section P Procedures and Tables Procedures Page 1 Standard Operating Procedures Throughout the laboratory portion of this course, you will be required to perform a number of operations
Lab 1. Instrumentation Familiarity: Using Micropipetters and Serological Pipettes
 Instrumentation Familiarity: Using Micropipetters and Serological Pipettes Introduction: In molecular biology we sometimes need to measure volumes as small as one millionth of a liter (a liter is about
Instrumentation Familiarity: Using Micropipetters and Serological Pipettes Introduction: In molecular biology we sometimes need to measure volumes as small as one millionth of a liter (a liter is about
Hands-On Experiment Density and Measurement
 Hands-On Experiment Density and Measurement GOALS: 1. To measure liquid volume as accurately as possible with graduated cylinders. 2. To measure the volume of irregular shaped solid objects by liquid volume
Hands-On Experiment Density and Measurement GOALS: 1. To measure liquid volume as accurately as possible with graduated cylinders. 2. To measure the volume of irregular shaped solid objects by liquid volume
VITLAB Volumetric instruments
 VITLAB Volumetric instruments Standard Operating Procedure (SOP) VITLAB GmbH Linus-Pauling-Str.1 63762 Grossostheim Germany Telefon: +49 6026 97799-0 Fax: +49 6026 97799-30 E-mail: info@vitlab.com Internet:
VITLAB Volumetric instruments Standard Operating Procedure (SOP) VITLAB GmbH Linus-Pauling-Str.1 63762 Grossostheim Germany Telefon: +49 6026 97799-0 Fax: +49 6026 97799-30 E-mail: info@vitlab.com Internet:
LABORATORY TECHNIQUES. Pouring Liquids
 LABORATORY TECHNIQUES Working in the chemistry laboratory you will be handling potentially dangerous substances and performing unfamiliar tasks. This section provides you with a guide to the safe laboratory
LABORATORY TECHNIQUES Working in the chemistry laboratory you will be handling potentially dangerous substances and performing unfamiliar tasks. This section provides you with a guide to the safe laboratory
Laboratory #2 Pipetting Technique and Micropipette Calibration Skills=40 pts
 Laboratory #2 Pipetting Technique and Micropipette Calibration Skills=40 pts Objectives: Upon completion of this unit, the student should be able to: 1. List and describe 3 categories of pipets. 2. List
Laboratory #2 Pipetting Technique and Micropipette Calibration Skills=40 pts Objectives: Upon completion of this unit, the student should be able to: 1. List and describe 3 categories of pipets. 2. List
Laboratory Activity Measurement and Density. Average deviation = Sum of absolute values of all deviations Number of trials
 Laboratory Activity Measurement and Density Background: Measurements of mass and volume are very common in the chemistry laboratory. The analytical balance is used to measure mass, and the graduated cylinder,
Laboratory Activity Measurement and Density Background: Measurements of mass and volume are very common in the chemistry laboratory. The analytical balance is used to measure mass, and the graduated cylinder,
CHM Introductory Laboratory Experiment (r17sd) 1/13
 CHM 110 - Introductory Laboratory Experiment (r17sd) 1/13 Purpose This introductory exercise will familiarize you with a few of the measurements we make in the chemistry laboratory and the level of uncertainty
CHM 110 - Introductory Laboratory Experiment (r17sd) 1/13 Purpose This introductory exercise will familiarize you with a few of the measurements we make in the chemistry laboratory and the level of uncertainty
Bante820 Portable Dissolved Oxygen Meter Instruction Manual
 Bante820 Portable Dissolved Oxygen Meter Instruction Manual BANTE INSTRUMENTS CO., LTD Bante820 Portable Dissolved Oxygen Meter 1 Introduction Thank you for selecting the Bante820 portable dissolved oxygen
Bante820 Portable Dissolved Oxygen Meter Instruction Manual BANTE INSTRUMENTS CO., LTD Bante820 Portable Dissolved Oxygen Meter 1 Introduction Thank you for selecting the Bante820 portable dissolved oxygen
CHM 100 / Introductory Laboratory Experiment (r10) 1/11
 CHM 100 / 110 - Introductory Laboratory Experiment (r10) 1/11 Purpose This introductory exercise will familiarize you with a few of the measurements we make in the chemistry laboratory and the level of
CHM 100 / 110 - Introductory Laboratory Experiment (r10) 1/11 Purpose This introductory exercise will familiarize you with a few of the measurements we make in the chemistry laboratory and the level of
BASIC LABORATORY TECHNIQUES (Revised )
 BASIC LABORATORY TECHNIQUES (Revised 1-2-16) (See Appendix II: Summary for making Spreadsheets and Graphs with Excel and Appendix III parts C, C1 and C2: Significant figures, scientific notation and rounding)
BASIC LABORATORY TECHNIQUES (Revised 1-2-16) (See Appendix II: Summary for making Spreadsheets and Graphs with Excel and Appendix III parts C, C1 and C2: Significant figures, scientific notation and rounding)
BASIC LABORATORY TECHNIQUES (Revised )
 BASIC LABORATORY TECHNIQUES (Revised 1-6-13) A. WEIGHING The determination of the quantity of matter in a sample is most directly determined by measuring its mass. The process by which we determine the
BASIC LABORATORY TECHNIQUES (Revised 1-6-13) A. WEIGHING The determination of the quantity of matter in a sample is most directly determined by measuring its mass. The process by which we determine the
QAM-I-117 Volumetric Equipment Calibration Verification
 1. Applicability and Purpose This procedure applies to all adjustable and fixed volume pipetters and any labware used to deliver measured volumes of liquid by laboratory analysts at the Texas Institute
1. Applicability and Purpose This procedure applies to all adjustable and fixed volume pipetters and any labware used to deliver measured volumes of liquid by laboratory analysts at the Texas Institute
Technical Procedure for General Laboratory Equipment
 Technical Procedure for General Laboratory Equipment 1.0 Purpose - This procedure specifies the required elements for the use of general laboratory equipment. 2.0 Scope - This procedure applies to all
Technical Procedure for General Laboratory Equipment 1.0 Purpose - This procedure specifies the required elements for the use of general laboratory equipment. 2.0 Scope - This procedure applies to all
PREPARING GAS SAMPLES IN LARGE PLASTIC SYRINGES
 PREPARING GAS SAMPLES IN LARGE PLASTIC SYRINGES A wide variety of gases can be prepared safely inside a 60 ml plastic syringe. Here you will practice making carbon dioxide, so that you know the technique
PREPARING GAS SAMPLES IN LARGE PLASTIC SYRINGES A wide variety of gases can be prepared safely inside a 60 ml plastic syringe. Here you will practice making carbon dioxide, so that you know the technique
VITLAB micropipette. Standard Operating Procedure (SOP)
 VITLAB micropipette Standard Operating Procedure (SOP) VITLAB GmbH Linus-Pauling-Str.1 63762 Grossostheim Germany tel: +49 6026 97799-0 fax: +49 6026 97799-30 info@vitlab.com www.vitlab.com Table of contents
VITLAB micropipette Standard Operating Procedure (SOP) VITLAB GmbH Linus-Pauling-Str.1 63762 Grossostheim Germany tel: +49 6026 97799-0 fax: +49 6026 97799-30 info@vitlab.com www.vitlab.com Table of contents
Measuring Mass and Volume
 Measuring Mass and Volume Experiment 2 Expt 2 Measurement.wpd INTENT The purpose of this experiment is to introduce some fundamental aspects of the measurement making process as well as to introduce some
Measuring Mass and Volume Experiment 2 Expt 2 Measurement.wpd INTENT The purpose of this experiment is to introduce some fundamental aspects of the measurement making process as well as to introduce some
Chapter Pipette service & maintenance. Pipette specifications according to ISO Repair in the lab or return for service?
 Pipette specifications according to ISO 8655 Chapter 5 5.1 The ISO 8655 standard gives the accuracy and precision limits as both absolute and relative values. Specifications will depend on the technique
Pipette specifications according to ISO 8655 Chapter 5 5.1 The ISO 8655 standard gives the accuracy and precision limits as both absolute and relative values. Specifications will depend on the technique
Transferpette. Testing Instructions (SOP) 1. Introduction. October 1998
 Transferpette Testing Instructions (SOP) October 1998 1. Introduction The standards ISO DIS 8655 and DIN 12650 describe both the design and the testing of the piston operated pipette Transferpette. The
Transferpette Testing Instructions (SOP) October 1998 1. Introduction The standards ISO DIS 8655 and DIN 12650 describe both the design and the testing of the piston operated pipette Transferpette. The
CHEM 321 Experiment 1
 CHEM 321 Experiment 1 Basics Review and Calibration of Volumetric Glassware There are three types of containers used in lab to contain or deliver liquids: volumetric, ordinary and disposable glassware.
CHEM 321 Experiment 1 Basics Review and Calibration of Volumetric Glassware There are three types of containers used in lab to contain or deliver liquids: volumetric, ordinary and disposable glassware.
Standard Operating Procedure. Air Displacement Pipette Calibration
 University of Colorado at Denver October 28, 2003 - Revision 1.00 Page 1 of 7 1 Background: Standard Operating Procedure An accurate pipette is one of the most important tools in performing accurate analytical
University of Colorado at Denver October 28, 2003 - Revision 1.00 Page 1 of 7 1 Background: Standard Operating Procedure An accurate pipette is one of the most important tools in performing accurate analytical
1. Determining Solution Concentration
 In this exercise you will determine the concentration of salt solutions by measuring samples with known concentration and making a calibration curve. You will review units of concentration, and how to
In this exercise you will determine the concentration of salt solutions by measuring samples with known concentration and making a calibration curve. You will review units of concentration, and how to
Bante821 Portable Dissolved Oxygen Meter Instruction Manual
 Bante821 Portable Dissolved Oxygen Meter Instruction Manual BANTE INSTRUMENTS CO., LTD Bante821 Portable Dissolved Oxygen Meter 1 Introduction Thank you for selecting the Bante821 portable dissolved oxygen
Bante821 Portable Dissolved Oxygen Meter Instruction Manual BANTE INSTRUMENTS CO., LTD Bante821 Portable Dissolved Oxygen Meter 1 Introduction Thank you for selecting the Bante821 portable dissolved oxygen
Determination of the Gas-Law Constant (R) using CO2
 Determination of the Gas-Law Constant (R) using CO2 EXPERIMENT 11 Prepared by Edward L. Brown and Miranda Raines, Lee University The student will become familiar with ideal gases and how their properties
Determination of the Gas-Law Constant (R) using CO2 EXPERIMENT 11 Prepared by Edward L. Brown and Miranda Raines, Lee University The student will become familiar with ideal gases and how their properties
Determination of Zn using Atomic Absorption with Multiple Standard Additions
 1. Purpose Determination of Zn using Atomic Absorption with Multiple Standard Additions This procedure will determine the concentration of zinc at the parts-per-million level using flame atomic absorption
1. Purpose Determination of Zn using Atomic Absorption with Multiple Standard Additions This procedure will determine the concentration of zinc at the parts-per-million level using flame atomic absorption
Transferpette. Lightweight precise rugged reliable!
 Lightweight precise rugged reliable! Transferpette Solutions for S cience F I R S T C L A S S B R A N D The Transferpette S microliter pipettes provide all features required for pipetting in Life Science
Lightweight precise rugged reliable! Transferpette Solutions for S cience F I R S T C L A S S B R A N D The Transferpette S microliter pipettes provide all features required for pipetting in Life Science
Quantos Automated Dosing Solution Preparation Precise concentrations Process compliance Minimize out-of-specs
 Quantos Automated Dosing Solution Preparation Precise concentrations Process compliance Minimize out-of-specs Gravimetric Sample Preparation The Alternative to Volumetric Flasks From Volumetric to Gravimetric
Quantos Automated Dosing Solution Preparation Precise concentrations Process compliance Minimize out-of-specs Gravimetric Sample Preparation The Alternative to Volumetric Flasks From Volumetric to Gravimetric
Transferpette. Lightweight precise rugged reliable!
 Lightweight precise rugged reliable! Transferpette Solutions for S cience F I R S T C L A S S B R A N D The Transferpette S microliter pipettes provide all features required for pipetting in Life Science
Lightweight precise rugged reliable! Transferpette Solutions for S cience F I R S T C L A S S B R A N D The Transferpette S microliter pipettes provide all features required for pipetting in Life Science
DOscan10 Pocket Dissolved Oxygen Tester Instruction Manual
 DOscan10 Pocket Dissolved Oxygen Tester Instruction Manual BANTE INSTRUMENTS CO., LTD DOscan10 Pocket Dissolved Oxygen Tester 1 Thank you for selecting the DOscan10 pocket dissolved oxygen tester. This
DOscan10 Pocket Dissolved Oxygen Tester Instruction Manual BANTE INSTRUMENTS CO., LTD DOscan10 Pocket Dissolved Oxygen Tester 1 Thank you for selecting the DOscan10 pocket dissolved oxygen tester. This
Bante810 Benchtop Dissolved Oxygen Meter Instruction Manual
 Bante810 Benchtop Dissolved Oxygen Meter Instruction Manual BANTE INSTRUMENTS CO., LTD Bante810 Benchtop Dissolved Oxygen Meter 1 Introduction Thank you for selecting the Bante810 benchtop dissolved oxygen
Bante810 Benchtop Dissolved Oxygen Meter Instruction Manual BANTE INSTRUMENTS CO., LTD Bante810 Benchtop Dissolved Oxygen Meter 1 Introduction Thank you for selecting the Bante810 benchtop dissolved oxygen
Pipettor. User Manual
 Pipettor User Manual Product Code Description Single Chanel Pipettor Variable 550.002.055 Volume 0.5 to 10ul 550.002.060 2 to 20ul 550.002.065 10 to 100ul 550.002.070 20 to 200ul 550.002.075 100 to 100ul
Pipettor User Manual Product Code Description Single Chanel Pipettor Variable 550.002.055 Volume 0.5 to 10ul 550.002.060 2 to 20ul 550.002.065 10 to 100ul 550.002.070 20 to 200ul 550.002.075 100 to 100ul
DURING the course of certain investigations it became
 VOLUMETRIC DETERMINATION OF ETHER OR CYCLOPROPANE, CARBON DIOXIDE, NITROUS OXIDE AND OXYGEN IN ANESTHETIC MIXTURES By F. J. PRIME DURING the course of certain investigations it became necessary to be able
VOLUMETRIC DETERMINATION OF ETHER OR CYCLOPROPANE, CARBON DIOXIDE, NITROUS OXIDE AND OXYGEN IN ANESTHETIC MIXTURES By F. J. PRIME DURING the course of certain investigations it became necessary to be able
METHOD 21 - DETERMINATION OF VOLATILE ORGANIC COMPOUND LEAKS. 1.2 Scope. This method is applicable for the
 1151 METHOD 21 - DETERMINATION OF VOLATILE ORGANIC COMPOUND LEAKS 1.0 Scope and Application. 1.1 Analytes. Analyte Volatile Organic Compounds (VOC) CAS No. No CAS number assigned 1.2 Scope. This method
1151 METHOD 21 - DETERMINATION OF VOLATILE ORGANIC COMPOUND LEAKS 1.0 Scope and Application. 1.1 Analytes. Analyte Volatile Organic Compounds (VOC) CAS No. No CAS number assigned 1.2 Scope. This method
GCMSD-Headspace Analysis SOP
 Before you start GCMSD-Headspace Analysis SOP Method and Sequences names are restricted to 50 characters (MSD program will crash otherwise) The GC oven and Injection Ports need to be cooled to 50 oc to
Before you start GCMSD-Headspace Analysis SOP Method and Sequences names are restricted to 50 characters (MSD program will crash otherwise) The GC oven and Injection Ports need to be cooled to 50 oc to
Tips for Proper Pipette handling and Maintenance. D. Muruganand, Ph.D ILQA - Bangalore, Feb 2014
 Tips for Proper Pipette handling and Maintenance D. Muruganand, Ph.D ILQA - Bangalore, Feb 2014 Liquid handling tools Manual pipettes Electronic pipette and dispenser Automated systems Principles of Pipetting
Tips for Proper Pipette handling and Maintenance D. Muruganand, Ph.D ILQA - Bangalore, Feb 2014 Liquid handling tools Manual pipettes Electronic pipette and dispenser Automated systems Principles of Pipetting
The use of the analytical balance, and the buret.
 1211L Experiment 1. Density 2015 by H. Patterson Instructor Notes: Students make measurements individually then share data to make the graph. There are four volumetric measurements to be studied; 3.00
1211L Experiment 1. Density 2015 by H. Patterson Instructor Notes: Students make measurements individually then share data to make the graph. There are four volumetric measurements to be studied; 3.00
INTRODUCTION TO THE SPECTROPHOTOMETER AND PIPETTING SKILLS
 INTRODUCTION TO THE SPECTROPHOTOMETER AND PIPETTING SKILLS Section A: Intro to the spectrophotometer A commonly used instrument in the analysis of cellular extracts is the Spectrophotometer. Today you
INTRODUCTION TO THE SPECTROPHOTOMETER AND PIPETTING SKILLS Section A: Intro to the spectrophotometer A commonly used instrument in the analysis of cellular extracts is the Spectrophotometer. Today you
II. MATERIALS REQUIRED FOR PERFORMANCE TESTING
 TSP-016 P/N L038-058 4-001 Rev. D February 8, 2000 Page 1 of 14 DISTRIBUTION: VistaLab Technologies Customers EQUIPMENT: MLA Pipettes SUBJECT: Pipette Performance Verification Protocol PURPOSE: To evaluate
TSP-016 P/N L038-058 4-001 Rev. D February 8, 2000 Page 1 of 14 DISTRIBUTION: VistaLab Technologies Customers EQUIPMENT: MLA Pipettes SUBJECT: Pipette Performance Verification Protocol PURPOSE: To evaluate
Experiment #2. Density and Measurements
 Experiment #2. Density and Measurements Goals 1. To measure and record length, volume and mass accurately with the correct number of significant figures 2. To use significant figures correctly in calculations.
Experiment #2. Density and Measurements Goals 1. To measure and record length, volume and mass accurately with the correct number of significant figures 2. To use significant figures correctly in calculations.
Determination of Zn using Atomic Absorption with Multiple Standard Additions
 Determination of Zn using Atomic Absorption with Multiple Standard Additions 1. Purpose This procedure will determine the concentration of zinc at the parts-per-million level using flame atomic absorption
Determination of Zn using Atomic Absorption with Multiple Standard Additions 1. Purpose This procedure will determine the concentration of zinc at the parts-per-million level using flame atomic absorption
Evaluation copy. Interdependence of Plants and Animals. computer OBJECTIVES MATERIALS
 Interdependence of Plants and Animals Computer 14 Plants and animals share many of the same chemicals throughout their lives. In most ecosystems, O 2, CO 2, water, food and nutrients are exchanged between
Interdependence of Plants and Animals Computer 14 Plants and animals share many of the same chemicals throughout their lives. In most ecosystems, O 2, CO 2, water, food and nutrients are exchanged between
FOURIER TRANSFORM INFRARED SPECTROSCOPY
 Purpose and Scope: This document describes the procedures and policies for using the MSE Bruker FTIR Spectrometer. The scope of this document is to establish user procedures. Instrument maintenance and
Purpose and Scope: This document describes the procedures and policies for using the MSE Bruker FTIR Spectrometer. The scope of this document is to establish user procedures. Instrument maintenance and
FLUORESCENCE DETERMINATION OF OXYGEN
 FLUORESCENCE DETERMINATION OF OXYGEN Objectives This experiment will familiarize the students with the principles of fluorescence quenching as well as the Ocean Optics FOXY Fiber Optic Sensor System. Spectra
FLUORESCENCE DETERMINATION OF OXYGEN Objectives This experiment will familiarize the students with the principles of fluorescence quenching as well as the Ocean Optics FOXY Fiber Optic Sensor System. Spectra
Check-In, Lab Safety, Balance and Volumetric Glassware Use, Introduction to Statistics, and MSDS Familiarization
 Check-In, Lab Safety, Balance and Volumetric Glassware Use, Introduction to Statistics, and MSDS Familiarization Before coming to lab: Read Chapter 2 of your textbook before coming to lab. This chapter
Check-In, Lab Safety, Balance and Volumetric Glassware Use, Introduction to Statistics, and MSDS Familiarization Before coming to lab: Read Chapter 2 of your textbook before coming to lab. This chapter
Oct 1, 2002 LAB MANUAL SPECIFIC GRAVITY & ABSORPTION of FINE AGGREGATE AASHTO Designation T 84 (Mn/DOT Modified)
 Oct 1, 2002 LAB MANUAL 1205.0 1205 SPECFC GRAVTY & ABSORPTON of FNE AGGREGATE AASHTO Designation T 84 (Mn/DOT Modified) 1205.1 GENERAL This test method is intended for use in determining the bulk and apparent
Oct 1, 2002 LAB MANUAL 1205.0 1205 SPECFC GRAVTY & ABSORPTON of FNE AGGREGATE AASHTO Designation T 84 (Mn/DOT Modified) 1205.1 GENERAL This test method is intended for use in determining the bulk and apparent
Real World Metric System
 Real World Metric System Learning Objectives 1. Identify the metric units of weight, length, volume and temperature. 2. Make measurements using the metric system. 3. Convert values between different metric
Real World Metric System Learning Objectives 1. Identify the metric units of weight, length, volume and temperature. 2. Make measurements using the metric system. 3. Convert values between different metric
Ch. 2: Tools of the Trade
 Ch. 2: Tools of the Trade Outline: 2-1 Safety 2-2 Notebook 2-3 Analytical Balance 2-4 Burets 2-5 Volumetric Flasks 2-6 Pipets and Syringes 2-7 Filtration 2-8 Drying 2-9 Calibration of Volumetric Glassware
Ch. 2: Tools of the Trade Outline: 2-1 Safety 2-2 Notebook 2-3 Analytical Balance 2-4 Burets 2-5 Volumetric Flasks 2-6 Pipets and Syringes 2-7 Filtration 2-8 Drying 2-9 Calibration of Volumetric Glassware
Determination of Sodium using Atomic Emission
 Determination of Sodium using Atomic Emission 1. Purpose The purpose of this procedure is to determine the concentration of sodium ion in parts per million in an unknown sample. 2. Background Atomic emission
Determination of Sodium using Atomic Emission 1. Purpose The purpose of this procedure is to determine the concentration of sodium ion in parts per million in an unknown sample. 2. Background Atomic emission
25/04/56. LTC Carmen A. Bell Chief, Laboratory Operations USAMC-AFRIMS
 Pipette Theory and Maintenance LTC Carmen A. Bell Chief, Laboratory Operations USAMC-AFRIMS carmen.bell@afrims.org +6602-696-2725 1 References Pharma Express: http://pharma.financialexpress.com/2008063
Pipette Theory and Maintenance LTC Carmen A. Bell Chief, Laboratory Operations USAMC-AFRIMS carmen.bell@afrims.org +6602-696-2725 1 References Pharma Express: http://pharma.financialexpress.com/2008063
Gaseous oxygen technical grade Specification
 CDC 09 (5010) P3 Rev. TZS 217:1984 FINALIZED TANZANIA STANDARD Gaseous oxygen technical grade Specification TANZANIA BUREAU OF STANDARDS. 2 nd edition TBS 2017 0. FOREWORD 0.1. This Tanzania Draft Standard
CDC 09 (5010) P3 Rev. TZS 217:1984 FINALIZED TANZANIA STANDARD Gaseous oxygen technical grade Specification TANZANIA BUREAU OF STANDARDS. 2 nd edition TBS 2017 0. FOREWORD 0.1. This Tanzania Draft Standard
RayPette Plus Autoclavable Pipette. User Manual
 RayPette Plus Autoclavable Pipette User Manual CONTENTS 1. YOUR NEW PIPETTE... 1 1.1. Adjustable volume pipettes... 1 1.3 Fully autoclavable... 2 2. UNPACKING... 3 3. INSTALLING THE PIPETTE HOLDER... 4
RayPette Plus Autoclavable Pipette User Manual CONTENTS 1. YOUR NEW PIPETTE... 1 1.1. Adjustable volume pipettes... 1 1.3 Fully autoclavable... 2 2. UNPACKING... 3 3. INSTALLING THE PIPETTE HOLDER... 4
Read this first. Zetasizer nano series Self installation and Quick start guide MRK825-02
 ! Read this first Zetasizer nano series Self installation and Quick start guide I N S T R U M E N T S MRK825-02 Zetasizer Nano series Self installation and Quick start guide MAN0383 Issue 1.1 July 2007
! Read this first Zetasizer nano series Self installation and Quick start guide I N S T R U M E N T S MRK825-02 Zetasizer Nano series Self installation and Quick start guide MAN0383 Issue 1.1 July 2007
Using the Akta Prime plus October 22, 2012
 Some starting precautions: 1. Vacuum filter all buffers. Removes any large particles/debris that may clog your column De-gases the buffers 2. Clarify lysates first by centrifugation and then filtration
Some starting precautions: 1. Vacuum filter all buffers. Removes any large particles/debris that may clog your column De-gases the buffers 2. Clarify lysates first by centrifugation and then filtration
USER'S GUIDE. Nanoflow Probe. Red Stripe. 3-4mm. ptfe Sleeve. To Injector. ZU1XC Fused Silica TTF115. Fused Silica. ptfe Sleeve
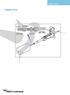 USER'S GUIDE Nanoflow Probe 3-4mm Red Stripe ptfe Sleeve To Injector ZU1XC Fused Silica 20µm 90µm TTF115 ptfe Sleeve Fused Silica 25µm 375µm Micromass UK Limited Floats Road Wythenshawe M23 9LZ Tel: +44
USER'S GUIDE Nanoflow Probe 3-4mm Red Stripe ptfe Sleeve To Injector ZU1XC Fused Silica 20µm 90µm TTF115 ptfe Sleeve Fused Silica 25µm 375µm Micromass UK Limited Floats Road Wythenshawe M23 9LZ Tel: +44
Ensuring pipette & weighing device performance: Verification & Calibration
 Ensuring pipette & weighing device performance: Verification & Calibration Wan Melissa Diyana bt Wan Normazlan Central Research Laboratories, Faculty of Medicine terms What is preventive maintenance? Regularly
Ensuring pipette & weighing device performance: Verification & Calibration Wan Melissa Diyana bt Wan Normazlan Central Research Laboratories, Faculty of Medicine terms What is preventive maintenance? Regularly
Setting up and running a column
 Setting up and running a column PART 1: What you need and theory PART 2: Using a bellow and packing the column PART 3: Loading sample to silica PART 4: Running column PART 5: TLC analysis and combining
Setting up and running a column PART 1: What you need and theory PART 2: Using a bellow and packing the column PART 3: Loading sample to silica PART 4: Running column PART 5: TLC analysis and combining
Operating Instructions METTLER TOLEDO Pipette Check Application for AX and MX/UMX Balances Version 1.xx
 Operating Instructions METTLER TOLEDO Pipette Check Application for AX and MX/UMX Balances Version 1.xx Contents 1 Introducing the Pipette Check application... 3 2 important notes... 3 3 Selecting the
Operating Instructions METTLER TOLEDO Pipette Check Application for AX and MX/UMX Balances Version 1.xx Contents 1 Introducing the Pipette Check application... 3 2 important notes... 3 3 Selecting the
UNITY 2 TM. Air Server Series 2 Operators Manual. Version 1.0. February 2008
 UNITY 2 TM Air Server Series 2 Operators Manual Version 1.0 February 2008 1. Introduction to the Air Server Accessory for UNITY 2...2 1.1. Summary of Operation...2 2. Developing a UNITY 2-Air Server method
UNITY 2 TM Air Server Series 2 Operators Manual Version 1.0 February 2008 1. Introduction to the Air Server Accessory for UNITY 2...2 1.1. Summary of Operation...2 2. Developing a UNITY 2-Air Server method
Standard Practice for Calibration of Laboratory Volumetric Apparatus 1
 Designation: E 542 0 Standard Practice for Calibration of Laboratory Volumetric Apparatus This standard is issued under the fixed designation E 542; the number immediately following the designation indicates
Designation: E 542 0 Standard Practice for Calibration of Laboratory Volumetric Apparatus This standard is issued under the fixed designation E 542; the number immediately following the designation indicates
Register your instrument! Eppendorf Xplorer / Eppendorf Xplorer plus. Adjustment
 lorer /Eppendorf N) t Xplorer plus Register your instrument! www.eppendorf.com/myeppendorf Eppendorf Xplorer / Eppendorf Xplorer plus Adjustment Copyright 2015 Eppendorf AG, Germany. All rights reserved,
lorer /Eppendorf N) t Xplorer plus Register your instrument! www.eppendorf.com/myeppendorf Eppendorf Xplorer / Eppendorf Xplorer plus Adjustment Copyright 2015 Eppendorf AG, Germany. All rights reserved,
The pipettes cover a volume range from 0.1µl to 10ml.
 CONTENTS 1. YOUR NEW PIPETTE 1 1.1. Adjustable volume pipettes 1 1.2. Fixed volume pipettes 2 1.3 Fully autoclavable 3 2. UNPACKING 3 3. INSTALLING THE PIPETTE HOLDER 4 4. PIPETTE COMPONENTS 5 5. PIPETTE
CONTENTS 1. YOUR NEW PIPETTE 1 1.1. Adjustable volume pipettes 1 1.2. Fixed volume pipettes 2 1.3 Fully autoclavable 3 2. UNPACKING 3 3. INSTALLING THE PIPETTE HOLDER 4 4. PIPETTE COMPONENTS 5 5. PIPETTE
Fisherbrand. Finnpipette INSTRUCTIONS FOR USE PARK LANE Pittsburgh PA
 Fisherbrand Finnpipette INSTRUCTIONS FOR USE 1 0. 0 1 0. 0 0 2000 PARK LANE Pittsburgh PA 15275 800-766-7000 1 CONTENTS PRODUCT DESCRIPTION 3 PIPETTE OPERATION 4 PIPETTING TECHNIQUES 4, 5 CALIBRATION 6,
Fisherbrand Finnpipette INSTRUCTIONS FOR USE 1 0. 0 1 0. 0 0 2000 PARK LANE Pittsburgh PA 15275 800-766-7000 1 CONTENTS PRODUCT DESCRIPTION 3 PIPETTE OPERATION 4 PIPETTING TECHNIQUES 4, 5 CALIBRATION 6,
Qi Litre Ultrasonic Cleaner
 Qi-300 17Litre Ultrasonic Cleaner Ultrawave s Qi-Series are robust and reliable, delivering outstanding cleaning performance in a wide range of industries. From precision, pharmaceutical and medical device
Qi-300 17Litre Ultrasonic Cleaner Ultrawave s Qi-Series are robust and reliable, delivering outstanding cleaning performance in a wide range of industries. From precision, pharmaceutical and medical device
Elcometer 138. Bresle Kit and Patches. Operating Instructions
 Elcometer 138 English Bresle Kit and Patches Operating Instructions R English The Conductivity Meter model B-173 has been tested in accordance with EU regulations governing Electro-magnetic compliance
Elcometer 138 English Bresle Kit and Patches Operating Instructions R English The Conductivity Meter model B-173 has been tested in accordance with EU regulations governing Electro-magnetic compliance
Purpose. Introduction
 Purpose The objective of this experiment is to determine the density of an unknown liquid and solid. The students will become familiar with the techniques for measuring mass and volume of several samples
Purpose The objective of this experiment is to determine the density of an unknown liquid and solid. The students will become familiar with the techniques for measuring mass and volume of several samples
RayBio Human vwf ELISA Kit
 RayBio Human vwf ELISA Kit Catalog #: ELH-vWF User Manual Last revised April 15, 2016 Caution: Extraordinarily useful information enclosed ISO 13485 Certified 3607 Parkway Lane, Suite 100 Norcross, GA
RayBio Human vwf ELISA Kit Catalog #: ELH-vWF User Manual Last revised April 15, 2016 Caution: Extraordinarily useful information enclosed ISO 13485 Certified 3607 Parkway Lane, Suite 100 Norcross, GA
SOP for use of the SX19 Stopped-Flow Instrument
 SOP for use of the SX19 Stopped-Flow Instrument The main components of the stopped flow system are: Always book the system before use. Starting up and preparing the SX19 for use: 1. Turn on the compressed
SOP for use of the SX19 Stopped-Flow Instrument The main components of the stopped flow system are: Always book the system before use. Starting up and preparing the SX19 for use: 1. Turn on the compressed
Lab Skills Practice: Pipetting Small Volumes. B3 Summer Science Camp at Olympic High School 2016
 Lab Skills Practice: Pipetting Small Volumes B3 Summer Science Camp at Olympic High School 2016 Pipetter types Serological and micropipettes are used to accurately transfer small liquid volumes (micro-liter
Lab Skills Practice: Pipetting Small Volumes B3 Summer Science Camp at Olympic High School 2016 Pipetter types Serological and micropipettes are used to accurately transfer small liquid volumes (micro-liter
EXPERIMENT 1 TOOLS FOR LAB MEASUREMENT
 EXPERIMENT 1 TOOLS FOR LAB MEASUREMENT INTRODUCTION A course in chemistry, one of the physical sciences, differs from a course in, say, literature or history. A main difference is that chemistry usually
EXPERIMENT 1 TOOLS FOR LAB MEASUREMENT INTRODUCTION A course in chemistry, one of the physical sciences, differs from a course in, say, literature or history. A main difference is that chemistry usually
Inquiry Module 1: Checking the calibration of a micropipette
 Inquiry Module 1: Checking the calibration of a micropipette 1. Introduction Larger volumes (1mL and more) are usually measured using pipets or measuring cylinders. Such cylinders and pipets are labelled
Inquiry Module 1: Checking the calibration of a micropipette 1. Introduction Larger volumes (1mL and more) are usually measured using pipets or measuring cylinders. Such cylinders and pipets are labelled
Elcometer 138. Bresle Kit and Patches. Operating Instructions
 Elcometer 138 English Bresle Kit and Patches Operating Instructions R English This product meets the Electromagnetic Compatibility Directive. The product is Class B, Group 1 ISM equipment according to
Elcometer 138 English Bresle Kit and Patches Operating Instructions R English This product meets the Electromagnetic Compatibility Directive. The product is Class B, Group 1 ISM equipment according to
General European OMCL Network (GEON) QUALITY MANAGEMENT DOCUMENT
 General European OMCL Network (GEON) QUALITY MANAGEMENT DOCUMENT PA/PH/OMCL (09) 64 R5 QUALIFICATION OF EQUIPMENT ANNEX 6: QUALIFICATION OF PISTON PIPETTES Full document title and reference Document type
General European OMCL Network (GEON) QUALITY MANAGEMENT DOCUMENT PA/PH/OMCL (09) 64 R5 QUALIFICATION OF EQUIPMENT ANNEX 6: QUALIFICATION OF PISTON PIPETTES Full document title and reference Document type
UNCC Biotechnology and Bioinformatics Camp. Dr. Jennifer Weller Summer 2010
 UNCC Biotechnology and Bioinformatics Camp Dr. Jennifer Weller Summer 2010 Using Micropipettes Lab Intro Micropipettes How do you control volume in the microliter range? How do you verify that the volume
UNCC Biotechnology and Bioinformatics Camp Dr. Jennifer Weller Summer 2010 Using Micropipettes Lab Intro Micropipettes How do you control volume in the microliter range? How do you verify that the volume
ali-q TM Gravimetric Verification Procedure
 ali-q TM Gravimetric Verification Procedure Introduction This document describes the recommended procedure for verifying performance of the ali-q aliquoting pipet controller using the industry standard-
ali-q TM Gravimetric Verification Procedure Introduction This document describes the recommended procedure for verifying performance of the ali-q aliquoting pipet controller using the industry standard-
Register your instrument! Eppendorf Research plus. User Adjustment
 search nt plus stment N) Register your instrument! www.eppendorf.com/myeppendorf User Adjustment Copyright 2013 Eppendorf AG, Hamburg. No part of this publication may be reproduced without the prior permission
search nt plus stment N) Register your instrument! www.eppendorf.com/myeppendorf User Adjustment Copyright 2013 Eppendorf AG, Hamburg. No part of this publication may be reproduced without the prior permission
Dissolved Oxygen in Water. Evaluation copy. Table 1. Temperature Range ( C) Trout Smallmouth bass Caddisfly larvae
 Dissolved Oxygen in Water Computer 12A Although water is composed of oxygen and hydrogen atoms, biological life in water depends upon another form of oxygen molecular oxygen. Oxygen is used by organisms
Dissolved Oxygen in Water Computer 12A Although water is composed of oxygen and hydrogen atoms, biological life in water depends upon another form of oxygen molecular oxygen. Oxygen is used by organisms
Soil Water Characteristic Curve (SWCC) Using the WP4C
 Energy GeoEngineering Laboratory Soil Water Characteristic Curve (SWCC) Using the WP4C Prepared by: Josbel Cordero & Geralds Useche Note: This pictorial manual is not intended to replace the official Decagon
Energy GeoEngineering Laboratory Soil Water Characteristic Curve (SWCC) Using the WP4C Prepared by: Josbel Cordero & Geralds Useche Note: This pictorial manual is not intended to replace the official Decagon
Abbe 5 Refractometer
 Abbe 5 Refractometer User Guide DECLARATION OF CONFORMITY According to BS EN ISO/IEC 17050-1 Manufacturer's Name Manufacturer's Address Bellingham & Stanley Ltd. Longfield Road, Tunbridge Wells, Kent TN2
Abbe 5 Refractometer User Guide DECLARATION OF CONFORMITY According to BS EN ISO/IEC 17050-1 Manufacturer's Name Manufacturer's Address Bellingham & Stanley Ltd. Longfield Road, Tunbridge Wells, Kent TN2
Any laboratory is equipped with specific tools, equipment,
 Use of Laboratory Equipment and Supplies 3 When you have completed this exercise, you will be able to: 1. Use a balance. 2. Use pipettes and graduated cylinders to measure the volume of liquids. 3. Use
Use of Laboratory Equipment and Supplies 3 When you have completed this exercise, you will be able to: 1. Use a balance. 2. Use pipettes and graduated cylinders to measure the volume of liquids. 3. Use
Heavy Duty Dissolved Oxygen Meter
 User's Manual Heavy Duty Dissolved Oxygen Meter Model 407510 Test Equipment Depot - 800.517.8431-99 Washington Street Melrose, MA 02176 FAX 781.665.0780 - TestEquipmentDepot.com Introduction Congratulations
User's Manual Heavy Duty Dissolved Oxygen Meter Model 407510 Test Equipment Depot - 800.517.8431-99 Washington Street Melrose, MA 02176 FAX 781.665.0780 - TestEquipmentDepot.com Introduction Congratulations
FRANZ CELL TEST OF NANO GLUTATHIONE WITH HPLC ANALYSIS
 FRANZ CELL TEST OF NANO GLUTATHIONE WITH HPLC ANALYSIS Analysis Conducted BY CC RESEARCH (P) Ltd., For Nanoceutical Solutions Inc. USA 2950 Thousand Oaks drive, Suite 3, San Antonio, Texas EXECUTIVE SUMMARY
FRANZ CELL TEST OF NANO GLUTATHIONE WITH HPLC ANALYSIS Analysis Conducted BY CC RESEARCH (P) Ltd., For Nanoceutical Solutions Inc. USA 2950 Thousand Oaks drive, Suite 3, San Antonio, Texas EXECUTIVE SUMMARY
Part 1: Distillation and Analysis of an Unknown Alcohol Mixture
 Experiment DE: Part 1 Experiment DE has multiple goals, including -separation and quantification of a 2-component mixture (fractional distillation and GC); preliminary ID of both components -preparation
Experiment DE: Part 1 Experiment DE has multiple goals, including -separation and quantification of a 2-component mixture (fractional distillation and GC); preliminary ID of both components -preparation
Accuracy and Precision
 Accuracy and Precision Introduction Scientists use many skills as they investigate the world around them. They make observations by gathering information with their senses. Some observations are simple.
Accuracy and Precision Introduction Scientists use many skills as they investigate the world around them. They make observations by gathering information with their senses. Some observations are simple.
MSD 96-Well MULTI-ARRAY and MULTI-SPOT Human Granulocyte Colony Stimulating Factor (hg-csf) Ultrasensitive Assay
 MSD 96-Well MULTI-ARRAY and MULTI-SPOT Human Granulocyte Colony Stimulating Factor (hg-csf) Ultrasensitive Assay Summary This assay measures Human Granulocyte Colony Stimulating Factor (G-CSF) in a 96-well
MSD 96-Well MULTI-ARRAY and MULTI-SPOT Human Granulocyte Colony Stimulating Factor (hg-csf) Ultrasensitive Assay Summary This assay measures Human Granulocyte Colony Stimulating Factor (G-CSF) in a 96-well
Purpose and Scope: Responsibilities: Warnings and precautions: Background Beers law
 Purpose and Scope: This document describes the procedures and policies for using the MSE Bruker FTIR Spectrometer. The scope of this document is to establish user procedures. Instrument maintenance and
Purpose and Scope: This document describes the procedures and policies for using the MSE Bruker FTIR Spectrometer. The scope of this document is to establish user procedures. Instrument maintenance and
Human Factor X Chromogenic Activity Kit
 AssaySense Human Factor X Chromogenic Activity Kit Assaypro LLC 3400 Harry S Truman Blvd St. Charles, MO 63301 T (636) 447-9175 F (636) 395-7419 www.assaypro.com For any questions regarding troubleshooting
AssaySense Human Factor X Chromogenic Activity Kit Assaypro LLC 3400 Harry S Truman Blvd St. Charles, MO 63301 T (636) 447-9175 F (636) 395-7419 www.assaypro.com For any questions regarding troubleshooting
Pipetting Small Volumes
 Pipetting Small Volumes Introduction Serological and micropipettes are used to accurately transfer small liquid volumes (micro-liter to milli-liter) accurately and precisely. Continuously adjustable Can
Pipetting Small Volumes Introduction Serological and micropipettes are used to accurately transfer small liquid volumes (micro-liter to milli-liter) accurately and precisely. Continuously adjustable Can
FTIR Users Group Meeting FTIR development
 FTIR Users Group Meeting FTIR development A bit of history From Oofty. To Spectronus A bit of history Oofty, Univ. Of Wollongong, 1995 They developed a method of trace gas analysis based on 1-cm -1 resolution
FTIR Users Group Meeting FTIR development A bit of history From Oofty. To Spectronus A bit of history Oofty, Univ. Of Wollongong, 1995 They developed a method of trace gas analysis based on 1-cm -1 resolution
Ultra-Low Copper Concentrations Determined by Rotating Disk Electrode Stripping Voltammetry
 Ultra-Low Copper Concentrations Determined by Rotating Disk Electrode Stripping Voltammetry 1. Purpose Copper ion concentrations in the parts-per-billion (ppb) range will be determined using anodic stripping
Ultra-Low Copper Concentrations Determined by Rotating Disk Electrode Stripping Voltammetry 1. Purpose Copper ion concentrations in the parts-per-billion (ppb) range will be determined using anodic stripping
BS ml Bottle Sampling Unit
 BS500 500ml Bottle Sampling Unit User Guide www.mpfiltri.co.uk 200.307-EN Covers Model Numbers BS500 SAFETY WARNING Hydraulic systems contain dangerous fluids at high pressures and temperatures. Installation,
BS500 500ml Bottle Sampling Unit User Guide www.mpfiltri.co.uk 200.307-EN Covers Model Numbers BS500 SAFETY WARNING Hydraulic systems contain dangerous fluids at high pressures and temperatures. Installation,
EXPERIMENT 2. Laboratory Procedures INTRODUCTION
 EXPERIMENT 2 Laboratory Procedures INTRODUCTION Begin each experiment by taking the necessary safety precautions. All materials that will not be used in the lab should be placed out of the laboratory working
EXPERIMENT 2 Laboratory Procedures INTRODUCTION Begin each experiment by taking the necessary safety precautions. All materials that will not be used in the lab should be placed out of the laboratory working
