DIVING PHYSICS EXAMPLE QUESTIONS
|
|
|
- Denis Peters
- 5 years ago
- Views:
Transcription
1 DIVING PHYSICS EXAMPLE QUESTIONS PLEASE NOTE: 1 bar = 10 Meter in Salt water 1 bar = 10.2 Meter in Fresh water. Will be GIVEN to you for calculations. 10m in Salt water = 1 bar 10m in Fresh water = 0.98 bar (1/10.2 = 0.98) 1 Litre Salt Water = Kilograms 1 Litre Fresh Water = Kilograms 1. Q: For every meter of depth in fresh water, pressure increases by: A: From the info given, we know that 1 bar in fresh water is reached at 10.2m depth. Therefore, 1 / 10.2 = 0.98 bar per 10 meter. From this we take 0.98 / 10 and our answer is that for every meter of depth in fresh water, pressure increases by bar. (0.98 / 10) 2. Q: The absolute pressure at a depth of 40m in salt water is: A: We know that for every 10metres of salt water, pressure increases by 1 bar. Taking into account the 1 bar atmospheric pressure; we calculate pressure in salt water, as follows: (Depth / 10) +1 Therefore pressure would be at 10m: (10/10) + 1 = 2bar 20m: (20/10) + 1 = 3bar 30m: (30/10) + 1 = 4bar 40m: (40/10) + 1 = 5bar Our answer is therefore at 40msw the pressure is 5 bars 3. Q: A known volume of air in a flexible container expands the most during a salt water ascent from: A: Pressure changes from 5bar to 4bar when ascending from 40msw to 30msw. 5/4 = 1.25 Pressure changes from 4bar to 3bar when ascending from 30msw to 20msw. 4/3 = 1.33 Pressure changes from 3bar to 2bar when ascending from 20msw to 10msw. 3/2 = 1.50 Pressure changes from 2bar to 1bar when ascending from 10msw to surface. 2/1 = 2.00 THEREFORE the largest pressure difference is from 10msw to the surface.
2 4. Q: An object with a volume of 117 litres, weighing 145kg is located at a depth of 30m in the ocean. How many 25 litre lift bags weighing 2.5kg each would it take to bring the object to the surface? A: According to Archimedes Principle, a body submersed in water is buoyed up by a force equal to the weight of the water it displaces. Therefore, the object weighing 145kg is buoyed up by (117 x 1.025kg which is the weight of salt water, = kg lift (buoyancy) = kg needed to lift the object. Our lift bags have a capacity of 25 litres each and weigh 2.5kg each therefore the lift capacity is only (25*1.025)-2.5 = kg lift capacity PER BAG? = 1.95 therefore, 1 lift bag is not enough. We need TWO lift bags for this exercise. Q: Air consumption: a. Decreases as depth increases b. Increases as depth increases c. Remains the same as depth increases d. Varies inversely with depth 5. A: The air delivery system supplies air to the diver at ambient pressure. As the pressure increases during descent, the air delivery system automatically adjusts and supplies more air from the cylinder. A diver s usage will vary directly with depth. The diver will consume twice as much air with each breath at 10msw (2 bar) than he will at sea level (1bar), Three times as much at 20msw (3bar) Four times as much at 30msw (4 bar) 6. Q: What would be the surface air consumption rate (SAC) of a diver with a 200bar, 15 litre? cylinder who uses 15bar in 10 minutes at a depth of 5 meters in the ocean? SAC = AIR USED AIR USED = START PRESSURE END PRESSURE WHERE USED? A: SAC = (AIR USED) X CYLINDER SIZE DEPTH (BAR) X TIME (MINUTES) SAC = (15bar) x 15l 1.5bar x 10 minutes SAC = 225 / 15 = 15 litres per minute.
3 7. Q: The ocean depth at which the partial pressure of oxygen in air equals 1.6bar is meter. Assuming O2 = 20%. A: 1.6 bar / 0.2 (Oxygen fraction) = 8. 8 bar = 70msw 8. Q: Breathing 0.5% carbon monoxide at a depth of 40m in the ocean is equivalent to breathing what % of carbon monoxide at the surface. A: 0.5% x 5bar (40msw = 5bar) = 2.5% 9. Q: An object that is 1.5m square and high (1.5 x 1.5 x 1.5) is floating in the ocean. 20cm of the object is above the waterline. What is the minimum line strength? that would be needed to lift the object out of the water? A: 1.5 x 1.5 x 1.5 = 3.375m3 20cm is above the waterline therefore 1.5 x 1.5 x 0.2 is above the waterline. 1.5 x 1.5 x 0.2 = 0.45m3 Thus = 2.925m3 is submersed 1.5 x 1.5 x 1.3 = 2.925m x (because ocean water weighs 1.025kg per litre) = Therefore we would need a 3.000kg line to lift/move this object. 10. Q: If a diver and his equipment weighs 87 kg when neutrally buoyant in salt water, the diver needs to add / remove kg to be neutrally buoyant for freshwater diving with the same equipment. A: 87kg x = = 2.18 The diver would have to REMOVE 2.2KG in order to be neutrally buoyant in fresh water. 11. Q: If 2 litres of air is needed in your buoyancy compensator to maintain neutral buoyancy 10m in the ocean, how much will your buoyancy increase if you ascend 3m? A: 2litres of air 1.7 bar (at 7msw) x 2 bar (diver was initially at 2 bar at 10msw) 2 / 1.7 = x 2 = = 0.35 Buoyancy will change by 0.35l.
4 12. Q: A lift bag that weighs 1kg and contains 20 litres of air at a depth of 20m in the ocean would provide kg of buoyancy upon reaching the surface provided no air escaped during ascent. A: NOTE: The CAPACITY is not given therefore we assume the capacity is limitless. 20 litres of air at 20m will expand. 20litres at 3bar will expand to 3 x its original volume. Therefore 20 x 3 = 60 HOWEVER keep into account the weight bag weighs 1kg and therefore can only carry 59l of air. 60 1= Q: The pressure increases per meter of depth in saltwater by: A: For every 10metres, it is 1 bar. Therefore 1bar / 10msw = 0.1bar per 1meter salt water. 14. At an altitude of 5,500 meters, the atmospheric pressure is approximately HALF of the atmospheric pressure at sea level. WE USE THIS INFORMATION WHEN FLYING. 15. Q: If a scuba cylinder contains 12 litres of air at a pressure of 200bar, how much air does the cylinder contain at 150bar if the temperature remains constant? A: 12l cylinder x 150 bar = (12x150) 1800 bar litters. 16. Q: If a diver takes a sealed, rigid, cubical container measuring 50cm per side to a depth of 5m in to the ocean, the total crushing force (assume the wall thickness is zero) on the container would be about: A: Well, how much pressure is at 5msw? It s 1.5bar. It will remain 1.5bar no matter what you submerse. 17. Q: The partial pressure law requires the sum of the partial pressures to always equal the pressure. A: Dalton s law uses the term total pressure to describe the sum of the partial pressures of each gas in a mixture of gases. Total pressure is synonymous with absolute pressure. Always use absolute pressure, the term used in physics, for pressure calculations. The answer is TOTAL or ABSOLUTE.
5 18. Q: The absolute pressure at a depth of 20m in fresh water at an altitude of 1000m (atmospheric pressure is 0.9bar) is bar. A: FRESH WATER 1 bar is attained at 10.2 metres therefore 20mfw = 20/ /10.2 = 1.96 Atmospheric pressure is given as 0.9bar = 2.86 Therefore the answer is 2.86bar 19. Q: An object floats when it displaces water weighing the weight of the object.. A: A. As much as B. Less than. C. None of the answers are correct D. More than Let s think about this logically. An object is buoyed up by a force equal to the weight of the water it displaces. Coins, gold, and metals these are very dense and therefore sink to the bottom easily. Polystyrene is less dense, therefore floats easily. A small coin displaces less water, therefore it sinks. The inverse of this is an object that displaces MORE water than what it weighs, will FLOAT. So, if we force an empty, sealed plastic bottle into a bucket of water, the sealed bottle will displace more water than what the bottle weighs and therefore it will surface and float the moment we let go of it. If the weight of the water displaced by an object weighs more than the object itself, the object will be buoyed up by a force equal to the difference between the weight of the object and the weight of the water it displaces. 20. Q: Assuming a maximum partial pressure limitation of 1.4 ATA, the maximum depth for a 40% Nitrox mixture is m of seawater. A: PPO2 Depth (bar) EANx O2 Fraction PPO2 is given as 1.4. O2 Fraction is given as 0.40 (40%) 1.4/ 0.4= bar = 25msw
6 21. Q: What would be the Surface Respiratory Volume (SRV) of a diver with a 200 bar, 15 litre cylinder? who uses 15 bars in 10 min. at a depth of 5 msw in the ocean? A: (15bar x 15l cylinder) / (10 minutes x 1.5bar depth) 225 /15 15litres per minute 22. Q: An object with a volume of 30 litres, weighing 70 kg is embedded in the sediment at a depth of 30 meters in the ocean harbour. This object is best lifted to the surface by: A: 1. Two 40 litres lift bags weighing 3kg each Volume = 30l 30 x (weight of salt 2. A line tied to a boat at the surface water) = Three 250 litres lift bags weighing 20 kg each 1 lift bag provides (40-3) 37kg of lift. We 4. Three 500 litres lift bags and two 25 l lift bags we therefore need 2 x 40l lift bags. 23. Q: The ocean depth at which the partial pressure of oxygen in air equals 1.6 bars is assuming O2 = 20,9%. A: 1.6 / = 7.665bar 7.665bar 1bar atmospheric pressure = 6.665bar water pressure x 10 = 66.5msw
PHYSICS. Light: Sound:
 PHYSICS Light: The speed of light changes as it passes through different things such as air, glass and water. This affects the way we see things underwater with a diving mask. As the light passes through
PHYSICS Light: The speed of light changes as it passes through different things such as air, glass and water. This affects the way we see things underwater with a diving mask. As the light passes through
1/21/2014. Diving Physics. Introduction
 Diving Physics Topics in this Chapter Introduction Principles of Pressure Properties of Water Temperature Archimedes Principle Charles Law & Guy-Lussac s Law Combined & Ideal Gas Laws Dalton s Law Henry
Diving Physics Topics in this Chapter Introduction Principles of Pressure Properties of Water Temperature Archimedes Principle Charles Law & Guy-Lussac s Law Combined & Ideal Gas Laws Dalton s Law Henry
In the liquid phase, molecules can flow freely from position to position by sliding over one another. A liquid takes the shape of its container.
 In the liquid phase, molecules can flow freely from position to position by sliding over one another. A liquid takes the shape of its container. In the liquid phase, molecules can flow freely from position
In the liquid phase, molecules can flow freely from position to position by sliding over one another. A liquid takes the shape of its container. In the liquid phase, molecules can flow freely from position
In the liquid phase, molecules can flow freely from position. another. A liquid takes the shape of its container. 19.
 In the liquid phase, molecules can flow freely from position to position by sliding over one another. A liquid takes the shape of its container. In the liquid phase, molecules can flow freely from position
In the liquid phase, molecules can flow freely from position to position by sliding over one another. A liquid takes the shape of its container. In the liquid phase, molecules can flow freely from position
Advanced Nitrox Exam
 A Guide to Advanced Nitrox: The Full Spectrum of Nitrox Mixtures Advanced Nitrox Exam Student s Name Date Instructor s Name Score... 1. To develop a technical mindset a diver must base their limits for
A Guide to Advanced Nitrox: The Full Spectrum of Nitrox Mixtures Advanced Nitrox Exam Student s Name Date Instructor s Name Score... 1. To develop a technical mindset a diver must base their limits for
Chapter 9. Forces and Fluids
 Chapter 9 Forces and Fluids Key Terms hydraulic systems incompressible mass neutral buoyancy pascal pneumatic systems pressure unbalanced forces weight Archimedes principle average density balanced forces
Chapter 9 Forces and Fluids Key Terms hydraulic systems incompressible mass neutral buoyancy pascal pneumatic systems pressure unbalanced forces weight Archimedes principle average density balanced forces
Science 8 Chapter 9 Section 1
 Science 8 Chapter 9 Section 1 Forces and Buoyancy (pp. 334-347) Forces Force: anything that causes a change in the motion of an object; a push or pull on an object balanced forces: the condition in which
Science 8 Chapter 9 Section 1 Forces and Buoyancy (pp. 334-347) Forces Force: anything that causes a change in the motion of an object; a push or pull on an object balanced forces: the condition in which
Fluids always move from high pressure to low pressure. Air molecules pulled by gravity = atmospheric pressure
 9.1 Fluids Under Pressure Fluids always move from high pressure to low pressure w Fluids under pressure and compressed gases are used for a variety of everyday tasks Air molecules pulled by gravity = atmospheric
9.1 Fluids Under Pressure Fluids always move from high pressure to low pressure w Fluids under pressure and compressed gases are used for a variety of everyday tasks Air molecules pulled by gravity = atmospheric
Example A: A 400-N force is applied to a tabletop over a square area with side-length L = 20-cm.
 Physics 17 Part H Fluids and Pressure Uni-Directional Pressure The pressure that is applied over an area is the force applied, divided by the area. A 400-N force is applied to a tabletop over a square
Physics 17 Part H Fluids and Pressure Uni-Directional Pressure The pressure that is applied over an area is the force applied, divided by the area. A 400-N force is applied to a tabletop over a square
Unit 1 Lesson 5 Fluids and Pressure. Copyright Houghton Mifflin Harcourt Publishing Company
 Feel the Pressure! What are fluids? A fluid is any material that can flow and that takes the shape of its container. A fluid can flow because its particles easily move past each other. Liquids and gases,
Feel the Pressure! What are fluids? A fluid is any material that can flow and that takes the shape of its container. A fluid can flow because its particles easily move past each other. Liquids and gases,
From and
 From http://www.school-for-champions.com/science/fluidpressure.htm and http://www.school-forchampions.com/science/fluidfloating.htm by Ron Kurtus, School for Champions Pressure in Fluids by Ron Kurtus
From http://www.school-for-champions.com/science/fluidpressure.htm and http://www.school-forchampions.com/science/fluidfloating.htm by Ron Kurtus, School for Champions Pressure in Fluids by Ron Kurtus
Open Water 20 - Physics
 Open Water 20 - Physics www.diveraid.com PHYSICS Open Water 20 - Physics INTRODUCTION... 2 SECTION OBJECTIVES... 2 BUOYANCY... 2 PRESSURE, VOLUME AND DENSITY RELATIONSHIPS... 5 GAS MATTERS... 12 PARTIAL
Open Water 20 - Physics www.diveraid.com PHYSICS Open Water 20 - Physics INTRODUCTION... 2 SECTION OBJECTIVES... 2 BUOYANCY... 2 PRESSURE, VOLUME AND DENSITY RELATIONSHIPS... 5 GAS MATTERS... 12 PARTIAL
3) Water dissipates body heat about times faster than air. a) 4 b) 7 c) 20 d) 200
 PHYSICS 1) If you raise the pressure of a gas in contact with a liquid - a) Gas bubbles form b) The liquid dissolves in the gas c) The gas dissolves into the liquid d) The liquid will evaporate 2) Objects
PHYSICS 1) If you raise the pressure of a gas in contact with a liquid - a) Gas bubbles form b) The liquid dissolves in the gas c) The gas dissolves into the liquid d) The liquid will evaporate 2) Objects
Chapter 10 Fluids. Which has a greater density? Ch 10: Problem 5. Ch 10: Problem Phases of Matter Density and Specific Gravity
 Chapter 10 Fluids 10-1 Phases of Matter The three common phases of matter are solid, liquid, and gas. A solid has a definite shape and size. A liquid has a fixed volume but can be any shape. A gas can
Chapter 10 Fluids 10-1 Phases of Matter The three common phases of matter are solid, liquid, and gas. A solid has a definite shape and size. A liquid has a fixed volume but can be any shape. A gas can
How to Calculate Your SAC Rate
 How to Calculate Your SAC Rate... and Why You Should Care Scott Grizzard University of South Florida Department of Mathematics October 7, 2015 Motivating Questions I want to ascend safely, making a three
How to Calculate Your SAC Rate... and Why You Should Care Scott Grizzard University of South Florida Department of Mathematics October 7, 2015 Motivating Questions I want to ascend safely, making a three
PHYS 101 Previous Exam Problems
 PHYS 101 Previous Exam Problems CHAPTER 14 Fluids Fluids at rest pressure vs. depth Pascal s principle Archimedes s principle Buoynat forces Fluids in motion: Continuity & Bernoulli equations 1. How deep
PHYS 101 Previous Exam Problems CHAPTER 14 Fluids Fluids at rest pressure vs. depth Pascal s principle Archimedes s principle Buoynat forces Fluids in motion: Continuity & Bernoulli equations 1. How deep
Dec 6 3:08 PM. Density. Over the last two periods we discussed/observed the concept of density. What have we learned?
 Over the last two periods we discussed/observed the concept of density. What have we learned? is a ratio of mass to volume describes how much matter is packed into a space is a property of both solids
Over the last two periods we discussed/observed the concept of density. What have we learned? is a ratio of mass to volume describes how much matter is packed into a space is a property of both solids
Grade 8 Science: Unit 2-Fluids Chapter 9: Force, Pressure Area
 Grade 8 Science: Unit 2-Fluids Chapter 9: Force, Pressure Area Key Terms: hydraulic systems, incompressible, mass, neutral buoyancy, pascal, pneumatic systems, pressure, unbalanced forces, weight, Archimedes
Grade 8 Science: Unit 2-Fluids Chapter 9: Force, Pressure Area Key Terms: hydraulic systems, incompressible, mass, neutral buoyancy, pascal, pneumatic systems, pressure, unbalanced forces, weight, Archimedes
Planning Decompression Diving Gas Requirements in Open Water
 Planning Decompression Diving Gas Requirements in Open Water 1 Background During our Open Water classes we were taught that we should be on the surface with an amount of gas remaining, usually 35-50bar.
Planning Decompression Diving Gas Requirements in Open Water 1 Background During our Open Water classes we were taught that we should be on the surface with an amount of gas remaining, usually 35-50bar.
Fluids: Floating & Flying. Student Leaning Objectives 2/16/2016. Distinguish between force and pressure. Recall factors that allow floating
 Fluids: Floating & Flying (Chapter 3) Student Leaning Objectives Distinguish between force and pressure Recall factors that allow floating Differentiate between cohesion and adhesion Analyze Pascal s principle
Fluids: Floating & Flying (Chapter 3) Student Leaning Objectives Distinguish between force and pressure Recall factors that allow floating Differentiate between cohesion and adhesion Analyze Pascal s principle
7. TDI Advanced Wreck Diver Course
 7. TDI Advanced Wreck Diver Course 7.1 Introduction This course provides training and experience to competently conduct advanced wreck dives. This program includes penetration skills and techniques. Depths
7. TDI Advanced Wreck Diver Course 7.1 Introduction This course provides training and experience to competently conduct advanced wreck dives. This program includes penetration skills and techniques. Depths
Second Midterm Exam. Physics General Physics Lecture 20 - Fluids 11/9/2016. Fall 2016 Semester Prof. Matthew Jones
 Physics 22000 General Physics Lecture 20 - Fluids Fall 2016 Semester Prof. Matthew Jones 1 Second Midterm Exam Wednesday, November 16 th, 8:00-9:30 pm Location: Elliot Hall of Music -ELLT 116. Covering
Physics 22000 General Physics Lecture 20 - Fluids Fall 2016 Semester Prof. Matthew Jones 1 Second Midterm Exam Wednesday, November 16 th, 8:00-9:30 pm Location: Elliot Hall of Music -ELLT 116. Covering
3. How many kilograms of air is in the room?
 1. Astronomers use density as a clue to the composition of distant objects. Judging by the orbits of its moons the mass of Saturn is found to be 5.68 10 26 kg. (a) Use its mean radius 58 230 km to determine
1. Astronomers use density as a clue to the composition of distant objects. Judging by the orbits of its moons the mass of Saturn is found to be 5.68 10 26 kg. (a) Use its mean radius 58 230 km to determine
1. All fluids are: A. gases B. liquids C. gases or liquids D. non-metallic E. transparent ans: C
 Chapter 14: FLUIDS 1 All fluids are: A gases B liquids C gases or liquids D non-metallic E transparent 2 Gases may be distinguished from other forms of matter by their: A lack of color B small atomic weights
Chapter 14: FLUIDS 1 All fluids are: A gases B liquids C gases or liquids D non-metallic E transparent 2 Gases may be distinguished from other forms of matter by their: A lack of color B small atomic weights
Free Diving. During Constant Buoyancy Control Diving the athlete reaches the maximum depth and returns to the surface just by muscular strength.
 Ocean 11 SCUBA Free Diving For the pre-20th century Greek sponge-divers, equalizing of the ears was not necessary. These freedivers had burst their eardrums since early childhood, on purpose, through diving
Ocean 11 SCUBA Free Diving For the pre-20th century Greek sponge-divers, equalizing of the ears was not necessary. These freedivers had burst their eardrums since early childhood, on purpose, through diving
CHAPTER 31 IDEAL GAS LAWS
 CHAPTER 31 IDEAL GAS LAWS EXERCISE 144, Page 317 1. The pressure of a mass of gas is increased from 150 kpa to 750 kpa at constant temperature. Determine the final volume of the gas, if its initial volume
CHAPTER 31 IDEAL GAS LAWS EXERCISE 144, Page 317 1. The pressure of a mass of gas is increased from 150 kpa to 750 kpa at constant temperature. Determine the final volume of the gas, if its initial volume
Simulating Microgravity with Buoyancy A Space School Lesson Plan
 ASTRONAUT TRAINING...UNDERWATER Simulating Microgravity with Buoyancy A Space School Lesson Plan by Bill Andrake, Swampscott Middle School Swampscott, Massachusetts Science Lesson: Buoyancy - Based on
ASTRONAUT TRAINING...UNDERWATER Simulating Microgravity with Buoyancy A Space School Lesson Plan by Bill Andrake, Swampscott Middle School Swampscott, Massachusetts Science Lesson: Buoyancy - Based on
Force Pressure = Area
 Topics Pressure Liquids Buoyancy Archimedes Principle Flotation Pascal;s Principle Surface Tension Capillarity Pressure Force Pressure = Area Which has the greatest pressure? Units: N/m 2 -- named the
Topics Pressure Liquids Buoyancy Archimedes Principle Flotation Pascal;s Principle Surface Tension Capillarity Pressure Force Pressure = Area Which has the greatest pressure? Units: N/m 2 -- named the
mass of container full of air = g mass of container with extra air = g volume of air released = cm 3
 1992 Q32 The air pressure inside the passenger cabin of an airliner is 9 x 10 4 Pa when the airliner is at its cruising height. The pressure of the outside atmosphere at this height is 4 x 10 4 Pa. Calculate
1992 Q32 The air pressure inside the passenger cabin of an airliner is 9 x 10 4 Pa when the airliner is at its cruising height. The pressure of the outside atmosphere at this height is 4 x 10 4 Pa. Calculate
Density. Chapters 12-14: Phases of Matter. Example: Density. Conceptual Check. Springs 2/27/12. Mass Density vs. Weight Density
 Chapters 12-14: Phases of Matter Density Sequence of increasing molecule motion (and kinetic energy) Solid Liquid Gas The densities of most liquids and solids vary slightly with changes in temperature
Chapters 12-14: Phases of Matter Density Sequence of increasing molecule motion (and kinetic energy) Solid Liquid Gas The densities of most liquids and solids vary slightly with changes in temperature
Team Blue Immersion. TecRec Overview. Technical Instructor Course
 Team Blue Immersion TecRec 40-45-50 Overview Technical Instructor Course Contents DSAT Tec 40, 45 & 50 DSAT Tec 40, 45 & 50 Standards... 1 Discover Tec Prerequisites... 1 Tec 40 Prerequisites... 1 Tec
Team Blue Immersion TecRec 40-45-50 Overview Technical Instructor Course Contents DSAT Tec 40, 45 & 50 DSAT Tec 40, 45 & 50 Standards... 1 Discover Tec Prerequisites... 1 Tec 40 Prerequisites... 1 Tec
Quiz name: Chapter 13 Test Review - Fluids
 Name: Quiz name: Chapter 13 Test Review - Fluids Date: 1. All fluids are A gases B liquids C gasses or liquids D non-metallic E transparent 2. 1 Pa is A 1 N/m B 1 m/n C 1 kg/(m s) D 1 kg/(m s 2 ) E 1 N/m
Name: Quiz name: Chapter 13 Test Review - Fluids Date: 1. All fluids are A gases B liquids C gasses or liquids D non-metallic E transparent 2. 1 Pa is A 1 N/m B 1 m/n C 1 kg/(m s) D 1 kg/(m s 2 ) E 1 N/m
Density and Specific Gravity
 Fluids Phases of Matter Matter is anything that has mass and takes up space (volume). The three common phases of matter are solid, liquid, and gas. A solid has a definite shape and size. A liquid has a
Fluids Phases of Matter Matter is anything that has mass and takes up space (volume). The three common phases of matter are solid, liquid, and gas. A solid has a definite shape and size. A liquid has a
Try Scuba Diving. Try Scuba Diving. Welcome. Introductions. Program Overview. Paperwork. Academic Session. Pool/Confined Water Session
 Try Scuba Diving Welcome Introductions Program Overview Try Scuba Diving Academic Session Pool/Confined Water Session Open Water Session What You Can and Cannot Do Try Scuba Diving is NOT a Scuba Certification
Try Scuba Diving Welcome Introductions Program Overview Try Scuba Diving Academic Session Pool/Confined Water Session Open Water Session What You Can and Cannot Do Try Scuba Diving is NOT a Scuba Certification
Notes Chapter 3. Buoyancy
 Notes Chapter 3 Buoyancy Pressure in a Fluid 3.2 Pressure and the Buoyant Forces Liquids and gases are fluids materials that can flow and have no definite shape. Objects in a fluid experience a buoyant
Notes Chapter 3 Buoyancy Pressure in a Fluid 3.2 Pressure and the Buoyant Forces Liquids and gases are fluids materials that can flow and have no definite shape. Objects in a fluid experience a buoyant
Density, Pressure Learning Outcomes
 Density, Pressure Learning Outcomes Define density and pressure, and give their units. Solve problems about density and pressure. Discuss pressure in liquids and gases. State Boyle s Law. Demonstrate atmospheric
Density, Pressure Learning Outcomes Define density and pressure, and give their units. Solve problems about density and pressure. Discuss pressure in liquids and gases. State Boyle s Law. Demonstrate atmospheric
Page 1
 Contents: 1. Thrust and Pressure 2. Pressure in Fluids 3. Buoyancy 4. Why objects sink or Float when placed on surface of water? 5. Archimedes Principle 6. Relative Density Learning Objectives: The students
Contents: 1. Thrust and Pressure 2. Pressure in Fluids 3. Buoyancy 4. Why objects sink or Float when placed on surface of water? 5. Archimedes Principle 6. Relative Density Learning Objectives: The students
Key Terms Chapter 7. boiling boiling point change of state concentration condensation deposition evaporation flow rate fluid freezing point
 Foldable Activity Using the instructions on page 267 in your textbook on how to make foldables, write a key term on each front tab, and the definition on the inside (see example that I made up). You will
Foldable Activity Using the instructions on page 267 in your textbook on how to make foldables, write a key term on each front tab, and the definition on the inside (see example that I made up). You will
Density and Buoyancy Notes
 Density and Buoyancy Notes Measuring Mass and Volume 3.1 Density A balance can be used to measure the mass of an object. If the object is a liquid, pour it into a graduated cylinder to measure the volume.
Density and Buoyancy Notes Measuring Mass and Volume 3.1 Density A balance can be used to measure the mass of an object. If the object is a liquid, pour it into a graduated cylinder to measure the volume.
Archimedes Principle
 Archimedes Principle Objective In this experiment you will verify that the buoyant force on an object submerged in water is equal to the weight of the water displaced by the object. Apparatus Triple-beam
Archimedes Principle Objective In this experiment you will verify that the buoyant force on an object submerged in water is equal to the weight of the water displaced by the object. Apparatus Triple-beam
Name Class Date. (pp ) Write the letter of the correct answer in the space provided.
 Skills Worksheet Directed Reading A Section: Buoyancy and Density (pp. 412 419) 1. What is the upward force that fluids exert on all matter called? a. pascal force b. atmospheric pressure c. buoyant force
Skills Worksheet Directed Reading A Section: Buoyancy and Density (pp. 412 419) 1. What is the upward force that fluids exert on all matter called? a. pascal force b. atmospheric pressure c. buoyant force
9. Extended Range Instructor
 9. Extended Range Instructor 9.1 Introduction This course provides the training and experience required to be certified to competently teach air dives to 55 metres / 180 feet that require staged decompression,
9. Extended Range Instructor 9.1 Introduction This course provides the training and experience required to be certified to competently teach air dives to 55 metres / 180 feet that require staged decompression,
Fluids. James H Dann, Ph.D. Say Thanks to the Authors Click (No sign in required)
 Fluids James H Dann, Ph.D. Say Thanks to the Authors Click http://www.ck12.org/saythanks (No sign in required) To access a customizable version of this book, as well as other interactive content, visit
Fluids James H Dann, Ph.D. Say Thanks to the Authors Click http://www.ck12.org/saythanks (No sign in required) To access a customizable version of this book, as well as other interactive content, visit
Chapter 13 Fluids. Copyright 2009 Pearson Education, Inc.
 Chapter 13 Fluids Phases of Matter Density and Specific Gravity Pressure in Fluids Atmospheric Pressure and Gauge Pressure Pascal s Principle Units of Chapter 13 Measurement of Pressure; Gauges and the
Chapter 13 Fluids Phases of Matter Density and Specific Gravity Pressure in Fluids Atmospheric Pressure and Gauge Pressure Pascal s Principle Units of Chapter 13 Measurement of Pressure; Gauges and the
1. TDI Helitrox Diver Course
 1. TDI Helitrox Diver Course 1.1 Introduction The Helitrox course examines the theory, methods and procedures for planned stage decompression diving utilizing Helium in the breathing mixture. This program
1. TDI Helitrox Diver Course 1.1 Introduction The Helitrox course examines the theory, methods and procedures for planned stage decompression diving utilizing Helium in the breathing mixture. This program
Matter is made up of particles which are in continual random motion Misconception: Only when a substance is in its liquid or gas state do its
 Kinetic Theory of Matter Matter is made up of particles which are in continual random motion Misconception: Only when a substance is in its liquid or gas state do its particles move because in these two
Kinetic Theory of Matter Matter is made up of particles which are in continual random motion Misconception: Only when a substance is in its liquid or gas state do its particles move because in these two
Vacuum P=0. h=76 cm A B C. Barometer
 Recap: Pressure Pressure = Force per unit area (P = F /A; units: Pascals) Density of object = mass / volume (ρ = m /V; units: kg / m 3 ) Pascal s Law:Pressure is transmitted equally in all directions throughout
Recap: Pressure Pressure = Force per unit area (P = F /A; units: Pascals) Density of object = mass / volume (ρ = m /V; units: kg / m 3 ) Pascal s Law:Pressure is transmitted equally in all directions throughout
Any active TDI Helitrox Instructor who has been approved by TDI Headquarters Training Department
 10. Helitrox Diver 10.1 Introduction The Helitrox course examines the theory, methods and procedures for planned stage decompression diving utilizing Helium in the breathing mixture. This program is designed
10. Helitrox Diver 10.1 Introduction The Helitrox course examines the theory, methods and procedures for planned stage decompression diving utilizing Helium in the breathing mixture. This program is designed
Phys101 Lectures Fluids I. Key points: Pressure and Pascal s Principle Buoyancy and Archimedes Principle. Ref: 10-1,2,3,4,5,6,7.
 Phys101 Lectures 21-22 Fluids I Key points: Pressure and Pascal s Principle Buoyancy and Archimedes Principle Ref: 10-1,2,3,4,5,6,7. Page 1 10-1 Phases of Matter The three common phases of matter are solid,
Phys101 Lectures 21-22 Fluids I Key points: Pressure and Pascal s Principle Buoyancy and Archimedes Principle Ref: 10-1,2,3,4,5,6,7. Page 1 10-1 Phases of Matter The three common phases of matter are solid,
3. Moments and Pressure
 Leaving Cert Physics Long Questions 2017-2002 3. Moments and Pressure Remember to photocopy 4 pages onto 1 sheet by going A3 A4 and using back to back on the photocopier Contents Moments: ordinary level
Leaving Cert Physics Long Questions 2017-2002 3. Moments and Pressure Remember to photocopy 4 pages onto 1 sheet by going A3 A4 and using back to back on the photocopier Contents Moments: ordinary level
*See Supervised Diver Upgrade procedure #6.12 for certification requirements.
 6. Supervised Diver 6.1 Introduction This course is designed to give students the necessary skills to conduct open water dives in conditions similar to their training under the direct supervision of a
6. Supervised Diver 6.1 Introduction This course is designed to give students the necessary skills to conduct open water dives in conditions similar to their training under the direct supervision of a
Float a Big Stick. To investigate how objects float by analyzing forces acting on a floating stick
 Chapter 19: Liquids Flotation 53 Float a Big Stick Purpose To investigate how objects float by analyzing forces acting on a floating stick Required Equipment/Supplies Experiment vernier calipers 250-mL
Chapter 19: Liquids Flotation 53 Float a Big Stick Purpose To investigate how objects float by analyzing forces acting on a floating stick Required Equipment/Supplies Experiment vernier calipers 250-mL
10. TDI Advanced Trimix Instructor Course
 10. TDI Advanced Trimix Instructor Course 10.1Introduction The TDI Advanced Trimix Instructor Course provides the training required to competently and safely teach breathing gases containing helium for
10. TDI Advanced Trimix Instructor Course 10.1Introduction The TDI Advanced Trimix Instructor Course provides the training required to competently and safely teach breathing gases containing helium for
7. Open Water Scuba Diver
 7. Open Water Scuba Diver 7.1 Introduction This certification course is designed to give students the necessary skills to conduct open water dives in conditions similar to their training without the direct
7. Open Water Scuba Diver 7.1 Introduction This certification course is designed to give students the necessary skills to conduct open water dives in conditions similar to their training without the direct
8. TDI Trimix Diver Course
 8. TDI Trimix Diver Course 8.1 Introduction The TDI Trimix Course provides the training required to competently and safely utilize breathing gases containing helium for dives that require staged decompression,
8. TDI Trimix Diver Course 8.1 Introduction The TDI Trimix Course provides the training required to competently and safely utilize breathing gases containing helium for dives that require staged decompression,
Density, Pressure Learning Outcomes
 1 Density, Pressure Learning Outcomes Define density and pressure, and give their units. Solve problems about density and pressure. Discuss pressure in liquids and gases. State Boyle s Law. Demonstrate
1 Density, Pressure Learning Outcomes Define density and pressure, and give their units. Solve problems about density and pressure. Discuss pressure in liquids and gases. State Boyle s Law. Demonstrate
Exercises The Atmosphere (page 383) 20.2 Atmospheric Pressure (pages )
 Exercises 20.1 The Atmosphere (page 383) 1. The energizes the molecules in Earth s atmosphere. 2. Why is gravity important to Earth s atmosphere? 3. What would happen to Earth s atmosphere without the
Exercises 20.1 The Atmosphere (page 383) 1. The energizes the molecules in Earth s atmosphere. 2. Why is gravity important to Earth s atmosphere? 3. What would happen to Earth s atmosphere without the
Slide 1 / What is the density of an aluminum block with a mass of 4050 kg and volume of 1.5 m 3?
 Slide 1 / 68 1 What is the density of an aluminum block with a mass of 4050 kg and volume of 1.5 m 3? Slide 2 / 68 2 What is the mass of a rectangular shaped ice block with dimensions of 0.04m x 0.05m
Slide 1 / 68 1 What is the density of an aluminum block with a mass of 4050 kg and volume of 1.5 m 3? Slide 2 / 68 2 What is the mass of a rectangular shaped ice block with dimensions of 0.04m x 0.05m
3. Intro to Tech. 3.1 Introduction. 3.2 Qualifications of Graduates. 3.3 Who May Teach. 3.4 Student to Instructor Ratio
 3. Intro to Tech 3.1 Introduction The TDI Intro to Tech course introduces students to the world of technical diving. This course is designed as an introductory course to the TDI Advanced Nitrox course
3. Intro to Tech 3.1 Introduction The TDI Intro to Tech course introduces students to the world of technical diving. This course is designed as an introductory course to the TDI Advanced Nitrox course
4. Advanced Adventure Diver
 4. Advanced Adventure Diver 4.1 Introduction The purpose of this course is to give the diver an overview of 5 different specialties, 2 core, and 3 additional SDI Specialties. The two core specialties are,
4. Advanced Adventure Diver 4.1 Introduction The purpose of this course is to give the diver an overview of 5 different specialties, 2 core, and 3 additional SDI Specialties. The two core specialties are,
Pressure is defined as force per unit area. Any fluid can exert a force
 Physics Notes Chapter 9 Fluid Mechanics Fluids Fluids are materials that flow, which include both liquids and gases. Liquids have a definite volume but gases do not. In our analysis of fluids it is necessary
Physics Notes Chapter 9 Fluid Mechanics Fluids Fluids are materials that flow, which include both liquids and gases. Liquids have a definite volume but gases do not. In our analysis of fluids it is necessary
Additional Information
 Buoyancy Additional Information Any object, fully or partially immersed in a fluid, is buoyed up by a force equal to the weight of the fluid displaced by the object. Archimedes of Syracuse Archimedes principle
Buoyancy Additional Information Any object, fully or partially immersed in a fluid, is buoyed up by a force equal to the weight of the fluid displaced by the object. Archimedes of Syracuse Archimedes principle
Student Exploration: Archimedes Principle
 Name: Date: Student Exploration: Archimedes Principle Vocabulary: Archimedes principle, buoyant force, density, displace, mass, volume, weight Prior Knowledge Questions (Do these BEFORE using the Gizmo.)
Name: Date: Student Exploration: Archimedes Principle Vocabulary: Archimedes principle, buoyant force, density, displace, mass, volume, weight Prior Knowledge Questions (Do these BEFORE using the Gizmo.)
Diving at Altitude. Part One: Starting with Residual Nitrogen. By John Adsit January 30, 2018
 Diving at Altitude By John Adsit January 30, 2018 Diving at altitude demands adjustments to the procedures used at sea level. With moderate increases in altitude and with shallower dives, the differences
Diving at Altitude By John Adsit January 30, 2018 Diving at altitude demands adjustments to the procedures used at sea level. With moderate increases in altitude and with shallower dives, the differences
20 Gases. Gas molecules are far apart and can move freely between collisions.
 Gas molecules are far apart and can move freely between collisions. Gases are similar to liquids in that they flow; hence both are called fluids. In a gas, the molecules are far apart, allowing them to
Gas molecules are far apart and can move freely between collisions. Gases are similar to liquids in that they flow; hence both are called fluids. In a gas, the molecules are far apart, allowing them to
Exam Review Mass, Weight, Density, Buoyancy, States of Matter
 Exam Review Mass, Weight, Density, Buoyancy, States of Matter Volume An object s volume is the amount of space it takes up. The volume of a cup of water can change if you freeze it in to a solid or boil
Exam Review Mass, Weight, Density, Buoyancy, States of Matter Volume An object s volume is the amount of space it takes up. The volume of a cup of water can change if you freeze it in to a solid or boil
. In an elevator accelerating upward (A) both the elevator accelerating upward (B) the first is equations are valid
 IIT JEE Achiever 2014 Ist Year Physics-2: Worksheet-1 Date: 2014-06-26 Hydrostatics 1. A liquid can easily change its shape but a solid cannot because (A) the density of a liquid is smaller than that of
IIT JEE Achiever 2014 Ist Year Physics-2: Worksheet-1 Date: 2014-06-26 Hydrostatics 1. A liquid can easily change its shape but a solid cannot because (A) the density of a liquid is smaller than that of
24. Basic Closed Circuit Rebreather Decompression Procedures Instructor - Unit Specific
 24. Basic Closed Circuit Rebreather Decompression Procedures Instructor - Unit Specific 24.1 Introduction This is the instructor level certification course for instructors wishing to teach the unit specific
24. Basic Closed Circuit Rebreather Decompression Procedures Instructor - Unit Specific 24.1 Introduction This is the instructor level certification course for instructors wishing to teach the unit specific
Gas molecules are far apart. collisions The Atmosphere
 Gas molecules are far apart and can move freely between collisions. Gases are similar to liquids in that they flow; hence both are called fluids. In a gas, the molecules are far apart, allowing them to
Gas molecules are far apart and can move freely between collisions. Gases are similar to liquids in that they flow; hence both are called fluids. In a gas, the molecules are far apart, allowing them to
Shark Biology Buoyancy by Bill Andrake
 Shark Biology Buoyancy by Bill Andrake Science Lesson: Buoyancy - Based on Webisode 45 - Shark Biology Grade Level: 6-8 Time: Four (45-50 minute) class periods Introduction Jonathan narrates an educational
Shark Biology Buoyancy by Bill Andrake Science Lesson: Buoyancy - Based on Webisode 45 - Shark Biology Grade Level: 6-8 Time: Four (45-50 minute) class periods Introduction Jonathan narrates an educational
Chapter 9 Fluids and Buoyant Force
 Chapter 9 Fluids and Buoyant Force In Physics, liquids and gases are collectively called fluids. 3/0/018 8:56 AM 1 Fluids and Buoyant Force Formula for Mass Density density mass volume m V water 1000 kg
Chapter 9 Fluids and Buoyant Force In Physics, liquids and gases are collectively called fluids. 3/0/018 8:56 AM 1 Fluids and Buoyant Force Formula for Mass Density density mass volume m V water 1000 kg
Properties of Air. Air Takes Up Space
 Properties of Air Air Takes Up Space Take an empty ziploc bag, open it and pull it through the air like a parachute. Now close it, seal it and try to squish the bag. There s nothing in the bag, right?
Properties of Air Air Takes Up Space Take an empty ziploc bag, open it and pull it through the air like a parachute. Now close it, seal it and try to squish the bag. There s nothing in the bag, right?
Intro to Tech Instructor
 Intro to Tech Instructor 1 Introduction The TDI Intro to Tech Instructor course provides the training required to competently and safely introduce students to the world of technical diving. The objective
Intro to Tech Instructor 1 Introduction The TDI Intro to Tech Instructor course provides the training required to competently and safely introduce students to the world of technical diving. The objective
Chapter 5 Decompression, Dive Tables, and Dive Computers
 Chapter 5 Decompression, Dive Tables, and Dive Computers Nitrogen and Diving 1. If the partial pressure of nitrogen in the air you are breathing is greater than the pressure of nitrogen in your body, your
Chapter 5 Decompression, Dive Tables, and Dive Computers Nitrogen and Diving 1. If the partial pressure of nitrogen in the air you are breathing is greater than the pressure of nitrogen in your body, your
Unit A: Mix and Flow of Matter
 Unit A: Mix and Flow of Matter Science 8 1 Section 3.0 THE PROPERTIES OF GASES AND LIQUIDS CAN BE EXPLAINED BY THE PARTICLE MODEL OF MATTER. 2 1 Viscosity and the Effects of Temperature Topic 3.1 3 Viscosity
Unit A: Mix and Flow of Matter Science 8 1 Section 3.0 THE PROPERTIES OF GASES AND LIQUIDS CAN BE EXPLAINED BY THE PARTICLE MODEL OF MATTER. 2 1 Viscosity and the Effects of Temperature Topic 3.1 3 Viscosity
Up Up and Away on Beautiful Balloons: Scaling up from Party Favors to Scientific Payloads
 Up Up and Away on Beautiful Balloons: Scaling up from Party Favors to Scientific Payloads How many helium filled balloons would it take to lift you? To lift a big telescope? Take a guess for each and record
Up Up and Away on Beautiful Balloons: Scaling up from Party Favors to Scientific Payloads How many helium filled balloons would it take to lift you? To lift a big telescope? Take a guess for each and record
Hydrostatics. Physics 1425 Lecture 25. Michael Fowler, UVa
 Hydrostatics Physics 1425 Lecture 25 Michael Fowler, UVa Basic Concepts Density Pressure: Pascal s Principle The Crown and the Bathtub Around 250 BC, the king of Syracuse commissioned a new crown,and gave
Hydrostatics Physics 1425 Lecture 25 Michael Fowler, UVa Basic Concepts Density Pressure: Pascal s Principle The Crown and the Bathtub Around 250 BC, the king of Syracuse commissioned a new crown,and gave
Independent Study Assignments. Tec 40. Tec 40 Knowledge Development One. Instructor Guide Appendix
 Instructor Guide Appendix Independent Study Assignments Tec 40 Tec 40 Knowledge Development One Study assignment: Tec Deep Diver Manual, pgs xi, pg xiii Your Obligations and Responsibilities, pg xiv Diver
Instructor Guide Appendix Independent Study Assignments Tec 40 Tec 40 Knowledge Development One Study assignment: Tec Deep Diver Manual, pgs xi, pg xiii Your Obligations and Responsibilities, pg xiv Diver
1 Fluids and Pressure
 CHAPTER 3 1 Fluids and Pressure SECTION Forces in Fluids BEFORE YOU READ After you read this section, you should be able to answer these questions: What are fluids? What is atmospheric pressure? What is
CHAPTER 3 1 Fluids and Pressure SECTION Forces in Fluids BEFORE YOU READ After you read this section, you should be able to answer these questions: What are fluids? What is atmospheric pressure? What is
Buoyancy and Density. Buoyant Force and Fluid Pressure. Key Concept Buoyant force and density affect whether an object will float or sink in a fluid.
 2 Buoyancy and Density Key Concept Buoyant force and density affect whether an object will float or sink in a fluid. What You Will Learn All fluids exert an upward buoyant force on objects in the fluid.
2 Buoyancy and Density Key Concept Buoyant force and density affect whether an object will float or sink in a fluid. What You Will Learn All fluids exert an upward buoyant force on objects in the fluid.
Activity 4 Buoyancy in a Liquid /Archimedes' Principle F1003 Physics II ITESM Campus Aguascalientes January-May 2017 Dr. Juan-Manuel CAMPOS-SANDOVAL
 Activity 4 Buoyancy in a Liquid /Archimedes' Principle F1003 Physics II ITESM Campus Aguascalientes January-May 2017 Dr. Juan-Manuel CAMPOS-SANDOVAL Name MULTIPLE CHOICE. Choose the one alternative that
Activity 4 Buoyancy in a Liquid /Archimedes' Principle F1003 Physics II ITESM Campus Aguascalientes January-May 2017 Dr. Juan-Manuel CAMPOS-SANDOVAL Name MULTIPLE CHOICE. Choose the one alternative that
AP Physics B Ch 10 Fluids. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Name: Period: Date: AP Physics B Ch 10 Fluids 1) The three common phases of matter are A) solid, liquid, and vapor. B) solid, plasma, and gas. C) condensate, plasma, and gas. D) solid, liquid, and gas.
Name: Period: Date: AP Physics B Ch 10 Fluids 1) The three common phases of matter are A) solid, liquid, and vapor. B) solid, plasma, and gas. C) condensate, plasma, and gas. D) solid, liquid, and gas.
Chapter 10: Properties of Gases: The Air We Breathe
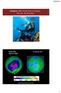 Chapter 10: Properties of Gases: The Air We Breathe South Pole Sept 24, 2006 15 February 2017 http://ozonewatch.gsfc.nasa.gov 1 Chapter Outline 10.1 10.2 10.3 The Properties of Gases Effusion and the Kinetic
Chapter 10: Properties of Gases: The Air We Breathe South Pole Sept 24, 2006 15 February 2017 http://ozonewatch.gsfc.nasa.gov 1 Chapter Outline 10.1 10.2 10.3 The Properties of Gases Effusion and the Kinetic
Clicker Question: Clicker Question: Clicker Question: Phases of Matter. Phases of Matter and Fluid Mechanics
 Newton's Correction to Kepler's First Law The orbit of a planet around the Sun has the common center of mass (instead of the Sun) at one focus. A flaw in Copernicus s model for the solar system was A:
Newton's Correction to Kepler's First Law The orbit of a planet around the Sun has the common center of mass (instead of the Sun) at one focus. A flaw in Copernicus s model for the solar system was A:
Chemistry: It s a gas
 Chemistry: It s a gas Part IV Molar mass of a gas Density of a gas Dalton s Law: high altitudes and scuba diving Kinetic Molecular Theory: fast gases are hot! I have a chemistry test on Friday, there is
Chemistry: It s a gas Part IV Molar mass of a gas Density of a gas Dalton s Law: high altitudes and scuba diving Kinetic Molecular Theory: fast gases are hot! I have a chemistry test on Friday, there is
Why do things float? Climate and Global Change. Introduction
 Why do things float? Introduction Archimedes of Syracuse (ca. 287-212 B.C.), a physical scientist, is credited with understanding two basic principles: When describing the mechanical advantage gained by
Why do things float? Introduction Archimedes of Syracuse (ca. 287-212 B.C.), a physical scientist, is credited with understanding two basic principles: When describing the mechanical advantage gained by
DENSITY AND BUOYANCY
 DENSITY AND BUOYANCY DENSITY - RECAP What is DENSITY? The amount of MASS contained in a given VOLUME Density describes how closely packed together the particles are in a substance Density Experiment SINK
DENSITY AND BUOYANCY DENSITY - RECAP What is DENSITY? The amount of MASS contained in a given VOLUME Density describes how closely packed together the particles are in a substance Density Experiment SINK
13. Advanced Trimix Diver
 13. Advanced Trimix Diver 13.1 Introduction The TDI Advanced Trimix course provides the training required to competently and safely utilize breathing gases containing helium for dives that require staged
13. Advanced Trimix Diver 13.1 Introduction The TDI Advanced Trimix course provides the training required to competently and safely utilize breathing gases containing helium for dives that require staged
Boyle s Law Practice Problems Name:
 Boyle s Law Practice Problems Name: 1. If a gas at 25.0 C occupies 3.60 L at a pressure of 1.00 atm, what will be its volume at a pressure of 2.50 atm? 2. 500.0 ml of a gas is collected at 745.0 mm Hg.
Boyle s Law Practice Problems Name: 1. If a gas at 25.0 C occupies 3.60 L at a pressure of 1.00 atm, what will be its volume at a pressure of 2.50 atm? 2. 500.0 ml of a gas is collected at 745.0 mm Hg.
4. TDI Advanced Nitrox Diver Course
 4. TDI Advanced Nitrox Diver Course 4.1 Introduction This course examines the use of EAN-21 through one hundred (100) percent oxygen for optimal mixes to a depth of forty (40) msw / one hundred thirty
4. TDI Advanced Nitrox Diver Course 4.1 Introduction This course examines the use of EAN-21 through one hundred (100) percent oxygen for optimal mixes to a depth of forty (40) msw / one hundred thirty
7. Advanced Nitrox Diver
 7. Advanced Nitrox Diver 7.1 Introduction This course examines the use of EAN-21 through 100 percent oxygen for optimal mixes to a depth of 40 metres / 130 feet. The objective of this course is to train
7. Advanced Nitrox Diver 7.1 Introduction This course examines the use of EAN-21 through 100 percent oxygen for optimal mixes to a depth of 40 metres / 130 feet. The objective of this course is to train
Chapter 10: Properties of Gases: The Air We Breathe
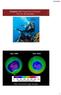 Chapter 10: Properties of Gases: The Air We Breathe Sept, 2006 Sept, 2016 http://ozonewatch.gsfc.nasa.gov 1 Chapter Outline 10.1 The Properties of Gases 10.2 The Kinetic Molecular Theory of Gases* 10.3
Chapter 10: Properties of Gases: The Air We Breathe Sept, 2006 Sept, 2016 http://ozonewatch.gsfc.nasa.gov 1 Chapter Outline 10.1 The Properties of Gases 10.2 The Kinetic Molecular Theory of Gases* 10.3
Overview. Prerequisites for the course. What will be covered throughout the course - Modules - Assembly & Pre/Post Care - Diving Skills
 Overview Prerequisites for the course What will be covered throughout the course - Modules - Assembly & Pre/Post Care - Diving Skills Requirements for Successful Completion Study Materials You Will Need
Overview Prerequisites for the course What will be covered throughout the course - Modules - Assembly & Pre/Post Care - Diving Skills Requirements for Successful Completion Study Materials You Will Need
1. Unlimited, so long as adequate facility, supplies and time are provided to ensure comprehensive and complete training of subject matter
 12. Trimix Diver 12.1 Introduction The TDI Trimix course provides the training required to competently and safely utilize breathing gases containing helium for dives that require staged decompression,
12. Trimix Diver 12.1 Introduction The TDI Trimix course provides the training required to competently and safely utilize breathing gases containing helium for dives that require staged decompression,
PRESSURE AND BUOYANCY
 PRESSURE AND BUOYANCY CONCEPT SUMMARY So far The pressure applied to a confined liquid is transmitted to every point in the liquid (Pascal's Principle). At any given point in a liquid the pressure is the
PRESSURE AND BUOYANCY CONCEPT SUMMARY So far The pressure applied to a confined liquid is transmitted to every point in the liquid (Pascal's Principle). At any given point in a liquid the pressure is the
13. Shallow Water Scuba Diver
 13. Shallow Water Scuba Diver 13.1 Introduction This entry-level certification course is designed to give students the necessary skills to safely conduct open water dives in conditions similar to their
13. Shallow Water Scuba Diver 13.1 Introduction This entry-level certification course is designed to give students the necessary skills to safely conduct open water dives in conditions similar to their
CHEM 1211 Gases-Part 1 Worksheet 4-2. Name(s):
 CHEM 1211 Gases-Part 1 Worksheet 4-2 Name(s): Pressure [1] The atmospheric pressure at the summit of Mt. McKinley is 581 mmhg on a certain day. What is the pressure in atmospheres, in atmospheres and in
CHEM 1211 Gases-Part 1 Worksheet 4-2 Name(s): Pressure [1] The atmospheric pressure at the summit of Mt. McKinley is 581 mmhg on a certain day. What is the pressure in atmospheres, in atmospheres and in
Page 1. Balance of Gravity Energy More compressed at sea level than at higher altitudes Moon has no atmosphere
 Earth s Atmosphere Gases and Plasmas Balance of Gravity Energy More compressed at sea level than at higher altitudes Moon has no atmosphere Magdeburg Hemispheres Weight of Air mass of air that would occupy
Earth s Atmosphere Gases and Plasmas Balance of Gravity Energy More compressed at sea level than at higher altitudes Moon has no atmosphere Magdeburg Hemispheres Weight of Air mass of air that would occupy
Unit 14 Gas Laws Funsheets
 Name: Period: Unit 14 Gas Laws Funsheets Part A: Vocabulary and Concepts- Answer the following questions. Refer to your notes and the PowerPoint for help. 1. List 5 different common uses for gases: a.
Name: Period: Unit 14 Gas Laws Funsheets Part A: Vocabulary and Concepts- Answer the following questions. Refer to your notes and the PowerPoint for help. 1. List 5 different common uses for gases: a.
AP Lab 11.3 Archimedes Principle
 ame School Date AP Lab 11.3 Archimedes Principle Explore the Apparatus We ll use the Buoyancy Apparatus in this lab activity. Before starting this activity check to see if there is an introductory video
ame School Date AP Lab 11.3 Archimedes Principle Explore the Apparatus We ll use the Buoyancy Apparatus in this lab activity. Before starting this activity check to see if there is an introductory video
