THE METABOLISM OF THE AMIDINE GROUP OF ARGINISE IN THE INTACT RAT
|
|
|
- Regina Webb
- 6 years ago
- Views:
Transcription
1 THE METABOLISM OF THE AMIDINE GROUP OF ARGINISE IN THE INTACT RAT BY DEWITT STETTRN, Ja., ASI) BEN BLOOM (Prom the National Institute oj Brthritis and Metabolic Diseases, National Institutes of Health, Public Health SertCe, Bethesda, Maryland) (Received for publication, October, 955) The present study is concerned with the metabolism of arginine in the intact rat. The amidine group of the arginine employed was labeled with two isotopes, N5 and C4. It was hoped that by the device of double labeling information beyond that presently available could be secured about the various fates of this amino acid. The first question about which information was sought Teas the possibility of loss and subsequent replacement of amidine nitrogen without concomitant exchange of amidine carbon. This might result, for example, from reversal of the citrulline + arginine reaction (, ) or from the action of an as yet undiscovered mammalian arginine desimidase. Such processes, if they occurred, would be expected to lower the ratio N5/C4 in the arginine of tissue proteins. The nitrogen of the amidine group of creatine is knolvn to arise from t.he amidine group of arginine (3, 4). It has not been established, however, whether in this transamidination reaction the amidine group migrates with I its skeleton (=N-C=N-) intact. Equality of the ratio N5/C4 in the amidine group of carcass creatine with the corresponding ratio in administered arginine would support the conclusion that C-N bonds within the amidine group were not ruptured incident to this transfer. Studies with arginine-amidine-n l5 (4) have yielded results in excellent agreement with the classical hypothesis of an ornithine-citrulline-arginine cycle. When urea-n5 was fed, it appeared to behave as a metabolic endproduct, little N5 being recovered in products other than urea. We know of no studies in which the fate of the amidine-carbon of arginine has been similarly followed, but several investigators have reported on the administration of urea-c4 (5, 6), and have found that a considerable fraction of this carbon is recoverable in expired COZ. The present experiments with doubly labeled arginine were designed to shed light on this apparent discrepancy.
2 74 AMIDINE GROUP OF ARGININE EXPERIMENTAL Synthesis oj 8~yilline-drnidine-Cl4,-P liyclrochloride-the synthesis of L-srginine monohydrochloride, doubly labeled in the amidine group with Cl- and Y5, followd in general the method of Murtx (7). This method involves the condcneat,ion of the caoppcr complex of ornithinc with O-methylisouronium chloride. Cyanogen bromide-u \vas prepared by addition of 4 mmoles of NaCN in a total volume of 4 ml. of H,O (8) to 40 mmoles of Brz, in a flask immersed in ice. The addition, which was made over a 4 hour period with stirring, consisted first of mmole of NaCY4N ( mc.),l followed by 40 mmoles of non-isotopic NaCN. During addition the temperature was maintained at 0 or below. The crystalline product was then distilled through a short stem into a receiver immersed in ice and fitted with a reflux condenser. All the material which could be distilled on a boiling water bath WLS collected. The crystalline distillate, which was slightly brown in color, was dissolved in 30 ml. of ether and treated with anhydrous CaClz for hour at 0. To the filtered ethereal solution of crude C? NBr was added a chilled solution containing 80 mmoles of W5H, (approximately G atom per cent excess) in 60 ml. of absolute methanol (9). A colorless solution resulted which, upon warming to room temperature, deposited crystals of N5H4Br. On the following day the crystals were removed by filtration, the filtrate was evaporated to dryness in vacua, and the residue was repeatedly extracted with port ions of boiling ether. The combined ether-insoluble material, presumably IV5H4Br, weighed 3.43 gm. (88 per cent of theory). The ether-soluble HZN-C=N was recovered after removal of solvent under XX and dried in vacua over I 05. It lyeighed.30 gm. (3 mmoles). This crude cyanamide was treated with about 5 ml. of cold absolute CHsOH and.5 gm. of anhydrous HCI (0). The mixture was left at room temperature for 5 days in a flask protected by a CaC& tube. The solvent was then removed under Nz, and the residue was dried. This amounted to 3.07 gm. (8 mmoles calculated as 0-methylisouronium chloride). 30 mmoles (6.5 gm.) of L-ornithine dihydrochloride ([or]: 4.35, concentration 4 per cent) (7) were suspended in water and brought into solution by adjustment of the ph to 7.0 with N NaOH. This clear solution (about 0 ml.) was treated with an excess of basic copper carbonate, boiled for 5 minutes, filtered, and cooled. G ml. of N NaOH were then added, and t,he entire solution of copper-ornit,hine complex was transferred onto Obtained from Tracerlab, Inc. Obtained from the Eastman Kodak Company.
3 D. STETTEN, JR., AND B. BLOOM 75 the dry 0-methylisouronium chloride described above. The resulting clear dark blue solution was kept at room temperature for 7 days. The solution was then brought to ph 3.5 with HCI, and the mixture was saturated with HZS, boiled to eliminate excess HZS, and filtered. To the filtrate plus mashings were added 75 mmoles (6. gm.) of flavianic acid dissolved in 65 ml. of hot water (). Immediate crystallization occurred, and after refrigeration for 4 hours the crude arginine flavianate was collected, washed with ice water, and then with ethanol. The crude arginine flavianate (9 gm.) was decomposed by treatment on the steam bath with 45 ml. of concentrated HCl solution for hours. After refrigeration for hours, the regenerated flavianic acid was removed by filtration, and the filtrate and washings were evaporated to a syrup in vacua, dissolved in boiling water, and decolorized with charcoal. Ethanol was cautiously added to the filtrate, the ph adjusted to about 7 by addition of aniline (la), and t he resultant oily precipitate seeded and scratched. After several days of refrigeration the precipitate was collected, washed with cold ethanol, and dissolved in 0 ml. of hot water. After decolorization with charcoal, the solution was treated with ethanol, and, upon seeding, white crystals of n-arginine monohydrochloride were deposited. After refrigeration these were filtered off, washed with ethanol, and dried in vucuo, first over CaC&, then over PZOG. The final yield was 3.48 gm. (6.5 mmoles = 4 per cent of theory based on. 40 mmoles of starting materials). The nitrogen content (Kjeldahl) of the arginine monohydrochloride was 6.6 per cent (calculated 6.8 after correct,ion for 5 atom per cent excess W5). [a]?.6 7. ( concentration.6 per cent as arginine in 6 N HCl). From the accumulated mother liquors of the last stages, an additional.0 gm. of recrystallized arginine nonoflavianat.e were secured. Feeding Experiments-Male rats of the Sherman strain weighing from 80 to 0 gm. were divided into three groups. Experiments -A and -B consisted of three and two rats, respectively. The rats of Experiment -A were maintained in a metabolism cage which permitted daily collection of urine. The rats of Experiment -B were maintained in a desiccator modified to permit daily collection of expired COZ. The urines of this latter group were preserved with diphenylmercuric borate and pooled for the duration of the experimental period. The treatment of the three rats in Experiment was identical with that in Experiment -A. For 3 days prior to experimentation the rat,s were given access to a diet consisting of 8 per cent casein, 5 per cent salt (Osborne-Mendel mixture No. l), 55 per cent corn starch, 7 per cent cellulose, 5 per cent yeast, 8 per cent corn oil, and per cent cod liver oil. To minimize scattering, the diet was made into a paste with water. At the beginning of the experi-
4 76 AMIDINE GROUP OF ARGININE mental period, sufficient isotopic arginine monohydrochloride was added to the diet so that the animals received an average of.5 mmoles of arginine per kilo of rat per day. The feeding of the isotopic diet was continued for 3 days. At the end of this period t,he tissues of rats of Experiments -A and -B were combined. Urine-Ammonia nitrogen was determined and isolated for N assay by aeration after addition of KzC03 (3). nitrogen was measured by a urease method (3). The NL5 and Cl4 contents of urea were obtained by analysis of the dixanthydryl derivative (4). The nitrogen contents (Kjeldahl) of the dixanthydryl urea derivatives ranged from 6.7 to 6.9 per cent (calculated 6.7). Nucleic Acids, Arginine, Aspartate, and Glutamate-Sodium nucleates were isolated from the internal organs (kidney, liver, thymus, testis, and spleen) by extraction with hot 0 per cent NaCl solution as described by Conzelman et al. (5). The product did not contain sufficient isotope to warrant further study. The residue was defatted by extraction with alcohol and ethyl ether and was dried. Arginine monoflavianate was isolated essentially as described by Vickery () and converted to arginine monohydrochloride according to the method of Cox (). The nitrogen content of the isolated arginine monohydrochloride was 6.6 per cent, Experiment, and 6., Experiment (calculated 6.6). The mother liquor from the arginine flavianate isolation was freed of flavianic acid by neutralization with Ba(OH) solution, filtration, and further treatment with charcoal. Glutamic acid hydrochloride and cupric aspartate were isolated as described by Stetten and Schoenheimer (6). The nitrogen content of the glutamic acid hydrochloride was 7.7 per cent (calculated 7.6) and of the cupric aspartate 7.3 (calculated 7.). Creatine-After removal of the organs and skin the carcasses were minced, and creatine was isolated as creatinine picrate according to Foster et al. (7). The creatinine picrate was converted to creatinine*znclz by the method of Benedict (8). The creatinine.znc contained 3.5 per cent nitrogen, Experiment, and 3.0 per cent, Experiment (calculated 3.). Amidine-N5, -Cl-The isotope content of the amidine group of arginine and of creatine was measured on samples of NH, and COz obtained by refluxing these products with Ba(OH) as described by Bloch et al. (9). Results Recovery of Isotope in Tissue Constituents-Comparison of the concentrations of CY and N5 in the arginine fed with that recovered from internal organs (Table I) reveals an approximate loo-fold dilution to have taken place in each of the experiments. The alkaline degradation of arginine tu
5 D. STETTEN, JR., AND B. BLOOM 77 liberate the C and IV of the amidine group gave the theoretical value for N5 on synthetic arginine, but the value for Cl4 Iv-as slightly less than ex- TABLE Isotope Distribution after Feeding Alginine-Cl4 and -W5 Arginine labeled in the amidine group with Cl4 and Nl was included in the diet of three groups of rats, Experiments l-a, I-B, and, at the level of.5 mmoles per kilo of rat per day for 3 days. The rats were sacrificed, and the internal organs and carcasses from Experiments -A and -B were pooled. For further details, see the experimental section. - Arginine, fed In amidinel Arginine, organs In amidinet Arginine, organs In amidinet Glutamate, organ Aspartate ( Creatine, carcasses In amidinet Creatine, carcasses In amidinet, urine, st day ( nd < 3rd lst-3rd da) I< st day < ([ nd I < 3rd T Experin nent No. -A -B - a (a) km per cent ezcess I hrrected N S* 65) peu cent C Cc) c.p.nz. per milliatonr c 4.59 x 0.56 X X lo3.3 x lo x x X lo3.99 x X lo3.66 x x lo4.46 x lo*.03 x 04.9 x x lo4 3.8 X lo4.55 x 04 per cen * Values in columns (b) and (d) are isotope concentrations adjusted to 00 in the amidine group of arginine fed. f The term amidine refers to NH and CO liberated after treatment with boiling Ba(OH) solution. petted. When the dilutions of the two isotopes of organ arginine are compared (last column, Table I), it would appear that the amidine carbon has undergone slightly greater dilution than the amidine nitrogen. As was anticipated from the results of Bloch (4), some lf was recovered in
6 78 AMIDINE GROUP OF ARGININE the dicarboxylic amino acids of the organs, more in glutamic than in aspartic acid. Nitrogen of the amidine group of creatine is known to arise from that of arginine (4). Evidence published to date does not permit a decision as to whether the amidine group,. comprising C atom and N atoms, is transferred intact. In the present experiments (Table I), the carcass creatine isolated contained both isotopic atoms with which the ingested arginine was labeled. The concentrations of isotope in the amidine group of creatine were less than per cent of the corresponding concentrations in the amidine group of ingested arginine. In each case, however, the ratio of the two isotopes in the amidine of creatine (last column, Table I) did not deviate significantly from that in the amidine of the arginine fed. This finding supports the view that, in this transamidinat ion reaction, the amidine group migrates intact. TABLE II Elimination of Amidine Calbon As COZ The expired COZ from the rats of Experiment I-B was collected in 4 hour portions. Consumed as arginine Excreted as COn Day No - activity Specific activity activity 3 I c.pm. c.p.m. $%r milliatom c c.p.m. 6. X lo x X 0 : x x X lo5 Distribution of Isotope in Excretory Products-Of the Cl4 ingested as amidine-labeled arginine, a considerable fraction appeared in respiratory COz. The specific activity of the CO? rose with the passage of time (Table II). In the 3 day period of study, two-thirds of the ingested radioactive carbon appeared as COZ, and, on the 3rd day of feeding, actually more Cl* was exhaled as COz than was ingested on that day. The magnitude of this conversion was unexpected in view of the fact that, in the accepted pathways of mammalian met,abolism of arginine, no direct route exists from amidine carbon to COZ. Of the N5 ingested as amidine-labeled arginine (Column f, Table III), 35 to 45 per cent was recovered in the total urinary nitrogen (last column, Table III) in the 3 day period. In those experiments in which daily urine collections were made, the ammonia of the urine contained 0.4 to 4 per cent as large a quantity of N5 as did the urea. The N5 concentrations in total urinary N as well as in urea and ammonia rose on successive days. It is of interest to compare the concentrations of Cl4 and of IV5 in urinary urea with those of the arginine fed (Table I). In contrast to the behavior
7 I). S ltlw:~, JR., ASD B. BLOOM 79 of W5, the Cl concentration in urea was irregular, wit,h a tendency to decline which was most marked on the 3rd day of each experiment,. The reason for this decline, in the face of a rising Xl5 concent,ration in urea and a rising specific activity in CO (Table II), is not apparent. This behavior is reflected in the values in the last column of Table I. Here TABLE Partition of Nitrogen and Isotope Distribution in Urinary Products Experiments I-A and were conducted on three rat,s each; Experiment -B was conducted on two rats. III Consumed Urine -A -B nay so Arginine (cl?la?nole sitrogen NH, NH3 XH, NH, NH3 NH, NH, s aom per cent eaxe.ss iy,; (RI x C/L) it is seen that the ratio of isotopes (X5/C? ). in urinary urea, close to unity initially, rises to a value of 3 to 4 by the 3rd day. From this it is clear that, of the constituent atoms of urinary urea, a larger fraction of t.he N t>han of the C is derived from the amidine group of ingested arginine. )scuss0s Three findings which have become apparent in the description of the results are presumably related. These are () that a major fraction of
8 730 AMIDINE GROUP OF ARGININE the amidine carbon of arginine fed to rats appears as expired C0, () that the ratio P5/C?4 in urinary urea rises significantly above the corresponding ratio in the amidine group of ingested isotopically labeled arginine, and (3) that, in the arginine isolated from internal organs, this same ratio appears to be slightly higher than in the material ingested. Whereas no mammalian enzymes are known to account for the direct conversion of amidine carbon of arginine to CO*, there is ample evidence (9) that ureases from bacteria in the gastrointestinal tract may degrade urea, which in turn arises from the action of arginase upon arginine. Of the products of gastrointestinal urea breakdown, the ammonia would be expected to enter the portal circulation, to be returned to the liver for resynthesis into arginine, ultimately appearing in large part as urea. The carbon dioxide, on the other hand, even assuming efficient intestinal absorption, would be mixed with a large reservoir of CO prior to reentry into the urea cycle, and its isotope would be greatly diluted. It is concluded that the metabolic inertia of urea in the intact animal, based on studies with Xl5 alone, is apparent rather than real and reflects the efficiency with which ammonia arising in the gastrointestinal tract is resynthesized into urea in the liver. SUMMARY L-hrginine-amidine-IV5-CY4 has been fed t o rats for 3 days. The ratio, Pp.34, in the amidine group of arginine isolated from organ proteins was somewhat higher than that in the material fed. The corresponding value in the amidine group of t)he carcass creatine was essentially equal to that in the arginine-fed group. The ratio PF5/C4 in the urinary urea of the st day was also approximately equal to that in the amidine of dietary arginine, but rose to values three to four times as high by the 3rd day. Over the 3 day period, two-thirds of the Cl4 ingested was exhaled as C40. These results indicate t)he absence of any process wherein amidine nitrogen of arginine is lost without coincident loss of amidine carbon. The transamidination from arginine ult imately to yield creatine proceeds without rupture of C-X bonds in the amidine group. In agreement with the findings of others, it is concluded that urea, arising from the amidine group of arginine, is subject to extensive catabolism in the intact animal, presumably in the gastrointestinal tract,. Much of the ammonia arising in this process is resynthesized into arginine, whence it reenters the urea cycle, whereas most of the CO is lost in the exhaled air. BIBLIOGRAPHY. Katner, S., in hlci~lro)-, W. II., and Glass, I<., Amino acid metabolism, Baltimore, 3 (955).
9 D. STETTEN, JR., AND B. BLOOM 73. Krebs, H. A., in Sumner, J. B., and Myrbllck, K., The enzymes, New York,, 866 (95). 3. Borsook, H., and Dubnoff, J. W., Science, 9,55 (940). 4. Bloch, K., J. Biol. Chem., 66,469 (946). 5. Dintzis, R. Z., and Hastings, A. B., Proc. Nut. Acad. SC., 39, 57 (953). 6. Chao, F., and Tarver, H., Proc. Sot. Exp. Biol. and Med., 84,406 (953). 7. Kurtz, A. C., J. Biol. Chem., 80, 53 (949). 8. Hartman, W. W., and Dreger, E. E., Org. Syntheses, ~0., 50 (943). 9. Bloch, H., Schoenheimer, R., and Rittenberg, D., J. Biol. Chem., 38,55 (94). 0. Stieglitz, J., Ber. them. Ges., 33, 57 (900).. Vickery, H. B., J. Biol. Chem., 3, 35 (940).. Cox, G. J., J. Biol. Chem., 78, 475 (98). 3. Hawk, P. B., Oser, B. L., and Summerson, W. H., Practical physiological chemistry, New York, 3th edition, 884,889 (954). 4. Fosse, R., Compt. rend. Acad., 67, 948 (93). 5. Conzelman, G. M., Jr., Mandel, H. G., and Smith, P. K., J. Biol. Chem., 0, 39 (953). 6. Stetten, M. R., and Schoenheimer, R., J. BioZ. Chem., 63, 3 (944). 7. Foster, G. L., Schoenheimer, R., and Rittenberg, D., J. BioZ. Chem., 7, 39 (939). 8. Benedict, S. R., J. BioZ. Chem., 8, 83 (94). 9. Kornberg, H. L., and Davies, R. E., Physiol. Rev., 36,69 (955).
10 THE METABOLISM OF THE AMIDINE GROUP OF ARGININE IN THE INTACT RAT DeWitt Stetten, Jr. and Ben Bloom J. Biol. Chem. 956, 0: Access the most updated version of this article at Alerts: When this article is cited When a correction for this article is posted Click here to choose from all of JBC's alerts This article cites 0 references, 0 of which can be accessed free at tml#ref-list-
STUDIES IN PROTEIN METABOLISM
 STUDIES IN PROTEIN METABOLISM XI. THE METABOLIC RELATION OF CREATINE AND CREATININE STUDIED WITH ISOTOPIC NITROGEN* BY KONRAD BLOCH AND RUDOLF SCHOENHEIMER (From the Department of Biochemistry, College
STUDIES IN PROTEIN METABOLISM XI. THE METABOLIC RELATION OF CREATINE AND CREATININE STUDIED WITH ISOTOPIC NITROGEN* BY KONRAD BLOCH AND RUDOLF SCHOENHEIMER (From the Department of Biochemistry, College
A MICROCOLORIMETRIC DETERMINATION OF CREATINE IN URINE BY THE JAFFE REACTION* HERTHA H. TAUSSKY
 A MICROCOLORIMETRIC DETERMINATION OF CREATINE IN URINE BY THE JAFFE REACTION* BY HERTHA H. TAUSSKY WITH THE TECHNICAL ASSISTANCE OF GLORIA KURZMANN (From the Russell Sage Institute of Pathology, Department
A MICROCOLORIMETRIC DETERMINATION OF CREATINE IN URINE BY THE JAFFE REACTION* BY HERTHA H. TAUSSKY WITH THE TECHNICAL ASSISTANCE OF GLORIA KURZMANN (From the Russell Sage Institute of Pathology, Department
DURING the course of certain investigations it became
 VOLUMETRIC DETERMINATION OF ETHER OR CYCLOPROPANE, CARBON DIOXIDE, NITROUS OXIDE AND OXYGEN IN ANESTHETIC MIXTURES By F. J. PRIME DURING the course of certain investigations it became necessary to be able
VOLUMETRIC DETERMINATION OF ETHER OR CYCLOPROPANE, CARBON DIOXIDE, NITROUS OXIDE AND OXYGEN IN ANESTHETIC MIXTURES By F. J. PRIME DURING the course of certain investigations it became necessary to be able
CHEM254 #4 The Synthesis of a Tertiary Alcohol Using a Pre-Made Grignard Reagent 1
 CHEM254 #4 The Synthesis of a Tertiary Alcohol Using a Pre-Made Grignard Reagent 1 Background In this project, we will perform a Grignard reaction using a pre-made Grignard reagent. Grignard reagents can
CHEM254 #4 The Synthesis of a Tertiary Alcohol Using a Pre-Made Grignard Reagent 1 Background In this project, we will perform a Grignard reaction using a pre-made Grignard reagent. Grignard reagents can
Effect of Various Protein Precipitants on Recoveries of Creatinine Added to Plasma
 Effect of Various Protein Precipitants on Recoveries of Creatinine Added to John F. Van Pilsum and M. Bovis NCOMPLETE RECOVERIES OF CREATININE added to plasma or serum have been reported by investigators
Effect of Various Protein Precipitants on Recoveries of Creatinine Added to John F. Van Pilsum and M. Bovis NCOMPLETE RECOVERIES OF CREATININE added to plasma or serum have been reported by investigators
Proskauer's reaction2 led to the discovery that acetyl methyl carbinol
 THE DIRECT DETERMINATION OF CREATINE IN PATHOLOGICAL URINE. BY G. STANLEY WALPOLE, B.Sc., F.I.C. (From the Wellcome Physiological Research Laboratories, Herne Hilly S.E.) THE investigation by Harden' into
THE DIRECT DETERMINATION OF CREATINE IN PATHOLOGICAL URINE. BY G. STANLEY WALPOLE, B.Sc., F.I.C. (From the Wellcome Physiological Research Laboratories, Herne Hilly S.E.) THE investigation by Harden' into
Chemistry 222 Fall 2012
 Experiment 2: Grignard Reaction Report Sheet Last Name: Siva First Name: Krishnaa Date: September 27, 2012 Section Number: 005 Objective: 1 mark (What is the purpose of this experiment?) The purpose of
Experiment 2: Grignard Reaction Report Sheet Last Name: Siva First Name: Krishnaa Date: September 27, 2012 Section Number: 005 Objective: 1 mark (What is the purpose of this experiment?) The purpose of
CREATININE AND CREATINE IN MUSCLE EXTRACTS.
 CREATININE AND CREATINE IN MUSCLE EXTRACTS. II. THE INFLUENCE OF THE REACTION OF THE MEDIUM ON THE CREATININE-CREATINE BALANCE IN INCUBATED EXTRACTS OF MUSCLE TISSUE OF THE ALBINO RAT. BY FREDERICK S.
CREATININE AND CREATINE IN MUSCLE EXTRACTS. II. THE INFLUENCE OF THE REACTION OF THE MEDIUM ON THE CREATININE-CREATINE BALANCE IN INCUBATED EXTRACTS OF MUSCLE TISSUE OF THE ALBINO RAT. BY FREDERICK S.
COMBINED SCIENCE 0653/6 CO-ORDINATED SCIENCES 0654/6
 Centre Number Candidate Number Candidate Name International General Certificate of Secondary Education UNIVERSITY OF CAMBRIDGE LOCAL EXAMINATIONS SYNDICATE COMBINED SCIENCE 0653/6 CO-ORDINATED SCIENCES
Centre Number Candidate Number Candidate Name International General Certificate of Secondary Education UNIVERSITY OF CAMBRIDGE LOCAL EXAMINATIONS SYNDICATE COMBINED SCIENCE 0653/6 CO-ORDINATED SCIENCES
6I unstable compound with phosphoric acid, which disappears during fatigue. (From the Department of Physiology, The University, Sheffield.
 6I2.744 ON THE CREATINE AND PHOSPHORUS CONTENT OF MUSCLE. BY MARION BROWN1 AND C. G. IMRIE. (From the Department of Physiology, The University, Sheffield.) IT is well known that the concentration of creatine
6I2.744 ON THE CREATINE AND PHOSPHORUS CONTENT OF MUSCLE. BY MARION BROWN1 AND C. G. IMRIE. (From the Department of Physiology, The University, Sheffield.) IT is well known that the concentration of creatine
19. The Grignard Reaction
 19. The Grignard eaction A. Introduction The Grignard reaction is an extremely valuable reaction in organic chemistry because it allows for the formation of carbon-carbon bonds. The reaction was discovered
19. The Grignard eaction A. Introduction The Grignard reaction is an extremely valuable reaction in organic chemistry because it allows for the formation of carbon-carbon bonds. The reaction was discovered
I. Colorimetric Determination of Trimethylamine as the
 Amines in Fish Muscle I. Colorimetric Determination of Trimethylamine as the Picrate Salt I ABSTRACT 1 A sensitive accurate colorimetric method for trimethylamine determination is presented, based on the
Amines in Fish Muscle I. Colorimetric Determination of Trimethylamine as the Picrate Salt I ABSTRACT 1 A sensitive accurate colorimetric method for trimethylamine determination is presented, based on the
A SIMPLE TITRATION METHOD FOR DETERMINING THE SPECIFIC GRAVITY ON ONE DROP OF URINE
 J. clin. Path. (1951), 4, 491. A SIMPLE TITRATION METHOD FOR DETERMINING THE SPECIFIC GRAVITY ON ONE DROP OF URINE BY From the Pathological Laboratory, the Peace Memorial Hospital, Watford (RECEIVED FOR
J. clin. Path. (1951), 4, 491. A SIMPLE TITRATION METHOD FOR DETERMINING THE SPECIFIC GRAVITY ON ONE DROP OF URINE BY From the Pathological Laboratory, the Peace Memorial Hospital, Watford (RECEIVED FOR
Environmental Engineering-I By Prof S S JAHAGIRDAR
 L- 28 Disinfection Part- I Environmental Engineering-I By Prof S S JAHAGIRDAR Contents Disinfection techniques- Ozonation, u/v radiation. Chemistry of chlorination, chlorine demand curve. Types of chlorination,
L- 28 Disinfection Part- I Environmental Engineering-I By Prof S S JAHAGIRDAR Contents Disinfection techniques- Ozonation, u/v radiation. Chemistry of chlorination, chlorine demand curve. Types of chlorination,
FlashCO2, CO2 at 23 $/ton
 FlashCO2, CO2 at 23 $/ton A cost effective solution of capturing CO2 from Steam Methane Reforming (SMR) Hydrogen production plants by the FlashCO2 process Introduction to a cost effective solution Driven
FlashCO2, CO2 at 23 $/ton A cost effective solution of capturing CO2 from Steam Methane Reforming (SMR) Hydrogen production plants by the FlashCO2 process Introduction to a cost effective solution Driven
François Auguste Victor Grignard, was a French chemist who discovered one of the world s first synthetic organometallic reactions.
 CHEM254 #6 The Synthesis of a Tertiary Alcohol Using a Pre-Made Grignard Reagent 1 Introduction/Background Image: François Auguste Victor Grignard (1871-1935) 1 François Auguste Victor Grignard, was a
CHEM254 #6 The Synthesis of a Tertiary Alcohol Using a Pre-Made Grignard Reagent 1 Introduction/Background Image: François Auguste Victor Grignard (1871-1935) 1 François Auguste Victor Grignard, was a
Honors Chemistry - Problem Set Chapter 13 Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT.
 Honors Chemistry - Problem Set Chapter 13 Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT. 1. Atmospheric pressure is 760 mm Hg. 2. The SI unit of pressure is
Honors Chemistry - Problem Set Chapter 13 Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT. 1. Atmospheric pressure is 760 mm Hg. 2. The SI unit of pressure is
The over-ventilated cat shows a similar adjustment to diminished. being over-ventilated, and he considered that on that account there was
 6I2.235:6I2.26I THE SOURCE OF COa EXPIRED AND THE SITE OF ITS RETENTION. BY LAURENCE IRVING, J. K. W. FERGUSON AND F. B. PLEWES. (From the Department of Physiology, University of Toronto.) AFTER evisceration
6I2.235:6I2.26I THE SOURCE OF COa EXPIRED AND THE SITE OF ITS RETENTION. BY LAURENCE IRVING, J. K. W. FERGUSON AND F. B. PLEWES. (From the Department of Physiology, University of Toronto.) AFTER evisceration
Noxious Fumes and Gases
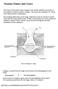 Noxious Fumes and Gases The tissues of the body require oxygen 0 2 for normal metabolic processes (ie. the oxidation of food to produce energy). They must also eliminate CO, which is the waste product
Noxious Fumes and Gases The tissues of the body require oxygen 0 2 for normal metabolic processes (ie. the oxidation of food to produce energy). They must also eliminate CO, which is the waste product
GUANIDINE AKD NITROUS ACID. I*
 GUANIDINE AKD NITROUS ACID. I* BY WILDER D. BANCROFT AKD BURTON C. BELDES 1. Introduction Organic chemists apparently have little idea and very little information as to what compound is formed when one
GUANIDINE AKD NITROUS ACID. I* BY WILDER D. BANCROFT AKD BURTON C. BELDES 1. Introduction Organic chemists apparently have little idea and very little information as to what compound is formed when one
Worksheet: Solubility
 1. According to your Reference Tables, which substance forms an unsaturated solution when 80 grams of the substance is dissolved in 100 grams of H 2 O at 10 C? (A) KI (B) KNO 3 (C) NaNO 3 (D) NaCl 2. The
1. According to your Reference Tables, which substance forms an unsaturated solution when 80 grams of the substance is dissolved in 100 grams of H 2 O at 10 C? (A) KI (B) KNO 3 (C) NaNO 3 (D) NaCl 2. The
Chemistry 1B Chapter 10 Worksheet - Daley. Name
 Name 1) The National Weather Service routinely supplies atmospheric pressure data to help pilots set their altimeters. The units the NWS uses for atmospheric pressure are inches of mercury. A barometric
Name 1) The National Weather Service routinely supplies atmospheric pressure data to help pilots set their altimeters. The units the NWS uses for atmospheric pressure are inches of mercury. A barometric
4/26/16. Section 1 Understanding Solutions. Solutions and Suspensions. Solutions and Suspensions. Solutions and Suspensions. Solvents and Solutes
 Section 1 Understanding Solutions On a hot day, you may think that a cool glass of plain water would be refreshing. However, if the glass were actually filled with plain water, it would taste stale. This
Section 1 Understanding Solutions On a hot day, you may think that a cool glass of plain water would be refreshing. However, if the glass were actually filled with plain water, it would taste stale. This
World of Chemistry Notes for Students [Chapter 13, page 1] Chapter 13 Gases
![World of Chemistry Notes for Students [Chapter 13, page 1] Chapter 13 Gases World of Chemistry Notes for Students [Chapter 13, page 1] Chapter 13 Gases](/thumbs/96/127526823.jpg) World of Chemistry Notes for Students [Chapter 3, page ] Chapter 3 Gases ) Sec 3.8 Kinetic Theory of Gases and the Nature of Gases The Kinetic Theory of Matter says that the tiny particles in all forms
World of Chemistry Notes for Students [Chapter 3, page ] Chapter 3 Gases ) Sec 3.8 Kinetic Theory of Gases and the Nature of Gases The Kinetic Theory of Matter says that the tiny particles in all forms
Experiment 18 Properties of Gases
 Experiment 18 Properties of Gases E18-1 E18-2 The Task In this experiment you will investigate some of the properties of gases, i.e. how gases flow, their phase changes and chemical reactivity. Skills
Experiment 18 Properties of Gases E18-1 E18-2 The Task In this experiment you will investigate some of the properties of gases, i.e. how gases flow, their phase changes and chemical reactivity. Skills
What do animals do to survive?
 What do animals do to survive? Section 26-1 All Animals have are carry out Eukaryotic cells with Heterotrophs Essential functions such as No cell walls Feeding Respiration Circulation Excretion Response
What do animals do to survive? Section 26-1 All Animals have are carry out Eukaryotic cells with Heterotrophs Essential functions such as No cell walls Feeding Respiration Circulation Excretion Response
Bt-Cry2Ab and Bt-Cry3Bb1 Multi-trait ELISA Kit DAS ELISA for the detection of Bt-Cry2Ab and Bt-Cry3Bb1 proteins Catalog number: PSM 26800
 List of contents Lot number Item 480 wells 4800 wells Antibody-coated 96-well microtiter plates 5 strip 50 solid Alkaline phosphatase enzyme conjugate, concentrated 0.550 ml 5 x 1.1 ml Peroxidase enzyme
List of contents Lot number Item 480 wells 4800 wells Antibody-coated 96-well microtiter plates 5 strip 50 solid Alkaline phosphatase enzyme conjugate, concentrated 0.550 ml 5 x 1.1 ml Peroxidase enzyme
Unit II Problem 4 Physiology: Diffusion of Gases and Pulmonary Circulation
 Unit II Problem 4 Physiology: Diffusion of Gases and Pulmonary Circulation - Physical principles of gases: Pressure of a gas is caused by the movement of its molecules against a surface (more concentration
Unit II Problem 4 Physiology: Diffusion of Gases and Pulmonary Circulation - Physical principles of gases: Pressure of a gas is caused by the movement of its molecules against a surface (more concentration
Name Chemistry Pre-AP
 Name Chemistry Pre-AP Notes: Gas Laws and Gas Stoichiometry Period Part 1: The Nature of Gases and The Gas Laws I. Nature of Gases A. Kinetic-Molecular Theory The - theory was developed to account for
Name Chemistry Pre-AP Notes: Gas Laws and Gas Stoichiometry Period Part 1: The Nature of Gases and The Gas Laws I. Nature of Gases A. Kinetic-Molecular Theory The - theory was developed to account for
Assessment of the Anabolic Effect of Androgens of the Edible Portion of the Shoot of Giginya Plant (Borassus aethiopum mart)
 Publications Available Online J. Sci. Res. 2 (2), 362-368 (2010) JOURNAL OF SCIENTIFIC RESEARCH www.banglajol.info/index.php/jsr Assessment of the Anabolic Effect of Androgens of the Edible Portion of
Publications Available Online J. Sci. Res. 2 (2), 362-368 (2010) JOURNAL OF SCIENTIFIC RESEARCH www.banglajol.info/index.php/jsr Assessment of the Anabolic Effect of Androgens of the Edible Portion of
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education
 UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education *9932384090* CO-ORDINATED SCIENCES 0654/51 Paper 5 Practical Test May/June 2011 2 hours Candidates
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education *9932384090* CO-ORDINATED SCIENCES 0654/51 Paper 5 Practical Test May/June 2011 2 hours Candidates
IASbaba ILP: Value Add Material - Matter MATTER. Iasbaba.com Page 1
 MATTER Iasbaba.com Page 1 LIQUID A. VAPOUR PRESSURE: Pressure of vapour in a closed container Changes majorly with temperature Has an exponential relationship with temperature Increases with increase in
MATTER Iasbaba.com Page 1 LIQUID A. VAPOUR PRESSURE: Pressure of vapour in a closed container Changes majorly with temperature Has an exponential relationship with temperature Increases with increase in
3 Solubility and Concentration
 CHAPTER 8 SECTION Solutions 3 Solubility and Concentration KEY IDEAS As you read this section, keep these questions in mind: What is solubility? What happens when you add more solute to a saturated solution?
CHAPTER 8 SECTION Solutions 3 Solubility and Concentration KEY IDEAS As you read this section, keep these questions in mind: What is solubility? What happens when you add more solute to a saturated solution?
CRYSTALLIZATION FOULING IN PACKED COLUMNS
 CRYSTALLIZATION FOULING IN PACKED COLUMNS D. Großerichter and J. Stichlmair Lehrstuhl für Fluidverfahrenstechnik, Technische Universität München, Munich, Germany ABSTRACT Fouling due to crystallization
CRYSTALLIZATION FOULING IN PACKED COLUMNS D. Großerichter and J. Stichlmair Lehrstuhl für Fluidverfahrenstechnik, Technische Universität München, Munich, Germany ABSTRACT Fouling due to crystallization
Before we can understand what dropsy is we must first understand two natural occurring scientific processes namely, Osmosis and Diffusion.
 Dropsy (Ascites) By Duncan Griffiths Before we can understand what dropsy is we must first understand two natural occurring scientific processes namely, Osmosis and Diffusion. Osmosis is the movement of
Dropsy (Ascites) By Duncan Griffiths Before we can understand what dropsy is we must first understand two natural occurring scientific processes namely, Osmosis and Diffusion. Osmosis is the movement of
An Important Change to Pinery s Water in March
 An Important Change to Pinery s Water in March Chloramine Conversion Project The Pinery Water District is scheduled to convert its water disinfection process to a chloramine treatment process March 2018.
An Important Change to Pinery s Water in March Chloramine Conversion Project The Pinery Water District is scheduled to convert its water disinfection process to a chloramine treatment process March 2018.
Author of: The Six-Pack Diet Plan: The Secrets to Getting Lean Abs and a Rock-Hard Body Permanently
 Interview with Rehan Jalali, President, Supplement Research Foundation Author of: The Six-Pack Diet Plan: The Secrets to Getting Lean Abs and a Rock-Hard Body Permanently Background Information: According
Interview with Rehan Jalali, President, Supplement Research Foundation Author of: The Six-Pack Diet Plan: The Secrets to Getting Lean Abs and a Rock-Hard Body Permanently Background Information: According
Water Balance Indexes Revised September 2017
 Water Balance Indexes Revised September 2017 Brought to you by the APSP Recreational Water Quality Committee (RWQC) I. INTRODUCTION There are several water balance indexes that are being used as tools
Water Balance Indexes Revised September 2017 Brought to you by the APSP Recreational Water Quality Committee (RWQC) I. INTRODUCTION There are several water balance indexes that are being used as tools
Bt-Cry1Ac & Bt-Cry2A Multi-trait ELISA Kit DAS ELISA for the detection of Bt-Cry1Ac & Bt-Cry2A proteins Catalog number: PSM 14900
 List of contents Lot number Item 480 wells 4800 wells Antibody-coated 96-well microtiter plates 5 strip 50 solid Alkaline phosphatase enzyme conjugate, concentrated 0.550 ml 5 x 1.1 ml Peroxidase enzyme
List of contents Lot number Item 480 wells 4800 wells Antibody-coated 96-well microtiter plates 5 strip 50 solid Alkaline phosphatase enzyme conjugate, concentrated 0.550 ml 5 x 1.1 ml Peroxidase enzyme
Pool Water Chemistry Guide
 Pool Water Chemistry Guide water test The water chemistry information in this guide will help you keep your pool water clean and clear, swimmer safe, and plaster friendly. the clear choice in pool care
Pool Water Chemistry Guide water test The water chemistry information in this guide will help you keep your pool water clean and clear, swimmer safe, and plaster friendly. the clear choice in pool care
REVISION: GASEOUS EXCHANGE & EXCRETION 11 SEPTEMBER 2013
 REVISION: GASEOUS EXCHANGE & EXCRETION 11 SEPTEMBER 2013 Lesson Description In this lesson we: Revise gaseous exchange in different animals and examine the structure of the kidney Key Concepts Important
REVISION: GASEOUS EXCHANGE & EXCRETION 11 SEPTEMBER 2013 Lesson Description In this lesson we: Revise gaseous exchange in different animals and examine the structure of the kidney Key Concepts Important
Paper No and Title. Paper 9: Organic Chemistry-III (Reaction Mechanism-2) Module no.13: Addition of Grignard reagent
 Subject Chemistry Paper No and Title Module No and Title Module Tag 9: Organic Chemistry-III (Reaction Mechanism-2) 13: Addition of Grignard reagent CHE_P9_M13 TABLE OF CONTENTS 1. Learning Outcomes 2.
Subject Chemistry Paper No and Title Module No and Title Module Tag 9: Organic Chemistry-III (Reaction Mechanism-2) 13: Addition of Grignard reagent CHE_P9_M13 TABLE OF CONTENTS 1. Learning Outcomes 2.
States of Matter. Q 7. Calculate the average of kinetic energy, in joules of the molecules in 8.0 g of methane at 27 o C. (IIT JEE Marks)
 Q 1. States of Matter Calculate density of NH 3 at 30 o C and 5 atm pressure Q 2. (IIT JEE 1978 3 Marks) 3.7 g of a gas at 25 o C occupied the same volume as 0.184g of hydrogen at 17 o C and at the same
Q 1. States of Matter Calculate density of NH 3 at 30 o C and 5 atm pressure Q 2. (IIT JEE 1978 3 Marks) 3.7 g of a gas at 25 o C occupied the same volume as 0.184g of hydrogen at 17 o C and at the same
Name:. Correct Questions = Wrong Questions =.. Unattempt Questions = Marks =
 Name:. Correct Questions = Wrong Questions =.. Unattempt Questions = Marks = Lab Problem 1 Design and carry out an experiment to determine the density of the plastic object you have been given. You may
Name:. Correct Questions = Wrong Questions =.. Unattempt Questions = Marks = Lab Problem 1 Design and carry out an experiment to determine the density of the plastic object you have been given. You may
Lung Volumes and Capacities
 Lung Volumes and Capacities Normally the volume of air entering the lungs during a single inspiration is approximately equal to the volume leaving on the subsequent expiration and is called the tidal volume.
Lung Volumes and Capacities Normally the volume of air entering the lungs during a single inspiration is approximately equal to the volume leaving on the subsequent expiration and is called the tidal volume.
9/28/2017 OBJ: SWBAT determine the solubility of 10 compounds. 1. What makes Magic Sand hydrophobic?
 9/28/2017 OBJ: SWBAT determine the solubility of 10 compounds 1. What makes Magic Sand hydrophobic? Solutions Pure Substances & Mixtures o What's the matter? o Pure Substances have a definite set of physical
9/28/2017 OBJ: SWBAT determine the solubility of 10 compounds 1. What makes Magic Sand hydrophobic? Solutions Pure Substances & Mixtures o What's the matter? o Pure Substances have a definite set of physical
CHAPTER 5 : THE AIR AROUND US
 CHAPTER 5 : THE AIR AROUND US The Composition of Air Air is a mixture consist of Nitrogen 78% Oxygen 21% Carbon dioxide 0.03% Inert gases 0.97% Water vapour Microorganism Dust The percentage of the constituents
CHAPTER 5 : THE AIR AROUND US The Composition of Air Air is a mixture consist of Nitrogen 78% Oxygen 21% Carbon dioxide 0.03% Inert gases 0.97% Water vapour Microorganism Dust The percentage of the constituents
Care of the swimming pool
 Care of the swimming pool The perfect care and maintenance of a swimming pool depends on two key elements: the water and the PVC membrane. In this way, by maintaining periodical control of the state of
Care of the swimming pool The perfect care and maintenance of a swimming pool depends on two key elements: the water and the PVC membrane. In this way, by maintaining periodical control of the state of
Slide 1. Slide 2. Slide 3 What do YOU know about oxygen? Dissolved Oxygen. Oxygen. I can t breath in the water but the fish can!
 Slide 1 Dissolved Oxygen I can t breath in the water but the fish can! Slide 2 Oxygen Oxygen is one of the fundamental resources required by life forms on Earth. Aquatic ecosystems have a wide assortment
Slide 1 Dissolved Oxygen I can t breath in the water but the fish can! Slide 2 Oxygen Oxygen is one of the fundamental resources required by life forms on Earth. Aquatic ecosystems have a wide assortment
For Establishing and Maintaining Your Spa using PROtech Spa Chemicals
 Spa Care For Establishing and Maintaining Your Spa using PROtech Spa Chemicals QUICK START Add water up to the level recommended by the manufacturer of your spa and let the pump circulate for at least
Spa Care For Establishing and Maintaining Your Spa using PROtech Spa Chemicals QUICK START Add water up to the level recommended by the manufacturer of your spa and let the pump circulate for at least
Pyrrole Chemistry. Part XII. The Mechanism of the Reaction Between the Pyrrole Grignard Reagent and Acylating Agents1
 Pyrrole Chemistry. Part XII. The Mechanism of the Reaction Between the Pyrrole Grignard Reagent and Acylating Agents1 C. E. LOADER AND HUGH J. ANDERSON Department of Chemistry, Memorial University, St.
Pyrrole Chemistry. Part XII. The Mechanism of the Reaction Between the Pyrrole Grignard Reagent and Acylating Agents1 C. E. LOADER AND HUGH J. ANDERSON Department of Chemistry, Memorial University, St.
A SIMPLE, ACCURATE METHOD OF ESTIMATING CARBON MONOXIDE IN BLOOD
 A SIMPLE, ACCURATE METHOD OF ESTIMATING CARBON MONOXIDE IN BLOOD BY F. J. W. ROUGHTOX (From the Physiological Laboratory, University of Cambridge, Cambridge, England) (Received for publication, October
A SIMPLE, ACCURATE METHOD OF ESTIMATING CARBON MONOXIDE IN BLOOD BY F. J. W. ROUGHTOX (From the Physiological Laboratory, University of Cambridge, Cambridge, England) (Received for publication, October
MONKEY ASSIMILATE STUDY 1
 MONKEY ASSIMILATE STUDY 1 PROCEDURES FOR STUDY Six healthy, lean, adult males volunteered for this study. None of the participants were following any particular protein-rich dietary regime, muscle-toning
MONKEY ASSIMILATE STUDY 1 PROCEDURES FOR STUDY Six healthy, lean, adult males volunteered for this study. None of the participants were following any particular protein-rich dietary regime, muscle-toning
Name:. Correct Questions = Wrong Questions =.. Unattempt Questions = Marks =
 Name:. Correct Questions = Wrong Questions =.. Unattempt Questions = Marks = Lab Problem 1 One way of using solar energy is to capture heat from the sun in a reservoir to be released later. You have been
Name:. Correct Questions = Wrong Questions =.. Unattempt Questions = Marks = Lab Problem 1 One way of using solar energy is to capture heat from the sun in a reservoir to be released later. You have been
CspB ELISA Kit DAS ELISA for the detection of CspB protein Catalog number: PSP 35500
 List of contents Lot number Item 480 wells 4800 wells Antibody-coated 96-well microtiter plates 5 strip 50 solid Peroxidase enzyme conjugate, concentrated 0.550 ml 5 x 1.1 ml RUB6 enzyme conjugate diluent
List of contents Lot number Item 480 wells 4800 wells Antibody-coated 96-well microtiter plates 5 strip 50 solid Peroxidase enzyme conjugate, concentrated 0.550 ml 5 x 1.1 ml RUB6 enzyme conjugate diluent
Are chloramines new? Why is my water supplier making the change to chloramines?
 In order to maintain quality water, the Central Coast Water Authority uses chloramines as disinfectant. Chloramines are a combination of chlorine and a small amount of ammonia. This web page provides answers
In order to maintain quality water, the Central Coast Water Authority uses chloramines as disinfectant. Chloramines are a combination of chlorine and a small amount of ammonia. This web page provides answers
SOLUBILITY OF A SOLID IN WATER
 1516L Experiment 2 SOLUBILITY OF A SOLID IN WATER Objectives In this experiment you will determine the solubility of potassium nitrate (KNO 3 ) in water at various temperatures. You will prepare a plot
1516L Experiment 2 SOLUBILITY OF A SOLID IN WATER Objectives In this experiment you will determine the solubility of potassium nitrate (KNO 3 ) in water at various temperatures. You will prepare a plot
MATERIAL SAFETY DATA SHEET
 1. IDENTIFICATION CATALOG NO. Detector AP Chemiluminescent Blotting Kit 54-30-01, 54-30-02 Issue Date:21 March, 2006 Revision Date: 21 January, 2004 Reagents That Contain Hazardous Materials Catalog No.
1. IDENTIFICATION CATALOG NO. Detector AP Chemiluminescent Blotting Kit 54-30-01, 54-30-02 Issue Date:21 March, 2006 Revision Date: 21 January, 2004 Reagents That Contain Hazardous Materials Catalog No.
GRADE 6: Materials 1. UNIT 6M.1 7 hours. Solubility. Resources. About this unit. Previous learning. Expectations. Key vocabulary and technical terms
 GRADE 6: Materials 1 Solubility UNIT 6M.1 7 hours About this unit This is the first of four units on materials in Grade 6. This unit builds on the study of the properties of water in Unit 5M.1. Unit 7M.1
GRADE 6: Materials 1 Solubility UNIT 6M.1 7 hours About this unit This is the first of four units on materials in Grade 6. This unit builds on the study of the properties of water in Unit 5M.1. Unit 7M.1
1 Designation of the substance or mixture, respectively, and of the company
 Metrohm AG CH-9100 Herisau Switzerland info@metrohm.com www.metrohm.com Material Safety Data Sheet Pursuant to Regulation (EC) No. 1272/2008 Storage solution, 250 ml June 19, 2013 8.106.7575EN fpe/ts Storage
Metrohm AG CH-9100 Herisau Switzerland info@metrohm.com www.metrohm.com Material Safety Data Sheet Pursuant to Regulation (EC) No. 1272/2008 Storage solution, 250 ml June 19, 2013 8.106.7575EN fpe/ts Storage
LEVANT WORMSEED FOR HOMOEOPATHIC PREPARATIONS CINA FOR HOMOEOPATHIC PREPARATIONS
 LEVANT WORMSEED FOR HOMOEOPATHIC PREPARATIONS CINA FOR HOMOEOPATHIC PREPARATIONS Artemisia cina ad praeparationes homoeopathicas DEFINITION Dried, non-blooming capitulum of Artemisia cina Berg. Content
LEVANT WORMSEED FOR HOMOEOPATHIC PREPARATIONS CINA FOR HOMOEOPATHIC PREPARATIONS Artemisia cina ad praeparationes homoeopathicas DEFINITION Dried, non-blooming capitulum of Artemisia cina Berg. Content
THE ANDERSONS SAFETY DATA SHEET DATE PREPARED: 2/3/06 CURRENT AS OF: 8/16/16
 THE ANDERSONS SAFETY DATA SHEET DATE PREPARED: 2/3/06 CURRENT AS OF: 8/16/16 SECTION 1: PRODUCT / SUPPLIER IDENTIFICATION PRODUCT CODE: PRODUCT NAME: PRODUCT USE: MFR INFO: The Andersons Professional Turf
THE ANDERSONS SAFETY DATA SHEET DATE PREPARED: 2/3/06 CURRENT AS OF: 8/16/16 SECTION 1: PRODUCT / SUPPLIER IDENTIFICATION PRODUCT CODE: PRODUCT NAME: PRODUCT USE: MFR INFO: The Andersons Professional Turf
TECHNICAL INFORMATION Ninhydrin Crystals Catalog Nos. NRP01A, NRP02B, NRP03C
 SIRCHIE Products Vehicles Training Copyright 2011 by SIRCHIE All Rights Reserved. TECHNICAL INFORMATION Ninhydrin Crystals Catalog Nos. NRP01A, NRP02B, NRP03C Application Development of latent fingerprints
SIRCHIE Products Vehicles Training Copyright 2011 by SIRCHIE All Rights Reserved. TECHNICAL INFORMATION Ninhydrin Crystals Catalog Nos. NRP01A, NRP02B, NRP03C Application Development of latent fingerprints
R-410A 50% R-32 50% R-125.
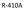 R-410A R-410A Facts Ozone friendly- No Chlorine Replacement for R-22, NOT a drop in! Higher Pressure Range-50 to 75% higher than R-22 Requires Special Lubricants R-410A It s a Blended Refrigerant. 50%
R-410A R-410A Facts Ozone friendly- No Chlorine Replacement for R-22, NOT a drop in! Higher Pressure Range-50 to 75% higher than R-22 Requires Special Lubricants R-410A It s a Blended Refrigerant. 50%
MATERIAL SAFETY DATA SHEET
 SECTION 1: PRODUCT AND COMPANY IDENTIFICATION PRODUCT NAME: PRODUCT CODES: MANUFACTURER: DIVISION: ADDRESS: INM MILKY BOND BASE COAT INMMBBC International Nail Manufacturers (inm) Division of Nail Cartel,
SECTION 1: PRODUCT AND COMPANY IDENTIFICATION PRODUCT NAME: PRODUCT CODES: MANUFACTURER: DIVISION: ADDRESS: INM MILKY BOND BASE COAT INMMBBC International Nail Manufacturers (inm) Division of Nail Cartel,
Hydrofluoric Acid SOP
 Introduction Hydrofluoric acid (HF) has a number of chemical, physical, and toxicological properties, which make handling this material especially hazardous. Anhydrous HF is a clear, colorless, fuming,
Introduction Hydrofluoric acid (HF) has a number of chemical, physical, and toxicological properties, which make handling this material especially hazardous. Anhydrous HF is a clear, colorless, fuming,
MATERIAL SAFETY DATA SHEET
 MATERIAL SAFETY DATA SHEET 1 IDENTIFICATION OF THE SUBSTANCE/MIXTURE AND OF THE COMPANY/UNDERTAKING 1.1 Product identifiers Product Name Zinc Benzoate NO. 553-72-0 1.2 Relevant identified uses of the substance
MATERIAL SAFETY DATA SHEET 1 IDENTIFICATION OF THE SUBSTANCE/MIXTURE AND OF THE COMPANY/UNDERTAKING 1.1 Product identifiers Product Name Zinc Benzoate NO. 553-72-0 1.2 Relevant identified uses of the substance
Gas Exchange ACTIVITY OVERVIEW SUMMARY KEY CONCEPTS AND PROCESS SKILLS KEY VOCABULARY. Teacher s Guide B-75 L A B O R ATO R Y
 Gas Exchange 17 40- to 2 50-minute sessions ACTIVITY OVERVIEW L A B O R ATO R Y SUMMARY This activity explores the role of the respiratory system in the regulation of gases in the blood. Students investigate
Gas Exchange 17 40- to 2 50-minute sessions ACTIVITY OVERVIEW L A B O R ATO R Y SUMMARY This activity explores the role of the respiratory system in the regulation of gases in the blood. Students investigate
SAFETY DATA SHEET. FLEET WASH A Revision 01/30/ PRODUCT AND COMPANY IDENTIFICATION 2. HAZARDOUS IDENTIFICATION
 1. PRODUCT AND COMPANY IDENTIFICATION Product Identifier Recommended use Distributed by: FLEET WASH A Cleaner DyChem International 560 North 500 West Salt Lake City, UT 84116 (800) 453-4606 Emergency Contact
1. PRODUCT AND COMPANY IDENTIFICATION Product Identifier Recommended use Distributed by: FLEET WASH A Cleaner DyChem International 560 North 500 West Salt Lake City, UT 84116 (800) 453-4606 Emergency Contact
PREPARATION AND PROPERTIES OF ATMOSPHERIC GASES I: Carbon Dioxide and Oxygen
 PREPARATION AND PROPERTIES OF ATMOSPHERIC GASES I: Carbon Dioxide and Oxygen.... :O = C = O:.... :O = O: INTRODUCTION The atmosphere consists predominantly of three gases -- nitrogen (N 2 ) 78%, oxygen
PREPARATION AND PROPERTIES OF ATMOSPHERIC GASES I: Carbon Dioxide and Oxygen.... :O = C = O:.... :O = O: INTRODUCTION The atmosphere consists predominantly of three gases -- nitrogen (N 2 ) 78%, oxygen
Aaron Vo 11/4/15 A03 Wednesday 2-5 PM Group 7/HS LAB 6- Discovering how ph/heat Change Affects Solubility
 Aaron Vo 11/4/15 A03 Wednesday 2-5 PM Group 7/HS LAB 6- Discovering how ph/heat Change Affects Solubility I. PURPOSE/OBJECTIVE: The purpose of lab 6 was to observe how altering the ph or heat to proteins
Aaron Vo 11/4/15 A03 Wednesday 2-5 PM Group 7/HS LAB 6- Discovering how ph/heat Change Affects Solubility I. PURPOSE/OBJECTIVE: The purpose of lab 6 was to observe how altering the ph or heat to proteins
Biology. Slide 1 of 53. End Show. Copyright Pearson Prentice Hall
 Biology 1 of 53 Chapter 33 Comparing Chordates 2 of 53 This chapter is a good revision of the material we saw during Unit III. 3 of 53 4 of 53 Controlling Body Temperature The control of body temperature
Biology 1 of 53 Chapter 33 Comparing Chordates 2 of 53 This chapter is a good revision of the material we saw during Unit III. 3 of 53 4 of 53 Controlling Body Temperature The control of body temperature
VOLUNTARY BREATHHOLDING. I. PULMONARY GAS
 VOLUNTARY BREATHHOLDING. I. PULMONARY GAS EXCHANGE DURING BREATHHOLDING'1 By CHARLES D. STEVENS, EUGENE B. FERRIS, JOSEPH P. WEBB, GEORGE L. ENGEL, AND MYRTLE LOGAN (From the Departments of Internal Medicine
VOLUNTARY BREATHHOLDING. I. PULMONARY GAS EXCHANGE DURING BREATHHOLDING'1 By CHARLES D. STEVENS, EUGENE B. FERRIS, JOSEPH P. WEBB, GEORGE L. ENGEL, AND MYRTLE LOGAN (From the Departments of Internal Medicine
Levels of CO2 in Arterial Blood of Carp under Carbon Dioxide Anesthesia
 J. Nutr. Sci. Vitaminol., 28, 35-39, 1982 Levels of CO2 in Arterial Blood of Carp under Carbon Dioxide Anesthesia Hisateru MITSUDA, Saburo UENO, Hiroshi MIZUNO, Tadashi UEDA, Hiromi FUJIKAWA, Tomoko NOHARA,
J. Nutr. Sci. Vitaminol., 28, 35-39, 1982 Levels of CO2 in Arterial Blood of Carp under Carbon Dioxide Anesthesia Hisateru MITSUDA, Saburo UENO, Hiroshi MIZUNO, Tadashi UEDA, Hiromi FUJIKAWA, Tomoko NOHARA,
SOLUBILITY OF A SOLID IN WATER
 1516L Experiment 1 SOLUBILITY OF A SOLID IN WATER Objectives In this experiment you will determine the solubility of potassium nitrate (KNO 3 ) in water at various temperatures. You will prepare a plot
1516L Experiment 1 SOLUBILITY OF A SOLID IN WATER Objectives In this experiment you will determine the solubility of potassium nitrate (KNO 3 ) in water at various temperatures. You will prepare a plot
: RSID Extraction Buffer
 SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product form Product name. Product code : Mixture : RSID Extraction Buffer : RSID02 1.2. Relevant
SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1. Product identifier Product form Product name. Product code : Mixture : RSID Extraction Buffer : RSID02 1.2. Relevant
The Variation of Muscle Oxygen Consumption With Velocity of Shortening
 The Variation of Muscle Oxygen Consumption With Velocity of Shortening R.J. BASKIN From the Department of Zoology, University of California, Davis ABSTRACT Total oxygen consumption following contraction
The Variation of Muscle Oxygen Consumption With Velocity of Shortening R.J. BASKIN From the Department of Zoology, University of California, Davis ABSTRACT Total oxygen consumption following contraction
Material Safety Data Sheet
 ES800 Gram Stain Kit Page 1 of 8 M S D S Material Safety Data Sheet 24 HOUR EMERGENCY TELEPHONE NUMBER CALL CHEMTREC 1-800-424-9300 189 Twin County Road Morgantown, PA 19543 (610) 524-5810 ES800 is a gram
ES800 Gram Stain Kit Page 1 of 8 M S D S Material Safety Data Sheet 24 HOUR EMERGENCY TELEPHONE NUMBER CALL CHEMTREC 1-800-424-9300 189 Twin County Road Morgantown, PA 19543 (610) 524-5810 ES800 is a gram
Spa Start Up & Maintenance
 Spa Start Up & Maintenance Your spa or hot tub is an investment in relaxation. Follow these simple steps for maximum enjoyment! Quality We offer a complete variety of products to support both of our chlorinating
Spa Start Up & Maintenance Your spa or hot tub is an investment in relaxation. Follow these simple steps for maximum enjoyment! Quality We offer a complete variety of products to support both of our chlorinating
Alfalfa Hay for Horses: Myths vs. Reality
 Alfalfa Hay for Horses: Myths vs. Reality Laurie Lawrence, Ph.D. Professor, Equine Nutrition Department of Animal and Food Sciences University of Kentucky Lexington, KY 40546-0215 llawrenc@uky.edu How
Alfalfa Hay for Horses: Myths vs. Reality Laurie Lawrence, Ph.D. Professor, Equine Nutrition Department of Animal and Food Sciences University of Kentucky Lexington, KY 40546-0215 llawrenc@uky.edu How
Date Printed: 03/22/2005 Date Updated: 03/06/2004 Version 1.2. Sigma-Aldrich Canada, Ltd
 SIGMA-ALDRICH MATERIAL SAFETY DATA SHEET Date Printed: 03/22/2005 Date Updated: 03/06/2004 Version 1.2 Section 1 - Product and Company Information Product Name ETHANOL-D, 99 ATOM % D Product Number 452556
SIGMA-ALDRICH MATERIAL SAFETY DATA SHEET Date Printed: 03/22/2005 Date Updated: 03/06/2004 Version 1.2 Section 1 - Product and Company Information Product Name ETHANOL-D, 99 ATOM % D Product Number 452556
SAFETY DATA SHEET according to Regulation (EG) no 1907/2006 Generic EU MSDS No country specific data REVISION DATE:
 SAFETY DATA SHEET according to Regulation (EG) 1907/2006 Generic EU MSDS No country specific data REVISION DATE: 2014-03-19 1. Identification of the substance/mixture and of the company/undertaking 1.1
SAFETY DATA SHEET according to Regulation (EG) 1907/2006 Generic EU MSDS No country specific data REVISION DATE: 2014-03-19 1. Identification of the substance/mixture and of the company/undertaking 1.1
2-AMINOANTHRACENE HAZARDS IDENTIFICATION Classification: According to regulation (EC) No 1272/2008 Labelling and
 2-AMINOANTHRACENE 1. IDENTIFICATION OF SUBSTANCE / PREPARATION AND THE COMPANY UNDERTAKING Chemical name: Product code: PPC-AA01, PPC-AA02 Synonyms: 2-Anthramine Recommended uses: For R&D purposes Supplier:
2-AMINOANTHRACENE 1. IDENTIFICATION OF SUBSTANCE / PREPARATION AND THE COMPANY UNDERTAKING Chemical name: Product code: PPC-AA01, PPC-AA02 Synonyms: 2-Anthramine Recommended uses: For R&D purposes Supplier:
What is photosynthesis? Photosynthesis II: Dissolved O2
 What is photosynthesis? Photosynthesis II: Dissolved O2 Measuring air dissolved pressure oxygen inside inside an active an active photosynthetic photosynthetic Objective The purpose of this activity is
What is photosynthesis? Photosynthesis II: Dissolved O2 Measuring air dissolved pressure oxygen inside inside an active an active photosynthetic photosynthetic Objective The purpose of this activity is
CLASS COPY-DO NOT WRITE ON
 Save Our Shells - Central Question: How does carbon dioxide affect salt water? CLASS COPY-DO NOT WRITE ON Overview of experiment: Exhaling carbon dioxide into a beaker of salt water mimics the gas exchange
Save Our Shells - Central Question: How does carbon dioxide affect salt water? CLASS COPY-DO NOT WRITE ON Overview of experiment: Exhaling carbon dioxide into a beaker of salt water mimics the gas exchange
Gas Laws. 2 HCl(aq) + CaCO 3 (s) H 2 O(l) + CO 2 (g) + CaCl 2 (aq) HCl(aq) + NaHCO 3 (s) H 2 O(l) + CO 2 (g) + NaCl(aq)
 Gas Laws Introduction: Although we cannot see gases, we can observe their behavior and study their properties. For example, we can watch a balloon filled with helium gas floating in air and conclude that
Gas Laws Introduction: Although we cannot see gases, we can observe their behavior and study their properties. For example, we can watch a balloon filled with helium gas floating in air and conclude that
(breed) Anadromous. wholly seawater
 Maintenance of Constancy of the Internal Environment during Fish Migration Between Fresh and Seawater Group 4: Kuan Yi Xian, Jeeann Lee Tien Ming Lee Tze Chuen Lee Wan-Jean Lee Yee Tjin Migration Seasonal
Maintenance of Constancy of the Internal Environment during Fish Migration Between Fresh and Seawater Group 4: Kuan Yi Xian, Jeeann Lee Tien Ming Lee Tze Chuen Lee Wan-Jean Lee Yee Tjin Migration Seasonal
Micro Channel Recuperator for a Reverse Brayton Cycle Cryocooler
 Micro Channel Recuperator for a Reverse Brayton Cycle Cryocooler C. Becnel, J. Lagrone, and K. Kelly Mezzo Technologies Baton Rouge, LA USA 70806 ABSTRACT The Missile Defense Agency has supported a research
Micro Channel Recuperator for a Reverse Brayton Cycle Cryocooler C. Becnel, J. Lagrone, and K. Kelly Mezzo Technologies Baton Rouge, LA USA 70806 ABSTRACT The Missile Defense Agency has supported a research
THE DEVELOPMENT OF LYMPH NODES AFTER INJECTION OF FLINT OF VARIABLE SIZE INTO THE LUNGS OF RATS
 Brit. J. industr. Med., 1954, 11, 145. THE DEVELOPMENT OF LYMPH NODES AFTER INJECTION OF FLINT OF VARIABLE SIZE INTO THE LUNGS OF RATS BY G. NAGELSCHMIDT, E. S. NELSON, E. J. KING, and C. V. HARRISON From
Brit. J. industr. Med., 1954, 11, 145. THE DEVELOPMENT OF LYMPH NODES AFTER INJECTION OF FLINT OF VARIABLE SIZE INTO THE LUNGS OF RATS BY G. NAGELSCHMIDT, E. S. NELSON, E. J. KING, and C. V. HARRISON From
Catabolism in Skeletal Muscle The Phosphagen System
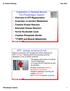 Catabolism in Skeletal Muscle The Phosphagen System Overview of ATP Regeneration Anaerobic vs Aerobic Metabolism Creatine Kinase Reaction Adenylate Kinase Reaction Purine Nucleotide Cycle Creatine Phosphate
Catabolism in Skeletal Muscle The Phosphagen System Overview of ATP Regeneration Anaerobic vs Aerobic Metabolism Creatine Kinase Reaction Adenylate Kinase Reaction Purine Nucleotide Cycle Creatine Phosphate
TESTOFEN HUMAN CLINICAL TRIAL GENCOR PACIFIC, INC. Copyright 2006 by Gencor Pacific, Inc.
 GENCOR PACIFIC, INC. 920 E. Orangethorpe Avenue, Suite B, Anaheim, CA 92801 Ph: 714.870.8723 714.870.8724 efax: 732.875.0306 drjit@gencorpacific.com gita@gencorpacific.com www.gencorpacific.com TESTOFEN
GENCOR PACIFIC, INC. 920 E. Orangethorpe Avenue, Suite B, Anaheim, CA 92801 Ph: 714.870.8723 714.870.8724 efax: 732.875.0306 drjit@gencorpacific.com gita@gencorpacific.com www.gencorpacific.com TESTOFEN
Chapter 5 TEST: Gases
 Chapter 5 TEST: Gases 1) Gases generally have A) low density B) high density C) closely packed particles D) no increase in volume when temperature is increased E) no decrease in volume when pressure is
Chapter 5 TEST: Gases 1) Gases generally have A) low density B) high density C) closely packed particles D) no increase in volume when temperature is increased E) no decrease in volume when pressure is
Date Printed: 06/08/2006 Date Updated: 01/26/2006 Version 1.7 L-HISTIDINE MONOHYDROCHLORIDE MONOHYDRATE, REAGENT GRADE, >= 98%
 SIGMA-ALDRICH MATERIAL SAFETY DATA SHEET Date Printed: 06/08/2006 Date Updated: 01/26/2006 Version 1.7 Section 1 - Product and Company Information Product Name Product Number Brand L-HISTIDINE MONOHYDROCHLORIDE
SIGMA-ALDRICH MATERIAL SAFETY DATA SHEET Date Printed: 06/08/2006 Date Updated: 01/26/2006 Version 1.7 Section 1 - Product and Company Information Product Name Product Number Brand L-HISTIDINE MONOHYDROCHLORIDE
Pco2 *20times = 0.6, 2.4, so the co2 carried in the arterial blood in dissolved form is more than the o2 because of its solubility.
 Physiology, sheet #9 Oxygen, is first dissolved in the plasma and the cytosol of the rbc, we have around blood constitutes 7% of our body weight, oxygen, in the capillaries is present in the rbc s and
Physiology, sheet #9 Oxygen, is first dissolved in the plasma and the cytosol of the rbc, we have around blood constitutes 7% of our body weight, oxygen, in the capillaries is present in the rbc s and
Example: 25 C = ( ) K = 298 K. Pressure Symbol: p Units: force per area 1Pa (Pascal) = 1 N/m 2
 Chapter 6: Gases 6.1 Measurements on Gases MH5, Chapter 5.1 Let s look at a certain amount of gas, i.e. trapped inside a balloon. To completely describe the state of this gas one has to specify the following
Chapter 6: Gases 6.1 Measurements on Gases MH5, Chapter 5.1 Let s look at a certain amount of gas, i.e. trapped inside a balloon. To completely describe the state of this gas one has to specify the following
The Respiratory System
 Respiration and Excretion Name Date Class The Respiratory System This section describes the parts of the respiratory system and how they work to help you breathe and speak. Use Target Reading Skills As
Respiration and Excretion Name Date Class The Respiratory System This section describes the parts of the respiratory system and how they work to help you breathe and speak. Use Target Reading Skills As
Safety Data Sheet 1. PRODUCT AND COMPANY IDENTIFICATION. Product name: Tris-HCl-SDS-2- Mercaptoethanol Product Number: 658
 SDS- Tris-HCl-SDS-2-Mercaptoethanol Page 1 of 8 Safety Data Sheet 1121 5 th Street NW Washington, DC 20001 www.edvotek.com Version 2 Revision Date 11/27/2013 1. PRODUCT AND COMPANY IDENTIFICATION Product
SDS- Tris-HCl-SDS-2-Mercaptoethanol Page 1 of 8 Safety Data Sheet 1121 5 th Street NW Washington, DC 20001 www.edvotek.com Version 2 Revision Date 11/27/2013 1. PRODUCT AND COMPANY IDENTIFICATION Product
Chem 110 General Principles of Chemistry
 CHEM110 Worksheet - Gases Chem 110 General Principles of Chemistry Chapter 9 Gases (pages 337-373) In this chapter we - first contrast gases with liquids and solids and then discuss gas pressure. - review
CHEM110 Worksheet - Gases Chem 110 General Principles of Chemistry Chapter 9 Gases (pages 337-373) In this chapter we - first contrast gases with liquids and solids and then discuss gas pressure. - review
ThermoSpas Hot Tub Glossary
 ThermoSpas Hot Tub Glossary Activator (Potassium Monopersulfate) - Also known as non-chlorine shock, it is a strong oxidizer capable of eliminating contaminants from your spa. It is a nonchlorine chemical
ThermoSpas Hot Tub Glossary Activator (Potassium Monopersulfate) - Also known as non-chlorine shock, it is a strong oxidizer capable of eliminating contaminants from your spa. It is a nonchlorine chemical
Composition of Exhaled Breath
 Composition of Exhaled Breath Exhaled Breath (~37 C) is mainly composed by gaseous substances: Water Vapor, CO 2, O 2, N 2, NO, CO, etc. Several volatile, low-volatile and non-volatile compounds of endogenous
Composition of Exhaled Breath Exhaled Breath (~37 C) is mainly composed by gaseous substances: Water Vapor, CO 2, O 2, N 2, NO, CO, etc. Several volatile, low-volatile and non-volatile compounds of endogenous
