CONTROL OF GILL VENTILATION AND AIR-BREATHING IN THE BOWFIN AMIA CALVA
|
|
|
- Mary Booth
- 5 years ago
- Views:
Transcription
1 The Journal of Experimental Biology 202, (1999) Printed in Great Britain The Company of Biologists Limited 1998 JEB CONTROL OF GILL VENTILATION AND AIR-BREATHING IN THE BOWFIN AMIA CALVA MICHAEL S. HEDRICK 1, * AND DAVID R. JONES 2 1 Department of Biological Sciences, California State University at Hayward, Hayward, CA 94542, USA and 2 Department of Zoology, University of British Columbia, Vancouver, British Columbia, Canada V6T 1Z4 * mhedrick@csuhayward.edu Accepted 19 October; published on WWW 7 December 1998 The purpose of this study was to investigate the roles of branchial and gas bladder reflex pathways in the control of gill ventilation and air-breathing in the bowfin Amia calva. We have previously determined that bowfin use two distinct air-breathing mechanisms to ventilate the gas bladder: type I air breaths are characterized by exhalation followed by inhalation, are stimulated by aquatic or aerial hypoxia and appear to regulate O 2 gas exchange; type II air breaths are characterized by inhalation alone and possibly regulate gas bladder volume and buoyancy. In the present study, we test the hypotheses (1) that gill ventilation and type I air breaths are controlled by O 2 -sensitive chemoreceptors located in the branchial region, and (2) that type II air breaths are controlled by gas bladder mechanosensitive stretch receptors. Hypothesis 1 was tested by examining the effects of partial or complete branchial denervation of cranial nerves IX and X to the gill arches on gill ventilation frequency (fg) and the proportion of type I air breaths during normoxia and hypoxia; hypothesis II was tested by gas bladder inflation and deflation. Following complete bilateral branchial denervation, fg did not differ from that of sham-operated control fish; in addition, fg was not significantly affected by aquatic hypoxia in sham-operated or denervated fish. In sham-operated fish, aquatic hypoxia significantly increased overall air-breathing frequency (fab) and the percentage of Summary type I breaths. In fish with complete IX X branchial denervation, fab was also significantly increased during aquatic hypoxia, but there were equal percentages of type I and type II air breaths. Branchial denervation did not affect the frequency of type I air breaths during aquatic hypoxia. Gas bladder deflation via an indwelling catheter resulted in type II breaths almost exclusively; furthermore, fab was significantly correlated with the volume removed from the gas bladder, suggesting a volume-regulating function for type II air breaths. These results indicate that chronic (3 4 weeks) branchial denervation does not significantly affect fg or type I air-breathing responses to aquatic hypoxia. Because type I air-breathing responses to aquatic hypoxia persist after IX X cranial nerve denervation, O 2 -sensitive chemoreceptors that regulate airbreathing may be carried in other afferent pathways, such as the pseudobranch. Gas bladder deflation reflexly stimulates type II breaths, suggesting that gas bladder volume-sensitive stretch receptors control this particular air-breathing mechanism. It is likely that type II air breaths function to regulate buoyancy when gas bladder volume declines during the inter-breath interval. Key words: hypoxia, air-breathing, gill ventilation, gas bladder, buoyancy, stretch receptor, bowfin, Amia calva. Introduction The bowfin Amia calva is a primitive actinopterygian fish with functional gills for aquatic respiration and a wellvascularized gas bladder for aerial ventilation. In response to aquatic hypoxia, Amia calva increase gill ventilation frequency and air-breathing, which serves to maintain oxygen homeostasis (Johansen et al., 1970; Randall et al., 1981; McKenzie et al., 1991; Hedrick and Jones, 1993). During air-breathing in Amia calva, gas flow is generated by a buccal force pump which has been described in detail using X-ray cine film and electromyographic analyses (Deyst and Liem, 1985). The latter study experimentally characterized the muscular basis for airbreathing that had been described by direct observations in earlier studies (Wilder, 1877; Johansen et al., 1970); specifically, that Amia calva breathe air by a mechanism that involves exhalation followed by an inhalation. Subsequent to those studies, Hedrick and Jones (1993) discovered a previously undescribed air-breathing mechanism in Amia calva that involves inhalation alone with no associated expiratory phase. In that study, the two air-breathing mechanisms were designated as type I and type II breaths. Type I breaths are characterized by the previously described exhalation inhalation sequence, and type II breaths are the newly described
2 88 M. S. HEDRICK AND D. R. JONES inhalation-only mechanism. Because type I breaths are preferentially stimulated by aquatic or aerial hypoxia (Hedrick and Jones, 1993), we hypothesized that type I breaths are regulated by O 2 -sensitive chemoreceptors (see Shelton et al., 1986). In our previous study, Amia calva exposed to aerial hyperoxia, regardless of aquatic oxygen levels, used predominantly type II breaths, which led us to hypothesize that type II breaths are regulated by gas bladder volume stretch receptor feedback (Milsom and Jones, 1985), rather than having an oxygen-acquisition function. The proximate function of type II breaths is thought to be adaptive for regulating buoyancy in the aquatic environment (Hedrick and Jones, 1993). The locations of the putative O 2 -sensitive chemoreceptors and the afferent pathways that mediate branchial and airbreathing reflexes in Amia calva and other air-breathing fishes are uncertain. Evidence from air-breathing and non-airbreathing fish (i.e. those that use exclusively branchial ventilation) suggests that hypoxic ventilatory reflexes are regulated by O 2 -sensitive chemoreceptors in the branchial region that monitor aquatic and/or intravascular P O (Shelton et al., 1986; Smatresk et al., 1986; Burleson et al., 1992). Because the afferent neural pathways from the presumed branchial locations for these chemoreceptors are carried in the glossopharyngeal (cranial nerve IX) and vagus (cranial nerve X) nerves, studies have attempted to abolish gill ventilatory responses to hypoxia by selective branchial denervation (Saunders and Sutterlin, 1971; Burleson and Smatresk, 1990); however, there is little information on the role of potential chemoreceptor pathways controlling air-breathing reflexes in fish (Smatresk et al., 1986; McKenzie et al., 1991). The purpose of the present study was to test in Amia calva the hypotheses (1) that increased gill ventilation and the frequency of type I air breaths during aquatic hypoxia are regulated by O 2 -sensitive chemoreceptors located in the branchial region with afferent pathways carried in cranial nerves IX and X; and (2) that type II air breaths are stimulated by reductions in gas bladder volume and are regulated by volume-sensitive gas bladder stretch receptors. Materials and methods Animals Twenty-two bowfin (Amia calva L.) were used in this study. Fish were netted in Lake Ontario, Canada, by commercial fishermen and air-freighted to the University of British Columbia, where they were kept indoors in large circular fibreglass tanks supplied with continuously running dechlorinated tap water that ranged in temperature from 6 to 13 C during the course of this study. Branchial denervation experiments Seventeen fish (mass 433±77 g, mean ± S.D.) were used for denervation experiments. The experiments were carried out from June to November. Surgical procedures were performed at the temperature at which the fish were normally maintained (10 13 C), but fish were acclimated to room temperature (20 22 C) for the experimental procedures. Fish were anaesthetized in 0.01 % tricaine methanesulphonate (MS-222; Sydell Laboratories, Vancouver, British Columbia, Canada) dissolved in dechlorinated water (ph ). When gill ventilation ceased, the fish was transferred to an operating table and artificially ventilated with a lower concentration of MS-222 (0.005 %) in oxygenated (100 % O 2 ) water. The branchial branches of cranial nerves IX (glossopharyngeal) and X (vagus) were exposed by lifting the operculum and cutting the thin membrane overlying the gill arches. Branches of nerve X innervate all four gill arches, whereas nerve IX innervates the first gill arch and pseudobranch (see Nilsson, 1984). Branches of cranial nerves IX and X to the four gill arches and pseudobranch were identified, separated from the surrounding fascia and cut with scissors. Bleeding was minimal and was controlled with cotton applicators. The procedure was performed bilaterally, and the membrane was closed with 3 4 sutures (3-0 silk). The entire procedure required approximately min to complete. The fish recovered in oxygenated, dechlorinated water until spontaneous gill ventilatory movements returned. Shamoperated control fish were treated in the same manner with anaesthetization and bilateral nerve exposure, but the nerves were not cut. After the procedure, the fish were returned to a holding tank maintained at the pre-surgical temperature (10 13 C) and allowed 3 4 weeks to recover. Protocol After the minimum 3 week recovery period, fish were brought into the laboratory in large (40 l) plastic containers at their normal holding temperature (10 13 C), and the water was allowed to warm slowly to room temperature (20 22 C) overnight while being continuously aerated. This acclimation procedure was complete by the following day (12 16 h), and the animals were held at room temperature for a total of h before being transferred to an aquarium for measurements. After acclimation, a fish was placed into an aquarium for h before any recordings were made. Airbreathing behaviour was recorded non-invasively by allowing the fish to breathe from a funnel located at the air water interface; air flowed continuously through the funnel at 200 ml min 1. Air-breathing was restricted to the funnel by blocking the aquarium surface with an acrylic cover. Air-breathing behaviour was recorded with a camera and video cassette recorder (JVC Canada, Scarsborough, Ontario, Canada) at 30 frames s 1 for each fish in 8 h sessions over a 3 day period with the following sequence: day 1, aquatic normoxia (P O >18.6 kpa); day 2, aquatic hypoxia (P O =6.52±0.13 kpa, mean ± S.E.M.); day 3, aquatic normoxia. After these sessions had been completed, the fish was killed by overanaesthetization with MS-222, and a post-mortem dissection was carried out to determine the extent of branchial denervation. Branchial nerves were identified and compared with a published description (Allis, 1897). Fish were categorised, on the basis of the dissection findings, as shamoperated control fish (SH; N=4), partially denervated (PD; N=5), in which nerve regrowth was indicated or not all the
3 Control of breathing in Amia calva 89 branchial branches had been cut, and total branchialdenervated fish (TD; N=8). Air-breathing events were counted directly from the video tape after each recording session; air breath type (type I or II) was characterized by specific ventilatory movements that were easily distinguished on video tape and were confirmed previously by pneumotachography (Hedrick and Jones, 1993). In the previous study, we found that expired ventilatory volume (V exp ) did not change in response to aquatic or aerial gas concentrations; therefore, V exp was not measured in this study. In the present study, pneumotachography was used to confirm type I or type II breaths that were not easily distinguished by direct observations from video tape. Gill ventilation frequency (fg) for each fish was counted directly from the video tape with a minimum 2 min recording used as a datum. Gill ventilation frequency was measured at least once, and as many as 20 times, during each 8 h recording session; however, a single mean value for each fish in each treatment was used in the analysis. It was often not possible to observe gill ventilatory movements if the fish was facing away from the camera or if the fish was moving excessively. Gas bladder inflation and deflation Five fish (mass 335±99 g, mean ± S.D.) were used for gas bladder inflation/deflation experiments. These experiments were carried out during January February when the water temperature was 6 8 C. Each fish was brought into the laboratory and acclimated to room temperature (20 22 C) as described above for h; after acclimation, each fish was anaesthetized with MS-222 and placed on a surgical table and artificially ventilated as described above. The glottis was identified in the dorsal gut wall and held open with forceps. A 50 cm length of polyethylene tubing (PE90; Intramedic) was introduced through the glottis and pneumatic duct and advanced approximately 3 5 cm into the gas bladder. The cannula was secured to the palate with suture (0 silk), passed through a grommet in the frontal bone and exited through the snout. The glottal aperture of Amia calva used in this study is approximately 5 mm in diameter (Davies et al., 1993); therefore, it is estimated that the total area of the glottis occluded by the cannula was less than 5 %. Following surgery, each fish was allowed to recover for 24 h in an aquarium containing continuously aerated, dechlorinated water at room temperature. Tests of gas bladder deflation and inflation were carried out with fish in normoxic (P O =19 21 kpa), hypoxic (P O =7 8 kpa) and hyperoxic (P O =36 40 kpa) water on separate days. The aquarium containing the fish was bubbled with oxygen/nitrogen mixtures from a gas-mixing pump (Wösthoff) until the desired P O was reached. The P O of the water was measured with a Clark-type oxygen electrode (Radiometer). Gas bladder volume manipulations were achieved with minimal stimulation from catheter handling or visual and vibrational disturbances. For inflation or deflation of the gas bladder, the catheter was connected to a plastic syringe, which was firmly secured to a flat surface near the aquarium, and a volume of gas was removed from, or injected into, the gas bladder. Following each deflation or inflation, air-breathing behaviour was observed for 10 min and recorded as the number of breaths taken as well as the type (type I or II) of breath taken by the fish. The walls of the aquarium were covered with an opaque material to within 5 cm of the water surface so that the fish would not be disturbed by visual inputs, but the experimenter could observe the air breath response at the surface. At least 30 min was allowed between deflation or inflation trials. Data analysis and statistics Values are presented as means ± S.E.M. unless stated otherwise. For gill denervation experiments (N=17), measurements during a single 8 h recording session included fg, the total number of air breaths, the breath type (type I or II) and the inter-breath interval (IBI, min) between type I air breaths. These measurements were obtained for each fish in the three groups (SH, PD and TD) during aquatic normoxia or hypoxia on each of the 3 days of recording. Data were analyzed using unpaired t-tests, and one-way and twoway analysis of variance (ANOVA) followed by the Student Newman Keuls (SNK) multiple-range test where significant differences were detected by ANOVA (Zar, 1974). Where Bartlett s test revealed a lack of homogeneity of variance, data were analyzed using a non-parametric ANOVA by ranks (Kruskal Wallis). Percentages of type I and type II air breaths for each fish were converted to their arcsine values before comparison using unpaired t-tests (Zar, 1974). For gas bladder deflations, least-squares linear regression was also used to examine the effects of gas bladder volume deflation on airbreathing frequency (fab). Significance was accepted at the 5 % level (P<0.05). All statistical comparisons were carried out using a commercially available statistical software package (GraphPad Prism version 2.0, San Diego, CA, USA). Results Gill ventilation and air-breathing responses to branchial denervation Table 1 summarizes the mean fg values recorded for surgically treated fish (SH, PD or TD) during aquatic normoxia and hypoxia. Gill ventilation frequency was significantly affected by branchial denervation (F 2,31 =9.6; P<0.002) and by Table 1. Mean gill ventilation frequency (cycles min 1 ) for sham-operated (SH; N=4), partially denervated (PD; N=5) and totally denervated (TD; N=8) Amia calva in normoxic (day 1 and day 3) and hypoxic water Normoxia (day 1) Hypoxia (day 2) Normoxia (day 3) SH 8.9± ± ±0.7 PD 12.2± ± ±0.5* TD 5.6± ± ±0.7 *Significantly different (P<0.05) from normoxia (day 1); significantly different (P<0.05) from PD fish during normoxia (day 1). Values are means ± S.E.M.
4 90 M. S. HEDRICK AND D. R. JONES Table 2. Mean air-breathing frequency (f AB ) and percentage of type I and type II breaths (of total breaths) for sham-operated (SH; N=4), partially denervated (PD; N=5) and totally denervated (TD; N=8) fish during normoxia (days 1 and 3) and hypoxia (day 2) Normoxia (day 1) Hypoxia (day 2) Normoxia (day 3) f AB Type I Type II f AB Type I Type II f AB Type I Type II (breaths h 1 ) (%) (%) (breaths h 1 ) (%) (%) (breaths h 1 ) (%) (%) SH 4.7± ± ±12.5* 9.7± ± ±9.7** 3.4± ± ±14.6 PD 3.5± ± ± ± ± ±5.3** 3.2± ± ±12.0 TD 9.1± ± ±6.9*** 12.6± ± ± ± ± ±8.1 *P<0.05; **P<0.02; ***P<0.001 compared with the percentage of type I breaths (unpaired t-test) for the same treatment. P<0.05 compared with both normoxic treatments (one-way ANOVA). P<0.05 compared with normoxia (day 1) in the same treatment. Values are means ± S.E.M. aquatic gas treatment (F 2,31 =5.3; P<0.05) owing to a reduction in fg in the second normoxic treatment for the PD fish (Table 1). fg in TD fish was depressed relative to that of PD fish during normoxia (day 1). In all three groups, there was no significant gill ventilatory response to aquatic hypoxia, and fg for TD fish did not differ from that of control (SH) fish (Table 1). Air-breathing frequency was approximately 3 9 breaths h 1 in all three groups during normoxia and increased significantly during aquatic hypoxia (Table 2). SH and TD fish in the first normoxic treatment (day 1) had a significantly greater proportion of type II breaths compared with type I breaths (P<0.05; unpaired t-test; Table 2), but in PD fish there was no difference between the numbers of type I and type II air breaths during normoxia. During aquatic hypoxia, SH and PD fish showed a significant change in the percentage of type I breaths relative to type II breaths (unpaired t-test; P<0.02). Both the increase in fab and an increased percentage of type I breaths during hypoxia have been noted previously in Amia calva (Hedrick and Jones, 1993). In contrast, TD fish showed no significant increase in the percentage of type I breaths during hypoxia (Table 2) despite the overall increase in fab compared with normoxia, and the increased frequency of type I breaths during hypoxia (Table 3). Direct Table 3. Mean interbreath interval (IBI; min) for type I air breaths for individual sham-operated (SH), partially denervated (PD) and totally denervated (TD) fish Fish Normoxia (day 1) P Hypoxia (day 2) P Normoxia (day 3) SH ±14.9 (41) ±5.9 (65) ±12.6 (23) ±14.4 (16) ±1.4 (69) ±6.0 (27) ±3.9 (51) ±23.3 (6) ±7.3 (39) ±56.6 (3) PD 1 117±6.4 (2) ±3.8 (38) ±13.7 (8) ±82.3 (10) ±4.8 (30) ±35.9 (3) ±5.6 (18) ±5.5 (3) ±4.3 (36)*** ±2.6 (56) NS 9.0±3.6 (54) ±28.4 (10) ±3.9 (36) NS 120±139 (2) TD ±21.9 (11) ±2.2 (59) ±29.6 (9) ±36.5 (4) ±4.4 (36) ±18.6 (9) ±9.8 (14) ±3.2 (41) ±12.5 (20) ±7.4 (22)* ±4.3 (34) NS 17.6±9.4 (25) ±9.2 (2) ±7.3 (17) ±15.2 (7) ±4.2 (22) ±34.3 (4) ±10.0 (31)* ±2.6 (53) NS 10.6±5.3 (45) ±4.0 (26) ±3.5 (37) ±10.1 (10) Significance level (P) between aquatic hypoxia IBI and both normoxic IBI values is given (Kruskal Wallis test). Unpaired t-test. *P<0.05; ***P<0.001 compared with normoxic (day 3) value (Kruskal Wallis test). Values are means ± S.D. (N). NS, no significant difference. A value of 0 indicates that the fish did not use type I breaths during the period of observation in this treatment.
5 Control of breathing in Amia calva 91 observations from the video tape analysis revealed that some TD fish had difficulty capturing and transferring inhaled air to the gas bladder during type I breaths, thus failing to fill the gas bladder sufficiently with inhaled air (see Discussion). The lack of an increased percentage of type I breaths in TD fish during hypoxia compared with SH and PD fish was masked by the larger percentage of type II breaths taken by five of the eight TD fish. For example, fab in one TD fish was approximately 6 breaths h 1 during normoxia and increased to approximately 9 breaths h 1 during hypoxia; however, the majority of these breaths (>75 %) were of type II. Table 3 shows the mean interbreath interval (IBI) for type I breaths for individual fish in each treatment. Two of the four SH fish and one of the five PD fish did not use any type I air breaths during the first normoxic treatment. IBI ranged from 13 to 117 min during normoxia and decreased to 7 28 min during hypoxia (Table 3). In all three groups, type I IBI decreased significantly during aquatic hypoxia (Kruskal Wallis test, Table 3), indicating a strong effect of aquatic hypoxia on type I fab. In only three of the 17 fish was IBI significantly greater during the first normoxic treatment compared with the second normoxic treatment, indicating that the hypoxic exposure on day 2 had little effect on the number or frequency of type I air breaths on day 3 (normoxia). Effects of gas bladder deflation and inflation Gas bladder deflation had a highly significant effect on fab in the five Amia calva used; fab was significantly correlated with the volume removed (V R ) from the gas bladder: fab=0.5v R +4.3 (Fig. 1; F 1,88 =35.7; P<0.001; r 2 =0.29). The overall slope of the regression (0.50±0.08) was significantly different from zero (t 88 =5.98; P<0.001). The effects of V R on fab during aquatic normoxia and hyperoxia were highly significant (normoxia: F 1,49 =16.2; P<0.001; hyperoxia: F 1,23 =15.0; P<0.001), with slopes of 0.38±0.1 and 0.54±0.14, fab (breaths h -1 ) V R (ml kg -1 ) Fig. 1. Air-breathing frequency fab (breaths h 1 ) as a function of the volume of gas removed V R (ml kg 1 ) from the gas bladder in Amia calva during aquatic normoxia (circles), hypoxia (squares) and hyperoxia (triangles). The solid line is the line of best fit using leastsquares linear regression analysis for all values (see Results). The relationship is significant at P< respectively. During aquatic hypoxia, however, V R did not have a significant effect on fab (F 1,11 =3.6; P<0.1) with a slope of 0.56±0.3. In every instance except one, regardless of aquatic P O, the initial response to deflation was a type II air breath, and the majority of these breaths (>50 %) were taken less than 1 min following deflation. Type I breaths were occasionally observed during the 10 min following deflation, but these occurred during aquatic hypoxia and were never the first or second breath in a series of air breaths. There appeared to be a volume threshold of approximately 3 ml kg 1 ; that is, changes to the volume of the gas bladder below this threshold volume did not elicit air-breathing. Large deflations typically caused the fish to sink to the bottom of the aquarium, and they then struggled to reach the surface to take an air breath, thus indicating qualitatively that deflation caused the fish to become negatively buoyant. Gas bladder inflation above the apparent volume threshold typically prevented an air breath attempt if the animal was approaching the water surface. Large inflations caused fish to rise rapidly towards the surface because of increased positive buoyancy and usually resulted in excess air being expelled from the mouth before the fish reached the air water interface. This allowed the fish to attain neutral or negative buoyancy and to return to the bottom of the aquarium. Discussion The important findings from this study are (1) that fg is depressed by complete chronic branchial denervation, but is not significantly altered by exposure to aquatic hypoxia; (2) that chronic total or partial branchial denervation does not abolish or attenuate type I breaths during aquatic hypoxia, suggesting that other branchial sites, such as the pseudobranch, may be responsible for the hypoxic ventilatory responses after complete branchial denervation; and (3) that deflation of the gas bladder resulted in type II breaths almost exclusively, and fab was positively correlated with the volume of gas removed from the gas bladder, suggesting a role for volume-sensitive gas bladder stretch receptors in regulating type II air breaths. Role of branchial reflex pathways in ventilation Afferent nerve activity from the first gill arch of teleosts has been shown to increase in response to hypoxia, low ph or chemical stimulants such as sodium cyanide (NaCN) (Laurent and Rouzeau, 1972; Milsom and Brill, 1986; Burleson and Milsom, 1990, 1993; Burleson et al., 1992). The afferent activity from the gill arches, carried in glossopharyngeal (IX) and vagal (X) pathways, is presumed to mediate the cardiovascular and ventilatory responses to hypoxia in conscious fish; however, denervation of these afferent branchial pathways has generally failed to abolish the ventilatory responses to hypoxia (Hughes and Shelton, 1962; Saunders and Sutterlin, 1971; Randall and Jones, 1973), suggesting that extrabranchial sites, possibly in the central nervous system (CNS), initiate hypoxia-induced ventilatory responses (Bamford, 1974; Jones, 1983). At present, there is no evidence to support a role for CNS chemoreceptors in
6 92 M. S. HEDRICK AND D. R. JONES mediating hypoxic or hypercapnic ventilatory reflexes in airbreathing or non-air-breathing fish (Graham et al., 1990; Hedrick et al., 1991). More recent experiments have shown that gill ventilation and air-breathing responses to hypoxia can be eliminated by complete branchial denervation combined with pseudobranch ablation (Burleson and Smatresk, 1990; McKenzie et al., 1991). Burleson and Smatresk (1990) reported that complete branchial denervation in channel catfish Ictalurus punctatus, a species that lacks a pseudobranch, was necessary to abolish hypoxic ventilatory reflexes, supporting the hypothesis that O 2 -sensitive chemoreceptors are localized peripherally. Their study also indicated that multiple branchial chemoreceptor sites are present because failure to denervate catfish completely resulted in an intact hypoxic ventilatory response. A similar study with Amia calva indicated that complete branchial denervation combined with pseudobranch ablation could abolish air-breathing responses to relatively brief (15 min) exposures to aquatic hypoxia and attenuate increases in gill ventilation frequency and opercular amplitude (McKenzie et al., 1991). Gill ventilation frequency was similar for fish in the present study to that in the study of McKenzie et al. (1991), despite differences in methodology. The present study used a noninvasive measurement of fg, whereas McKenzie et al. (1991) used direct opercular pressure measurements. Both studies also indicate that fg during normoxia is reduced by complete branchial denervation. We did not observe any changes in fg for control or denervated fish upon exposure to hypoxia, which contrasts with the significant increase in both fg and opercular amplitude in sham-operated fish in the study of McKenzie et al. (1991). Their study also showed that fg and opercular amplitude were attenuated, but not abolished, by branchial denervation. One drawback of the present study is that we were only able to measure fg and could not measure an index of ventilation volume, such as opercular pressure. It is therefore possible that fish in the present study increased their ventilation volume in response to hypoxia or relied more heavily on ventilation volume than on changes in opercular amplitude during hypoxia under the experimental conditions in this study. There are other important methodological differences between the present study and the study by McKenzie et al. (1991) that may also explain differences in air-breathing responses to hypoxia in sham-operated and denervated fish. One major difference between the two studies is the type of apparatus used to hold fish for measurements. The fish in the study by McKenzie et al. (1991) were placed into a smaller, shallow box with a forward air space for air-breathing similar to the type used in a previous study on the role of central chemoreceptors in air-breathing Amia calva (Hedrick et al., 1991). Although this type of apparatus is necessary for studies in which measurements of opercular and blood pressures are made via indwelling catheters, there is little room for movement by the fish. This might affect the type of air breaths used by the fish, particularly if buoyancy control is unnecessary in shallow water. For example, SH fish in our study had an overall fab of 3 5 breaths h 1 during aquatic normoxia and of approximately 10 breaths h 1 during aquatic hypoxia; these values are approximately twice the fab values reported for SH fish by McKenzie et al. (1991) or Hedrick et al. (1991) under similar conditions. A significant percentage of these breaths were of type II in the present study, particularly during normoxia (Table 2), indicating that, when fish are allowed more access to movement in a larger container, overall fab may be higher. If we remove type II breaths from our analysis, fab values in these studies are similar. The fish in the present study were allowed a longer recovery period (3 4 weeks) following surgery than the 48 h recovery time allowed in the study by McKenzie et al. (1991), which may have enabled alternative pathways (i.e. metabolic) or other cues to develop which could regulate hypoxic ventilatory responses. For example, McKenzie et al. (1991) found that branchially denervated Amia calva exposed to aquatic hypoxia showed a significant increase in plasma catecholamine concentration and that catecholamine infusion increased fg, but not fab. Although we did not measure catecholamine levels in the present study, there may be differences in the amount of stress (as indicated by catecholamine levels) experienced by the fish in the two studies, and this may in turn have had some impact on hypoxic ventilatory responses. Exposure to hypoxia was relatively brief (15 min) in the study by McKenzie et al. (1991), which may not have allowed full expression of airbreathing responses to hypoxia. In the present study, the mean IBI for type I air breaths for hypoxic TD fish was 14.6±2.8 min (mean ± S.E.M., N=8). Thus, relatively short hypoxic exposures may not allow observations of air-breathing responses in denervated fish, particularly if type I air breaths are used predominantly. Finally, and perhaps most importantly, we did not remove the pseudobranch from the Amia calva in our study, although the innervation from cranial nerve IX to the pseudobranch was cut. There is some evidence that the pseudobranch in some species is innervated by cranial nerve VII (Nilsson, 1984); thus, the possibility exists that some peripheral chemoreceptive tissue remained intact in our fish. Given the lack of evidence for CNS sites that could modulate air-breathing responses to hypoxia (Hedrick et al., 1991), it is likely that an intact pseudobranch allows expression of hypoxic ventilatory reflexes. The denervation procedure in our study appeared to have a detrimental effect on air-breathing behaviour and ventilatory mechanics that has not been noted previously. Our observations from video recordings of air breathing indicated that some TD fish could not capture and transfer inhaled air to the gas bladder in a normal fashion. More specifically, as inhaled air was transferred from the buccal cavity to the gas bladder, most was lost through the opercular cavity. Under normal circumstances, a small amount of gas is lost in this way, and this small loss of inhaled gas appears to be an important source of variation that masks an underlying periodicity present in type I air breaths (Hedrick et al., 1994). The failure to transfer inhaled gas adequately in denervated fish is probably due to the loss of afferent information from the gill arches. A significant amount of mechanoreceptive information
7 Control of breathing in Amia calva 93 is contained in gill arch afferents that transduce information about gill position and deformation (see Burleson et al., 1992). Our observations suggest that some aspect of this afferent information is necessary for the normal expression of the motor control involved in air-breathing behaviour; however, this remains to be tested experimentally. The failure to transfer inhaled air adequately to the gas bladder in TD fish resulted in a larger number of type II breaths (inhalation only) and a lack of an increase in the percentage of type I breaths during hypoxia (Table 2). In TD fish, stimulation of type I breaths by hypoxia (Table 3) caused qualitatively greater losses of inhaled gas compared with SH or PD fish, resulting in an increased frequency of type II breaths to restore gas bladder volume. This lends indirect support to our hypothesis that type II breaths function to maintain gas bladder volume within normal limits (see below). Expired ventilation volume (V exp ) was not measured for type I air breaths in the present study, but in a previous study on this species we found that V exp was unaffected by aquatic or aerial hypoxia (Hedrick and Jones, 1993). Thus, fab and the relative proportions of type I or type II breaths make a more important contribution to minute ventilation than does V exp. However, inhaled volume is the more important parameter with respect to both gas exchange and buoyancy regulation. Our observations that TD fish lost substantial amounts of inhaled gas through the opercular cavity would not be confirmed by measurements of V exp. From our observations, it was clear that large losses of gas resulted in decreased buoyancy and an increased proportion of type II air breaths during hypoxia (Table 2). Gas bladder volume regulation of air-breathing It is known that gas bladder deflation stimulates, and inflation inhibits, air-breathing responses in Amia calva (Johansen et al., 1970), gar Lepisosteus osseus (Smatresk and Cameron, 1982) and lungfish Protopterus annectens (Pack et al., 1992). The response is analogous to the Breuer Hering inspiratory-terminating reflex in mammals. These species have been shown to have slowly adapting and/or rapidly adapting mechanoreceptors that respond to volume changes in the gas bladder (DeLaney et al., 1983; Milsom and Jones, 1985; Smatresk and Azizi, 1987). Because these primitive airbreathing species are dependent upon gas bladder volume for buoyancy regulation, in addition to the role of the gas bladder as a gas exchanger, it is reasonable to assume that volumesensitive gas bladder stretch receptors provide information that is integrated into the overall air-breathing behaviour. The present study demonstrated a clear functional role for volumesensitive feedback in the regulation of air-breathing in Amia calva: reductions in gas bladder volume result in an increased frequency of type II breaths and a restoration of gas bladder volume (Fig. 1). Afferent information from gas bladder stretch receptors in Amia calva is carried in the ramus intestinalis branch of the vagus nerve; nerve fibre discharge is slowly adapting, increases in response to inflation and decreases or ceases in response to deflation (Milsom and Jones, 1985). Moreover, afferent nerve discharge is proportional to gas bladder volume in Amia calva; therefore, our observation that the increased frequency of type II breaths is also proportional to the reduction in gas bladder volume can be explained by the characteristics of gas bladder mechanoreceptor discharge. Our interpretation of the role of type II breaths in regulating gas bladder volume and thus playing an important role in buoyancy control is supported by a number of observations. First, Amia calva with bilateral denervation of the ramus intestinalis nerve fail to control buoyancy (Hedrick, 1991). Second, Amia calva exposed to aerial hyperoxia (100 % O 2 ) switch to exclusive use of type II breaths (Hedrick and Jones, 1993). Our interpretation of the latter result is that, when the contents of the gas bladder have a very high P O, there is greater diffusion of O 2 into venous blood, resulting in a high blood P O downstream from the gas bladder. Because O 2 from venous blood would diffuse across the gills into the water, there would be a continuous flux of O 2 from the gas bladder to the venous blood. The high blood P O under these conditions presumably silences the oxygen chemoreceptors that regulate type I breaths, yet diffusion of O 2 from the gas bladder to the blood will still result in a reduction in gas bladder volume. Third, we have estimated, using gas bladder and tidal volume data from Hedrick and Jones (1993), that a typical type II air breath would increase gas bladder P O by approximately 0.5 kpa during aerial normoxia. This small change in P O is unlikely to be detected by intravascular oxygen-sensitive chemoreceptors (Burleson et al., 1992); therefore, type II breaths are unlikely to contribute significantly to gas exchange. Alternatively, type I breaths, which are of larger volume and exchange approximately 30 % of the gas bladder contents with each breath, are sufficient to maintain the P O of approximately 7 11 kpa measured in the gas bladder of Amia calva (Johansen et al., 1970; Hedrick, 1991). Finally, results from computer model simulations (Hedrick and Katz, 1991) with firing thresholds for P O and gas bladder volume that regulate type I and type II breaths, respectively, produce realistic air-breathing periodicities that are similar to those observed from Amia calva during aquatic normoxia and hypoxia (Hedrick et al., 1994). Taken together, these observations indicate that the proximate function of type II breaths is to regulate gas bladder volume; volume regulation of the gas bladder is clearly important for maintaining buoyancy in the aquatic environment. Evolutionary implications Our current view of air-breathing in Amia calva is that this species evolved two air-breathing mechanisms for coping with the conflicting demands of buoyancy and gas exchange that occur when a single organ is used for both functions. This conflict arises because an effective gas exchanger requires high rates of diffusional exchange across its respiratory surface, whereas the effectiveness of a buoyancy-control organ relies on minimizing diffusional gas flux. Any primitive lung, or gasfilled structure, automatically imposes a change in buoyancy on the animal whether or not the inhaled gas is involved with gas exchange and metabolic demands. Amia calva appears to
8 94 M. S. HEDRICK AND D. R. JONES have evolved a strategy in which gas exchange is regulated by type I breaths, whereas the buoyancy/volume function of the gas bladder is regulated by type II breaths which simply replace the gas lost by diffusion across the respiratory surface to the blood. Although it is not known whether other airbreathing fishes have similar mechanisms to cope with the conflicting demands of gas exchange and buoyancy, it appears that buoyancy regulation has been a strong selection pressure for the development of type II air-breathing behaviour in Amia calva. We thank Dr C. Wood and S. Munger and for supplying the Amia calva used in this study. We thank Drs S. Katz and J. Graham for useful discussions related to this work and the input of J. Graham who read an earlier version of the manuscript. We are also grateful to an anonymous reviewer who improved the quality of the manuscript. This work was supported by an NSERC (Canada) operating grant to D.R.J. References Allis, E. P. (1897). The cranial muscles and cranial and first spinal nerves in Amia calva. J. Morph. 12, Bamford, O. S. (1974). Respiratory neurones in rainbow trout (Salmo gairdneri). Comp. Biochem. Physiol. 48A, Burleson, M. L. and Milsom, W. K. (1990). Propranolol inhibits O 2 receptor activity in trout. Am. J. Physiol. 258, R1089 R1091. Burleson, M. L. and Milsom, W. K. (1993). Sensory receptors in the first gill arch of rainbow trout. Respir. Physiol. 93, Burleson, M. L. and Smatresk, N. J. (1990). Effects of sectioning cranial nerves IX and X on cardiovascular and ventilatory reflex responses to hypoxia and NaCN in channel catfish. J. Exp. Biol. 154, Burleson, M. L., Smatresk, N. J. and Milsom, W. K. (1992). Afferent inputs associated with cardioventilatory control in fish. In Fish Physiology, vol. XIIB (ed. W. S. Hoar, D. J. Randall and A. P. Farrell), pp Orlando: Academic Press. Davies, P. J., Hedrick, M. S. and Jones, D. R. (1993). Neuromuscular control of the glottis in a primitive air-breathing fish, Amia calva. Am. J. Physiol. 264, R204 R210. DeLaney, R. G., Laurent, P., Galante, R., Pack, A. I. and Fishman, A. P. (1983). Pulmonary mechanoreceptors in the dipnoi lungfish Protopterus and Lepidosiren. Am. J. Physiol. 244, R418 R428. Deyst, K. A. and Liem, K. F. (1985). The muscular basis of aerial ventilation of the primitive lung of Amia calva. Respir. Physiol. 59, Graham, M. S., Turner, J. D. and Wood, C. M. (1990). Control of ventilation in the hypercapnic skate Raja ocellata. I. Blood and extradural fluid. Respir. Physiol. 80, Hedrick, M. S. (1991). Air-breathing in the bowfin (Amia calva L.). PhD thesis, University of British Columbia. 174pp. Hedrick, M. S., Burleson, M. L., Jones, D. R. and Milsom, W. K. (1991). An examination of central chemosensitivity in an airbreathing fish (Amia calva). J. Exp. Biol. 155, Hedrick, M. S. and Jones, D. R. (1993). The effects of altered aquatic and aerial respiratory gas concentrations on air-breathing patterns in a primitive fish (Amia calva). J. Exp. Biol. 181, Hedrick, M. S. and Katz, S. L. (1991). A model of aerial ventilation in an air-breathing fish (Amia calva). Am. Zool. 31, 68A. Hedrick, M. S., Katz, S. L. and Jones, D. R. (1994). Periodic airbreathing behaviour in a primitive fish revealed by spectral analysis. J. Exp. Biol. 197, Hughes, G. M. and Shelton, G. (1962). Respiratory mechanisms and their nervous control in fish. Adv. Comp. Physiol. Biochem. 1, Johansen, K., Hanson, D. and Lenfant, C. (1970). Respiration in the primitive air-breather Amia calva. Respir. Physiol. 9, Jones, D. R. (1983). Ontogeny and phylogeny of the oxygen response. Proc. Physiol. Soc. N.Z. 3, Laurent, P. and Rouzeau, J. D. (1972). Afferent neural activity from the pseudobranch of teleosts. Respir. Physiol. 14, McKenzie, D. J., Burleson, M. L. and Randall, D. J. (1991). The effects of branchial denervation and pseudobranch ablation on cardioventilatory control in an air-breathing fish. J. Exp. Biol. 161, Milsom, W. K. and Brill, R. W. (1986). Oxygen sensitive afferent information arising from the first gill arch of yellowfin tuna. Respir. Physiol. 66, Milsom, W. K. and Jones, D. R. (1985). Characteristics of mechanoreceptors in the air-breathing organ of the holostean fish, Amia calva. J. Exp. Biol. 117, Nilsson, S. (1984). Innervation and pharmacology of the gills. In Fish Physiology, vol. XA (ed. W. S. Hoar and D. J. Randall), pp New York: Academic Press. Pack, A. I., Galante, R. J. and Fishman, A. I. (1992). Role of lung inflation of air breath duration in African lungfish (Protopterus annectens). Am. J. Physiol. 262, R879 R884. Randall, D. J., Cameron, J. N., Daxboeck, C. and Smatresk, N. J. (1981). Aspects of bimodal gas exchange in the bowfin Amia calva L. (Actinopterygii: Amiiformes). Respir. Physiol. 43, Randall, D. J. and Jones, D. R. (1973). The effect of deafferentation of the pseudobranch on the respiratory responses to hypoxia and hyperoxia in the trout (Salmo gairdneri). Respir. Physiol. 17, Saunders, R. L. and Sutterlin, A. M. (1971). Cardiac and respiratory responses to hypoxia in the sea raven, Hemitripterus americanus and an investigation of possible control mechanisms. J. Fish. Res. Bd Can. 28, Shelton, G., Jones, D. R. and Milsom, W. K. (1986). Control of breathing in ectothermic vertebrates. In Handbook of Physiology, section 3, The Respiratory System, vol. II, Control of Breathing, part 2 (ed. A. P. Fishman, N. S. Cherniack, J. G. Widdicombe and S. R. Geiger), pp Bethesda, MD: American Physiological Society. Smatresk, N. J. and Azizi, S. Q. (1987). Characteristics of lung mechanoreceptors in spotted gar, Lepisosteus oculatus. Am. J. Physiol. 252, R1066 R1072. Smatresk, N. J., Burleson, M. L. and Azizi, S. Q. (1986). Chemoreflexive responses to hypoxia and NaCN in longnose gar: evidence for two chemoreceptor loci. Am. J. Physiol. 251, R116 R125. Smatresk, N. J. and Cameron, J. N. (1982). Respiration and acid base physiology of the spotted gar, a bimodal breather. III. Response to a transfer from fresh water to 50 % sea water and control of ventilation. J. Exp. Biol. 96, Wilder, B. G. (1877). On the respiration of Amia. Proc. Am. Acad. Advmt Sci. 26, Zar, J. H. (1974). Biostatistical Analysis. Englewood Cliffs, NJ: Prentice-Hall, Inc.
THE EFFECTS OF ALTERED AQUATIC AND AERIAL RESPIRATORY GAS CONCENTRATIONS ON AIR-BREATHING PATTERNS IN A PRIMITIVE FISH (AMIA CALVA)
 J. exp. Biol. 181, 81 94 (1993) Printed in Great Britain The Company of Biologists Limited 1993 81 THE EFFECTS OF ALTERED AQUATIC AND AERIAL RESPIRATORY GAS CONCENTRATIONS ON AIR-BREATHING PATTERNS IN
J. exp. Biol. 181, 81 94 (1993) Printed in Great Britain The Company of Biologists Limited 1993 81 THE EFFECTS OF ALTERED AQUATIC AND AERIAL RESPIRATORY GAS CONCENTRATIONS ON AIR-BREATHING PATTERNS IN
THE EFFECTS OF BRANCHIAL DENERVATION AND PSEUDOBRANCH ABLATION ON CARDIOVENTILATORY CONTROL IN AN AIR-BREATHING FISH
 J. exp. Biol. 161, 347-365 (1991) 347 Printed in Great Britain The Company of Biologists Limited 1991 THE EFFECTS OF BRANCHIAL DENERVATION AND PSEUDOBRANCH ABLATION ON CARDIOVENTILATORY CONTROL IN AN AIR-BREATHING
J. exp. Biol. 161, 347-365 (1991) 347 Printed in Great Britain The Company of Biologists Limited 1991 THE EFFECTS OF BRANCHIAL DENERVATION AND PSEUDOBRANCH ABLATION ON CARDIOVENTILATORY CONTROL IN AN AIR-BREATHING
AN EXAMINATION OF CENTRAL CHEMOSENSITIVITY IN AN AIR-BREATHING FISH (AMIA CALVA)
 J. exp. Biol. 155, 165-174 (1991) 165 Printed in Great Britain The Company of Biologists Limited 1991 AN EXAMINATION OF CENTRAL CHEMOSENSITIVITY IN AN AIR-BREATHING FISH (AMIA CALVA) BY M. S. HEDRICK,
J. exp. Biol. 155, 165-174 (1991) 165 Printed in Great Britain The Company of Biologists Limited 1991 AN EXAMINATION OF CENTRAL CHEMOSENSITIVITY IN AN AIR-BREATHING FISH (AMIA CALVA) BY M. S. HEDRICK,
THE EFFECT OF CHANGES IN BLOOD OXYGEN- CARRYING CAPACITY ON VENTILATION VOLUME IN THE RAINBOW TROUT (SALMO GAIRDNERI)
 J. exp. Biol. (1982), 97, 325~334 325 With 2 figures Printed in Great Britain THE EFFECT OF CHANGES IN BLOOD OXYGEN- CARRYING CAPACITY ON VENTILATION VOLUME IN THE RAINBOW TROUT (SALMO GAIRDNERI) BY FRANK
J. exp. Biol. (1982), 97, 325~334 325 With 2 figures Printed in Great Britain THE EFFECT OF CHANGES IN BLOOD OXYGEN- CARRYING CAPACITY ON VENTILATION VOLUME IN THE RAINBOW TROUT (SALMO GAIRDNERI) BY FRANK
Respiratory Physiology. Adeyomoye O.I
 Respiratory Physiology By Adeyomoye O.I Outline Introduction Hypoxia Dyspnea Control of breathing Ventilation/perfusion ratios Respiratory/barometric changes in exercise Intra-pulmonary & intra-pleural
Respiratory Physiology By Adeyomoye O.I Outline Introduction Hypoxia Dyspnea Control of breathing Ventilation/perfusion ratios Respiratory/barometric changes in exercise Intra-pulmonary & intra-pleural
THE EFFECTS OF REDUCED GILL AREA AND HYPEROXIA ON THE OXYGEN CONSUMPTION AND SWIMMING SPEED OF RAINBOW TROUT
 J. exp. Biol. 127, 349-354 (1987) 349 f Great Britain The Company of Biologists Limited 1987 THE EFFECTS OF REDUCED GILL AREA AND HYPEROXIA ON THE OXYGEN CONSUMPTION AND SWIMMING SPEED OF RAINBOW TROUT
J. exp. Biol. 127, 349-354 (1987) 349 f Great Britain The Company of Biologists Limited 1987 THE EFFECTS OF REDUCED GILL AREA AND HYPEROXIA ON THE OXYGEN CONSUMPTION AND SWIMMING SPEED OF RAINBOW TROUT
Extrabranchial chemoreceptors involved in respiratory reflexes in the neotropical fish Colossoma macropomum (the tambaqui)
 The Journal of Experimental Biology 25, 1765 1774 (22) Printed in Great Britain The ompany of Biologists Limited JEB43 1765 Extrabranchial chemoreceptors involved in respiratory reflexes in the neotropical
The Journal of Experimental Biology 25, 1765 1774 (22) Printed in Great Britain The ompany of Biologists Limited JEB43 1765 Extrabranchial chemoreceptors involved in respiratory reflexes in the neotropical
Figure 1. A schematic diagram of the human respiratory system.
 Introduction to Respiration In this experiment, you will investigate various aspects of normal breathing, hyperventilation, rebreathing the effect of changing airway resistance and ways in which to measure
Introduction to Respiration In this experiment, you will investigate various aspects of normal breathing, hyperventilation, rebreathing the effect of changing airway resistance and ways in which to measure
Lesson 28. Function - Respiratory Pumps in Air Breathers Buccal Force Pump Aspiration Pump - Patterns of Gas Transfer in Chordates
 Lesson 28 Lesson Outline: Evolution of Respiratory Mechanisms - Air Breathers Form - Accessory Air Breathing Organs Facultative vs Obligate - Lungs Function - Respiratory Pumps in Air Breathers Buccal
Lesson 28 Lesson Outline: Evolution of Respiratory Mechanisms - Air Breathers Form - Accessory Air Breathing Organs Facultative vs Obligate - Lungs Function - Respiratory Pumps in Air Breathers Buccal
POSTILLA PEABODY MUSEUM YALE UNIVERSITY NUMBER AUGUST LUNG VENTILATION IN DIPNOAN FISHES KEITH STEWART THOMSON
 POSTILLA PEABODY MUSEUM YALE UNIVERSITY NUMBER 122. 5 AUGUST 1968. LUNG VENTILATION IN DIPNOAN FISHES KEITH STEWART THOMSON POSTILLA Published by the Peabody Museum of Natural History, Yale University
POSTILLA PEABODY MUSEUM YALE UNIVERSITY NUMBER 122. 5 AUGUST 1968. LUNG VENTILATION IN DIPNOAN FISHES KEITH STEWART THOMSON POSTILLA Published by the Peabody Museum of Natural History, Yale University
Roger S. Seymour Æ Anthony P. Farrell Æ Keith Christian Æ Timothy D. Clark Æ Michael B. Bennett Æ Rufus M. G. Wells Æ John Baldwin
 DOI 10.1007/s00360-007-0156-5 ORIGINAL PAPER Continuous measurement of oxygen tensions in the air-breathing organ of Pacific tarpon (Megalops cyprinoides) in relation to aquatic hypoxia and exercise Roger
DOI 10.1007/s00360-007-0156-5 ORIGINAL PAPER Continuous measurement of oxygen tensions in the air-breathing organ of Pacific tarpon (Megalops cyprinoides) in relation to aquatic hypoxia and exercise Roger
RESPIRATION AND ACID-BASE PHYSIOLOGY OF THE SPOTTED GAR, A BIMODAL BREATHER
 J. exp. Biol. (1982), 96, 295-306 295 With 3 figures Printed in Great Britain RESPIRATION AND ACID-BASE PHYSIOLOGY OF THE SPOTTED GAR, A BIMODAL BREATHER III. RESPONSE TO A TRANSFER FROM FRESH WATER TO
J. exp. Biol. (1982), 96, 295-306 295 With 3 figures Printed in Great Britain RESPIRATION AND ACID-BASE PHYSIOLOGY OF THE SPOTTED GAR, A BIMODAL BREATHER III. RESPONSE TO A TRANSFER FROM FRESH WATER TO
(Received 16 January 1946)
 186 J. Physiol. (I946) I05, I86-I90 6I2.2I5.9 THE ABSORPTION OF FLUIDS FROM THE LUNGS BY F. C. COURTICE AND P. J. PHIPPS From the Experimental Station, Porton and the Laboratory of Physiology, Oxford (Received
186 J. Physiol. (I946) I05, I86-I90 6I2.2I5.9 THE ABSORPTION OF FLUIDS FROM THE LUNGS BY F. C. COURTICE AND P. J. PHIPPS From the Experimental Station, Porton and the Laboratory of Physiology, Oxford (Received
The physiological functions of respiration and circulation. Mechanics. exercise 7. Respiratory Volumes. Objectives
 exercise 7 Respiratory System Mechanics Objectives 1. To explain how the respiratory and circulatory systems work together to enable gas exchange among the lungs, blood, and body tissues 2. To define respiration,
exercise 7 Respiratory System Mechanics Objectives 1. To explain how the respiratory and circulatory systems work together to enable gas exchange among the lungs, blood, and body tissues 2. To define respiration,
RESPIRATORY REGULATION DURING EXERCISE
 RESPIRATORY REGULATION DURING EXERCISE Respiration Respiration delivery of oxygen to and removal of carbon dioxide from the tissue External respiration ventilation and exchange of gases in the lung Internal
RESPIRATORY REGULATION DURING EXERCISE Respiration Respiration delivery of oxygen to and removal of carbon dioxide from the tissue External respiration ventilation and exchange of gases in the lung Internal
SHORT COMMUNICATION EFFECT OF EXPERIMENTAL VENTILATION OF THE SKIN ON CUTANEOUS GAS EXCHANGE IN THE BULLFROG
 J. exp. Biol. 121, 445-449 (1986) 445 Printed in Great Britain The Company of Biologists Limited 1986 SHORT COMMUNICATION EFFECT OF EXPERIMENTAL VENTILATION OF THE SKIN ON CUTANEOUS GAS EXCHANGE IN THE
J. exp. Biol. 121, 445-449 (1986) 445 Printed in Great Britain The Company of Biologists Limited 1986 SHORT COMMUNICATION EFFECT OF EXPERIMENTAL VENTILATION OF THE SKIN ON CUTANEOUS GAS EXCHANGE IN THE
OXYGEN UPTAKE IN A SPONTANEOUSLY VENTILATING, BLOOD-PERFUSED TROUT PREPARATION
 J. exp. Biol. (198a), 101, 35-45 35 K'th 3 figures Wrmted in Great Britain OXYGEN UPTAKE IN A SPONTANEOUSLY VENTILATING, BLOOD-PERFUSED TROUT PREPARATION BY CHARLES DAXBOECKI, PETER S. DAVIE*, STEVE F.
J. exp. Biol. (198a), 101, 35-45 35 K'th 3 figures Wrmted in Great Britain OXYGEN UPTAKE IN A SPONTANEOUSLY VENTILATING, BLOOD-PERFUSED TROUT PREPARATION BY CHARLES DAXBOECKI, PETER S. DAVIE*, STEVE F.
Physiology of Respiration
 Physiology of Respiration External Respiration = pulmonary ventilation breathing involves 2 processes: inspiration expiration Inspiration an active process involves contraction of diaphragm innervated
Physiology of Respiration External Respiration = pulmonary ventilation breathing involves 2 processes: inspiration expiration Inspiration an active process involves contraction of diaphragm innervated
Lesson 27. Lesson Outline: Phylogenetic Trends in Respiratory System Form and Function
 Lesson 27 Lesson Outline: Phylogenetic Trends in Respiratory System Form and Function Objectives: Throughout the course what you need to master is an understanding of: 1) the form and function of structures,
Lesson 27 Lesson Outline: Phylogenetic Trends in Respiratory System Form and Function Objectives: Throughout the course what you need to master is an understanding of: 1) the form and function of structures,
AIR-BREATHING DURING ACTIVITY IN THE FISHES AMIA CALVA AND LEPISOSTEUS OCULATUS
 The Journal of Experimental Biology 2, 943 948 (998) Printed in Great Britain The Company of Biologists Limited 998 JEB32 943 AIR-BREATHING DURING ACTIVITY IN THE FISHES AMIA CALVA AND LEPISOSTEUS OCULATUS
The Journal of Experimental Biology 2, 943 948 (998) Printed in Great Britain The Company of Biologists Limited 998 JEB32 943 AIR-BREATHING DURING ACTIVITY IN THE FISHES AMIA CALVA AND LEPISOSTEUS OCULATUS
Gas Exchange in Animals. Uptake of O2 from environment and discharge of CO2. Respiratory medium! water for aquatic animals, air for terrestial
 Gas Exchange in Animals Uptake of O2 from environment and discharge of CO2 Respiratory medium! water for aquatic animals, air for terrestial Respiratory surface! skin, gills, lungs Circulatory System O2/CO2
Gas Exchange in Animals Uptake of O2 from environment and discharge of CO2 Respiratory medium! water for aquatic animals, air for terrestial Respiratory surface! skin, gills, lungs Circulatory System O2/CO2
Life 23 - Respiration in Air Raven & Johnson Ch. 53 (part)
 1 Life 23 - Respiration in Air Raven & Johnson Ch. 53 (part) Objectives 1: Compare the properties of air and water as media for respiration, and the consequences for the evolution of respiratory systems
1 Life 23 - Respiration in Air Raven & Johnson Ch. 53 (part) Objectives 1: Compare the properties of air and water as media for respiration, and the consequences for the evolution of respiratory systems
Human Biology Respiratory System
 Human Biology Respiratory System Respiratory System Responsible for process of breathing Works in cooperation with Circulatory system Three types: 1. Internal Respiration 2. External Respiration 3. Cellular
Human Biology Respiratory System Respiratory System Responsible for process of breathing Works in cooperation with Circulatory system Three types: 1. Internal Respiration 2. External Respiration 3. Cellular
RESPIRATION IN AN AIR-BREATHING FISH, THE CLIMBING PERCH, ANABAS TESTUDINEUS
 J. Exp. Biol. (9), S3, 2-29 2l With text-figures Printed in Great Britain RESPRATON N AN AR-BREATHNG FSH, THE CLMBNG PERCH, ANABAS TESTUDNEUS. RESPRATORY PATTERNS AND THE CONTROL OF BREATHNG BY G. M. HUGHES
J. Exp. Biol. (9), S3, 2-29 2l With text-figures Printed in Great Britain RESPRATON N AN AR-BREATHNG FSH, THE CLMBNG PERCH, ANABAS TESTUDNEUS. RESPRATORY PATTERNS AND THE CONTROL OF BREATHNG BY G. M. HUGHES
(a) (i) Describe how a large difference in oxygen concentration is maintained between a fish gill and the surrounding water.
 1. Answers should be written in continuous prose. Credit will be given for biological accuracy, the organisation and presentation of information and the way in which an answer is expressed. Fick s law
1. Answers should be written in continuous prose. Credit will be given for biological accuracy, the organisation and presentation of information and the way in which an answer is expressed. Fick s law
ON THE RECEPTOR FUNCTION OF THE SWIM BLADDER OF FISHES
 i6 ON THE RECEPTOR FUNCTION OF THE SWIM BLADDER OF FISHES BY CH. S. KOSHTOJANZ AND PH. D. VASSILENKO Section of Comparative Physiology, Timiriasev Biological Institute, Moscow {Received i April 1936) (With
i6 ON THE RECEPTOR FUNCTION OF THE SWIM BLADDER OF FISHES BY CH. S. KOSHTOJANZ AND PH. D. VASSILENKO Section of Comparative Physiology, Timiriasev Biological Institute, Moscow {Received i April 1936) (With
Recitation question # 05
 Recitation and Lab # 05 The goal of this recitations / labs is to review material related to the CV and respiratory lectures for the second test of this course. Info required to answer this recitation
Recitation and Lab # 05 The goal of this recitations / labs is to review material related to the CV and respiratory lectures for the second test of this course. Info required to answer this recitation
Biology 347 General Physiology Lab Basic Pulmonary Functions: Respirometry and Factors that Effect Respiration
 Biology 347 General Physiology Lab Basic Pulmonary Functions: Respirometry and Factors that Effect Respiration Objectives Students will measure breathing parameters in a resting subject. Students will
Biology 347 General Physiology Lab Basic Pulmonary Functions: Respirometry and Factors that Effect Respiration Objectives Students will measure breathing parameters in a resting subject. Students will
Chapter 13 The Respiratory System
 Chapter 13 The Respiratory System by Dr. Jay M. Templin Brooks/Cole - Thomson Learning Atmosphere Tissue cell External respiration Alveoli of lungs 1 Ventilation or gas exchange between the atmosphere
Chapter 13 The Respiratory System by Dr. Jay M. Templin Brooks/Cole - Thomson Learning Atmosphere Tissue cell External respiration Alveoli of lungs 1 Ventilation or gas exchange between the atmosphere
SHORT COMMUNICATION CONTINUOUS RECORDING OF ARTERIAL BLOOD P02 IN OCTOPUS VULGARIS DURING PROGRESSIVE HYPOXIA AND MOVEMENT
 J. exp. Biol. 117, 475-479 (1985) 475 Printed in Great Britain The Company of Biologists Limited 1985 SHORT COMMUNICATION CONTINUOUS RECORDING OF ARTERIAL BLOOD P02 IN OCTOPUS VULGARIS DURING PROGRESSIVE
J. exp. Biol. 117, 475-479 (1985) 475 Printed in Great Britain The Company of Biologists Limited 1985 SHORT COMMUNICATION CONTINUOUS RECORDING OF ARTERIAL BLOOD P02 IN OCTOPUS VULGARIS DURING PROGRESSIVE
These two respiratory media (air & water) impose rather different constraints on oxygen uptake:
 Topic 19: OXYGEN UPTAKE AND TRANSPORT (lectures 29-30) OBJECTIVES: 1. Be able to compare air vs. water as a respiratory medium with respect to oxygen content, diffusion coefficient, viscosity and water
Topic 19: OXYGEN UPTAKE AND TRANSPORT (lectures 29-30) OBJECTIVES: 1. Be able to compare air vs. water as a respiratory medium with respect to oxygen content, diffusion coefficient, viscosity and water
OXYGEN CONSUMPTION AND GILL WATER FLOW IN THE DOGFISH SCYLIORHINUS CANICULA L.
 jf. Exp. Biol. (1968), 49, 557-564 557 With 6 text-figures Printed in Great Britain OXYGEN CONSUMPTION AND GILL WATER FLOW IN THE DOGFISH SCYLIORHINUS CANICULA L. BY G. M. HUGHES* AND SHUN-ICHI UMEZAWAf
jf. Exp. Biol. (1968), 49, 557-564 557 With 6 text-figures Printed in Great Britain OXYGEN CONSUMPTION AND GILL WATER FLOW IN THE DOGFISH SCYLIORHINUS CANICULA L. BY G. M. HUGHES* AND SHUN-ICHI UMEZAWAf
GASEOUS EXCHANGE 17 JULY 2013
 GASEOUS EXCHANGE 17 JULY 2013 Lesson Description In this lesson we: Discuss what is gaseous exchange? Consider requirements of an efficient gaseous exchange surface. Look at diversity in gas exchange systems.
GASEOUS EXCHANGE 17 JULY 2013 Lesson Description In this lesson we: Discuss what is gaseous exchange? Consider requirements of an efficient gaseous exchange surface. Look at diversity in gas exchange systems.
Physiology Unit 4 RESPIRATORY PHYSIOLOGY
 Physiology Unit 4 RESPIRATORY PHYSIOLOGY In Physiology Today Respiration External respiration ventilation gas exchange Internal respiration cellular respiration gas exchange Respiratory Cycle Inspiration
Physiology Unit 4 RESPIRATORY PHYSIOLOGY In Physiology Today Respiration External respiration ventilation gas exchange Internal respiration cellular respiration gas exchange Respiratory Cycle Inspiration
Topic 13: Gas Exchange Ch. 42. Gas Exchange pp Gas Exchange. Gas Exchange in Fish pp Gas Exchange in Fish
 Topic 13: Gas Exchange Ch. 42 Fig. 42.24 Gas Exchange pp.979-989 Gas exchange involves the uptake of oxygen and the discharge of carbon dioxide (i.e. respiration or breathing). It is necessary for cellular
Topic 13: Gas Exchange Ch. 42 Fig. 42.24 Gas Exchange pp.979-989 Gas exchange involves the uptake of oxygen and the discharge of carbon dioxide (i.e. respiration or breathing). It is necessary for cellular
RESPIRATORY PHYSIOLOGY. Anaesthesiology Block 18 (GNK 586) Prof Pierre Fourie
 RESPIRATORY PHYSIOLOGY Anaesthesiology Block 18 (GNK 586) Prof Pierre Fourie Outline Ventilation Diffusion Perfusion Ventilation-Perfusion relationship Work of breathing Control of Ventilation 2 This image
RESPIRATORY PHYSIOLOGY Anaesthesiology Block 18 (GNK 586) Prof Pierre Fourie Outline Ventilation Diffusion Perfusion Ventilation-Perfusion relationship Work of breathing Control of Ventilation 2 This image
Lung Volumes and Capacities
 Lung Volumes and Capacities Normally the volume of air entering the lungs during a single inspiration is approximately equal to the volume leaving on the subsequent expiration and is called the tidal volume.
Lung Volumes and Capacities Normally the volume of air entering the lungs during a single inspiration is approximately equal to the volume leaving on the subsequent expiration and is called the tidal volume.
2. State the volume of air remaining in the lungs after a normal breathing.
 CLASS XI BIOLOGY Breathing And Exchange of Gases 1. Define vital capacity. What is its significance? Answer: Vital Capacity (VC): The maximum volume of air a person can breathe in after a forced expiration.
CLASS XI BIOLOGY Breathing And Exchange of Gases 1. Define vital capacity. What is its significance? Answer: Vital Capacity (VC): The maximum volume of air a person can breathe in after a forced expiration.
 Question 1: Define vital capacity. What is its significance? Vital capacity is the maximum volume of air that can be exhaled after a maximum inspiration. It is about 3.5 4.5 litres in the human body. It
Question 1: Define vital capacity. What is its significance? Vital capacity is the maximum volume of air that can be exhaled after a maximum inspiration. It is about 3.5 4.5 litres in the human body. It
Lab 3. The Respiratory System (designed by Heather E. M. Liwanag with T.M. Williams)
 Name Lab Partners Lab 3. The Respiratory System (designed by Heather E. M. Liwanag with T.M. Williams) Part 1. Lung Volumes and Capacities Objectives 1. Obtain graphical representation of lung capacities
Name Lab Partners Lab 3. The Respiratory System (designed by Heather E. M. Liwanag with T.M. Williams) Part 1. Lung Volumes and Capacities Objectives 1. Obtain graphical representation of lung capacities
Respiratory Systems: Ventilation & Gas Exchange
 Respiratory Systems: Ventilation & Gas Exchange Ventilation of Respiratory Surfaces Non-directional ventilation: Medium flows past gas exchange surface in an unpredictable pattern. Tidal Ventilation External
Respiratory Systems: Ventilation & Gas Exchange Ventilation of Respiratory Surfaces Non-directional ventilation: Medium flows past gas exchange surface in an unpredictable pattern. Tidal Ventilation External
Homeostasis and Negative Feedback Concepts and Breathing Experiments 1
 Homeostasis and Negative Feedback Concepts and Breathing Experiments 1 I. Homeostasis and Negative Feedback Homeostasis refers to the maintenance of relatively constant internal conditions. For example,
Homeostasis and Negative Feedback Concepts and Breathing Experiments 1 I. Homeostasis and Negative Feedback Homeostasis refers to the maintenance of relatively constant internal conditions. For example,
Chapter 17 The Respiratory System: Gas Exchange and Regulation of Breathing
 Chapter 17 The Respiratory System: Gas Exchange and Regulation of Breathing Overview of Pulmonary Circulation o Diffusion of Gases o Exchange of Oxygen and Carbon Dioxide o Transport of Gases in the Blood
Chapter 17 The Respiratory System: Gas Exchange and Regulation of Breathing Overview of Pulmonary Circulation o Diffusion of Gases o Exchange of Oxygen and Carbon Dioxide o Transport of Gases in the Blood
Gas Exchange & Circulation
 Why is gas exchange important? Gas Exchange & Circulation Read Ch. 42 start with 42.5: Gas Exchange in Animals Respiration: C 6 H 12 O 6 + O 2! Energy + CO 2 + H 2 O Photosynthesis: Energy + CO 2 + H 2
Why is gas exchange important? Gas Exchange & Circulation Read Ch. 42 start with 42.5: Gas Exchange in Animals Respiration: C 6 H 12 O 6 + O 2! Energy + CO 2 + H 2 O Photosynthesis: Energy + CO 2 + H 2
LAB 7 HUMAN RESPIRATORY LAB. Complete the charts on pgs. 67 and 68 and read directions for using BIOPAC
 66 LAB 7 HUMAN RESPIRATORY LAB Assignments: Due before lab: Quiz: Three Respiratory Interactive Physiology Animations pages 69 73. Complete the charts on pgs. 67 and 68 and read directions for using BIOPAC
66 LAB 7 HUMAN RESPIRATORY LAB Assignments: Due before lab: Quiz: Three Respiratory Interactive Physiology Animations pages 69 73. Complete the charts on pgs. 67 and 68 and read directions for using BIOPAC
IV. FROM AQUATIC TO ATMOSPHERIC BREATHING: THE TRACHEA & THE LUNG
 GAS EXCHANGE AND TRANSPORT I. INTRODUCTION: Heterotrophs oxidize carbon cmpds using O 2 to generate CO 2 & H 2 O. This is cellular respiration II. HOW GAS ENTERS A CELL A. The composition of air: 79% N
GAS EXCHANGE AND TRANSPORT I. INTRODUCTION: Heterotrophs oxidize carbon cmpds using O 2 to generate CO 2 & H 2 O. This is cellular respiration II. HOW GAS ENTERS A CELL A. The composition of air: 79% N
plethysmographic methods that when the subject was pinched on the upper
 24 J. Physiol. (I95I) II2, 24-2I 6I2.I5.6II.976 THE DECREASE IN HAND BLOOD FLOW FOLLOWING INFLATION OF AN ARTERIAL OCCLUSION CUFF ON THE OPPOSITE ARM BY IAN C. RODDIE From the Department of Physiology,
24 J. Physiol. (I95I) II2, 24-2I 6I2.I5.6II.976 THE DECREASE IN HAND BLOOD FLOW FOLLOWING INFLATION OF AN ARTERIAL OCCLUSION CUFF ON THE OPPOSITE ARM BY IAN C. RODDIE From the Department of Physiology,
Exam Key. NROSCI/BIOSC 1070 and MSNBIO 2070 Exam # 2 October 28, 2016 Total POINTS: % of grade in class
 NROSCI/BIOSC 1070 and MSNBIO 2070 Exam # 2 October 28, 2016 Total POINTS: 100 20% of grade in class 1) An arterial blood sample for a patient at sea level is obtained, and the following physiological values
NROSCI/BIOSC 1070 and MSNBIO 2070 Exam # 2 October 28, 2016 Total POINTS: 100 20% of grade in class 1) An arterial blood sample for a patient at sea level is obtained, and the following physiological values
Respiratory Lecture Test Questions Set 3
 Respiratory Lecture Test Questions Set 3 1. The pressure of a gas: a. is inversely proportional to its volume b. is unaffected by temperature changes c. is directly proportional to its volume d. does not
Respiratory Lecture Test Questions Set 3 1. The pressure of a gas: a. is inversely proportional to its volume b. is unaffected by temperature changes c. is directly proportional to its volume d. does not
Increase exchange rate by moving medium over respiratory surface
 Ventilation Increase exchange rate by moving medium over respiratory surface Water ~800x heavier than air, potentially expensive ventilation Oxygen per unit mass Air: 1.3 g contains 210 ml O 2 Water: 1000
Ventilation Increase exchange rate by moving medium over respiratory surface Water ~800x heavier than air, potentially expensive ventilation Oxygen per unit mass Air: 1.3 g contains 210 ml O 2 Water: 1000
Respiration. Figure 22: Schematic representation of the respiratory system
 Respiration One of the seven characteristics of something which is living is respiration. Strictly speaking, respiration is the process that takes place at cellular level and is one of three different
Respiration One of the seven characteristics of something which is living is respiration. Strictly speaking, respiration is the process that takes place at cellular level and is one of three different
OXYGEN UPTAKE IN AIR AND WATER IN THE AIR-BREATHING REEDFISH CALAMOICHTHYS CALABARICUS: ROLE OF SKIN, GILLS AND LUNGS
 J. exp. Biol. (1982), 97, 179-186 179 With 2 figures Printed in Great Britain OXYGEN UPTAKE IN AIR AND WATER IN THE AIR-BREATHING REEDFISH CALAMOICHTHYS CALABARICUS: ROLE OF SKIN, GILLS AND LUNGS BY R.
J. exp. Biol. (1982), 97, 179-186 179 With 2 figures Printed in Great Britain OXYGEN UPTAKE IN AIR AND WATER IN THE AIR-BREATHING REEDFISH CALAMOICHTHYS CALABARICUS: ROLE OF SKIN, GILLS AND LUNGS BY R.
Gas Exchange Respiratory Systems
 alveoli gills Gas Exchange Respiratory Systems elephant seals 2008-2009 Why do we need a respiratory system? respiration for respiration Need O 2 in for aerobic cellular respiration make ATP Need CO 2
alveoli gills Gas Exchange Respiratory Systems elephant seals 2008-2009 Why do we need a respiratory system? respiration for respiration Need O 2 in for aerobic cellular respiration make ATP Need CO 2
RESPIRATORY PHYSIOLOGY, PHYSICS AND PATHOLOGY IN RELATION TO ANAESTHESIA AND INTENSIVE CARE
 Course n : Course 3 Title: RESPIRATORY PHYSIOLOGY, PHYSICS AND PATHOLOGY IN RELATION TO ANAESTHESIA AND INTENSIVE CARE Sub-category: Intensive Care for Respiratory Distress Topic: Pulmonary Function and
Course n : Course 3 Title: RESPIRATORY PHYSIOLOGY, PHYSICS AND PATHOLOGY IN RELATION TO ANAESTHESIA AND INTENSIVE CARE Sub-category: Intensive Care for Respiratory Distress Topic: Pulmonary Function and
Gas Exchange ACTIVITY OVERVIEW SUMMARY KEY CONCEPTS AND PROCESS SKILLS KEY VOCABULARY. Teacher s Guide B-75 L A B O R ATO R Y
 Gas Exchange 17 40- to 2 50-minute sessions ACTIVITY OVERVIEW L A B O R ATO R Y SUMMARY This activity explores the role of the respiratory system in the regulation of gases in the blood. Students investigate
Gas Exchange 17 40- to 2 50-minute sessions ACTIVITY OVERVIEW L A B O R ATO R Y SUMMARY This activity explores the role of the respiratory system in the regulation of gases in the blood. Students investigate
A COMPARISON OF THERMAL POLYGONS FOR BRITISH FRESHWATER TELEOSTS
 178 A. ELLIOTT A COMPARISON OF THERMAL POLYGONS FOR BRITISH FRESHWATER TELEOSTS Introduction ALEX ELLIOTT (J. A. Elliott, 51312 Muirhead House, University of Stirling, Stirling FK9 4LG, Scotland.) Almost
178 A. ELLIOTT A COMPARISON OF THERMAL POLYGONS FOR BRITISH FRESHWATER TELEOSTS Introduction ALEX ELLIOTT (J. A. Elliott, 51312 Muirhead House, University of Stirling, Stirling FK9 4LG, Scotland.) Almost
ACUTE TEMPERATURE TOLERANCE OF JUVENILE CHINOOK SALMON FROM THE MOKELUMNE RIVER
 ACUTE TEMPERATURE TOLERANCE OF JUVENILE CHINOOK SALMON FROM THE MOKELUMNE RIVER Charles H. Hanson, Ph.D. Hanson Environmental, Inc. SUMMARY A series of static acute tests were performed to determine the
ACUTE TEMPERATURE TOLERANCE OF JUVENILE CHINOOK SALMON FROM THE MOKELUMNE RIVER Charles H. Hanson, Ph.D. Hanson Environmental, Inc. SUMMARY A series of static acute tests were performed to determine the
CHANGES IN ACTIVITY AND VENTILATION IN RESPONSE TO HYPOXIA IN UNRESTRAINED, UNOPERATED DOGFISH (SCYLIORHINUS CANICULA L.)
 * exp. Biol. 108, 411-418 (1984) 4 \ Jinted in Great Britain The Company of Biologists Limited 1984 CHANGES IN ACTIVITY AND VENTILATION IN RESPONSE TO HYPOXIA IN UNRESTRAINED, UNOPERATED DOGFISH (SCYLIORHINUS
* exp. Biol. 108, 411-418 (1984) 4 \ Jinted in Great Britain The Company of Biologists Limited 1984 CHANGES IN ACTIVITY AND VENTILATION IN RESPONSE TO HYPOXIA IN UNRESTRAINED, UNOPERATED DOGFISH (SCYLIORHINUS
Respiratory System Lab
 Respiratory System Lab Note: Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin. Take time to organize the materials you will need and set
Respiratory System Lab Note: Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin. Take time to organize the materials you will need and set
CARBON DIOXIDE ELIMINATION FROM SEMICLOSED SYSTEMS
 Brit. J. Anaesth. (1956), 28, 196 CARBON DIOXIDE ELIMINATION FROM SEMICLOSED SYSTEMS BY RUSSELL M. DAVIES, I. R. VERNER Queen Victoria Hospital, East Grinstead AND A. BRACKEN Research and Development Centre,
Brit. J. Anaesth. (1956), 28, 196 CARBON DIOXIDE ELIMINATION FROM SEMICLOSED SYSTEMS BY RUSSELL M. DAVIES, I. R. VERNER Queen Victoria Hospital, East Grinstead AND A. BRACKEN Research and Development Centre,
RESPIRATION OF AN AIR-BREATHING CATFISH CLARIAS BATRACHUS (LINN.)
 y. Exp. Bid. (i97i), 55» 421-434 421 With 6 text-figures Printed in Great Britain RESPIRATION OF AN AIR-BREATHING CATFISH CLARIAS BATRACHUS (LINN.) BY B. N. SINGH AND G. M. HUGHES Research Unit for Comparative
y. Exp. Bid. (i97i), 55» 421-434 421 With 6 text-figures Printed in Great Britain RESPIRATION OF AN AIR-BREATHING CATFISH CLARIAS BATRACHUS (LINN.) BY B. N. SINGH AND G. M. HUGHES Research Unit for Comparative
Conditioned Alarm Behavior in Fathead Minnows (Pimephales promelas) and Test Their Ability
 Conditioned alarm behavior in fathead minnows 1 Meera Alshamsi Prof, Wisenden June 27 th,11 Conditioned Alarm Behavior in Fathead Minnows (Pimephales promelas) and Test Their Ability of Differentiate Between
Conditioned alarm behavior in fathead minnows 1 Meera Alshamsi Prof, Wisenden June 27 th,11 Conditioned Alarm Behavior in Fathead Minnows (Pimephales promelas) and Test Their Ability of Differentiate Between
BREATHING AND EXCHANGE OF GASES
 96 BIOLOGY, EXEMPLAR PROBLEMS CHAPTER 17 BREATHING AND EXCHANGE OF GASES MULTIPLE CHOICE QUESTIONS 1. Respiration in insects is called direct because a. The cell exchange O 2 directly with the air in the
96 BIOLOGY, EXEMPLAR PROBLEMS CHAPTER 17 BREATHING AND EXCHANGE OF GASES MULTIPLE CHOICE QUESTIONS 1. Respiration in insects is called direct because a. The cell exchange O 2 directly with the air in the
Lab #2: Blood pressure and peripheral circulation
 Lab #2: Blood pressure and peripheral circulation Vertebrates have a closed circulatory system where the blood is always enclosed within blood vessels or the heart. Blood is pumped from the heart (the
Lab #2: Blood pressure and peripheral circulation Vertebrates have a closed circulatory system where the blood is always enclosed within blood vessels or the heart. Blood is pumped from the heart (the
Then the partial pressure of oxygen is x 760 = 160 mm Hg
 1 AP Biology March 2008 Respiration Chapter 42 Gas exchange occurs across specialized respiratory surfaces. 1) Gas exchange: the uptake of molecular oxygen (O2) from the environment and the discharge of
1 AP Biology March 2008 Respiration Chapter 42 Gas exchange occurs across specialized respiratory surfaces. 1) Gas exchange: the uptake of molecular oxygen (O2) from the environment and the discharge of
Chapter 4: Ventilation Test Bank MULTIPLE CHOICE
 Instant download and all chapters Test Bank Respiratory Care Anatomy and Physiology Foundations for Clinical Practice 3rd Edition Will Beachey https://testbanklab.com/download/test-bank-respiratory-care-anatomy-physiologyfoundations-clinical-practice-3rd-edition-will-beachey/
Instant download and all chapters Test Bank Respiratory Care Anatomy and Physiology Foundations for Clinical Practice 3rd Edition Will Beachey https://testbanklab.com/download/test-bank-respiratory-care-anatomy-physiologyfoundations-clinical-practice-3rd-edition-will-beachey/
Levels of CO2 in Arterial Blood of Carp under Carbon Dioxide Anesthesia
 J. Nutr. Sci. Vitaminol., 28, 35-39, 1982 Levels of CO2 in Arterial Blood of Carp under Carbon Dioxide Anesthesia Hisateru MITSUDA, Saburo UENO, Hiroshi MIZUNO, Tadashi UEDA, Hiromi FUJIKAWA, Tomoko NOHARA,
J. Nutr. Sci. Vitaminol., 28, 35-39, 1982 Levels of CO2 in Arterial Blood of Carp under Carbon Dioxide Anesthesia Hisateru MITSUDA, Saburo UENO, Hiroshi MIZUNO, Tadashi UEDA, Hiromi FUJIKAWA, Tomoko NOHARA,
alveoli Chapter 42. Gas Exchange elephant seals gills AP Biology
 alveoli Chapter 42. Gas Exchange gills elephant seals Gas exchange O 2 & CO 2 exchange exchange between environment & cells provides O 2 for aerobic cellular respiration need moist membrane need high
alveoli Chapter 42. Gas Exchange gills elephant seals Gas exchange O 2 & CO 2 exchange exchange between environment & cells provides O 2 for aerobic cellular respiration need moist membrane need high
SHORT COMMUNICATION FILAMENT POSITION IN FISH GILLS IS INFLUENCED BY A SMOOTH MUSCLE INNERVATED BY ADRENERGIC NERVES
 J. exp. Biol. 118, 433-437 (1985) 433 Printed in Great Britain The Company of Biologists Limited 1985 SHORT COMMUNICATION FILAMENT POSITION IN FISH GILLS IS INFLUENCED BY A SMOOTH MUSCLE INNERVATED BY
J. exp. Biol. 118, 433-437 (1985) 433 Printed in Great Britain The Company of Biologists Limited 1985 SHORT COMMUNICATION FILAMENT POSITION IN FISH GILLS IS INFLUENCED BY A SMOOTH MUSCLE INNERVATED BY
Coaching Applications The Effect of Intermittent Hypoxic Exposure plus Sea Level Swimming Training on Anaerobic Swimming Performance
 J. Swimming Research, Vol. 19:2 (2012) Coaching Applications plus Sea Level Swimming Training on Anaerobic Swimming Performance Miller A.N. 1 and George K. 2 Department of Biomolecular and Sports Science,
J. Swimming Research, Vol. 19:2 (2012) Coaching Applications plus Sea Level Swimming Training on Anaerobic Swimming Performance Miller A.N. 1 and George K. 2 Department of Biomolecular and Sports Science,
SOLUTIONS Homework #1
 SOLUTIONS Homework #1 INTRODUCTION TO ENGINEERING IN MEDICINE AND BIOLOGY ECEN 1001- Fall 2003 R. Mihran Due Tuesday, 9/9/03 Question 1: a) Chapter 1 of the text discusses the fact that a significant fraction
SOLUTIONS Homework #1 INTRODUCTION TO ENGINEERING IN MEDICINE AND BIOLOGY ECEN 1001- Fall 2003 R. Mihran Due Tuesday, 9/9/03 Question 1: a) Chapter 1 of the text discusses the fact that a significant fraction
AP Biology. Gas Exchange Respiratory Systems. Gas exchange. Why do we need a respiratory system? Optimizing gas exchange. Gas exchange in many forms
 alveoli Gas Exchange Respiratory Systems gills elephant seals 2008-2009 Why do we need a respiratory system? Need O 2 in food respiration for respiration for aerobic cellular respiration make ATP Need
alveoli Gas Exchange Respiratory Systems gills elephant seals 2008-2009 Why do we need a respiratory system? Need O 2 in food respiration for respiration for aerobic cellular respiration make ATP Need
Respiratory Response to Physiologic Challenges. Evaluation copy
 Respiratory Response to Physiologic Challenges Computer 20 The respiratory cycle of inspiration and expiration is controlled by complex mechanisms involving neurons in the cerebral cortex, brain stem,
Respiratory Response to Physiologic Challenges Computer 20 The respiratory cycle of inspiration and expiration is controlled by complex mechanisms involving neurons in the cerebral cortex, brain stem,
PICU Resident Self-Study Tutorial The Basic Physics of Oxygen Transport. I was told that there would be no math!
 Physiology of Oxygen Transport PICU Resident Self-Study Tutorial I was told that there would be no math! INTRODUCTION Christopher Carroll, MD Although cells rely on oxygen for aerobic metabolism and viability,
Physiology of Oxygen Transport PICU Resident Self-Study Tutorial I was told that there would be no math! INTRODUCTION Christopher Carroll, MD Although cells rely on oxygen for aerobic metabolism and viability,
Biology 347 General Physiology Lab Human Diving Response
 Biology 347 General Physiology Lab Human Diving Response Objectives Students will measure the heart and breathing rates of a subject at rest. Students will measure the heart and breathing rates of a subject
Biology 347 General Physiology Lab Human Diving Response Objectives Students will measure the heart and breathing rates of a subject at rest. Students will measure the heart and breathing rates of a subject
Systems of distribution
 Systems of distribution Outline Distribution of respiratory gases, and in blood Respiratory systems - transport of oxygen to tissues - radically different designs in mammals, birds, insects Vertebrate
Systems of distribution Outline Distribution of respiratory gases, and in blood Respiratory systems - transport of oxygen to tissues - radically different designs in mammals, birds, insects Vertebrate
BIOMODAL GAS EXCHANGE DURING VARIATION IN ENVIRONMENTAL OXYGEN AND CARBON DIOXIDE IN THE AIR BREATHING FISH TRICHOGASTER TRICHOPTERUS
 J. exp. Biol. (1979), 8a, 197-213 197 7 figures in Great Britain BIOMODAL GAS EXCHANGE DURING VARIATION IN ENVIRONMENTAL OXYGEN AND CARBON DIOXIDE IN THE AIR BREATHING FISH TRICHOGASTER TRICHOPTERUS BY
J. exp. Biol. (1979), 8a, 197-213 197 7 figures in Great Britain BIOMODAL GAS EXCHANGE DURING VARIATION IN ENVIRONMENTAL OXYGEN AND CARBON DIOXIDE IN THE AIR BREATHING FISH TRICHOGASTER TRICHOPTERUS BY
THE EFFECT OF PROGRESSIVE HYPOXIA ON RESPIRATION IN THE DOGFISH (SCYLIORHINUS CANICULA) AT DIFFERENT SEASONAL TEMPERATURES
 J. exp. Biol. (1975), 63, 117-130 With 7 figures Printed in Great Britain THE EFFECT OF PROGRESSIVE HYPOXIA ON RESPIRATION IN THE DOGFISH (SCYLIORHINUS CANICULA) AT DIFFERENT SEASONAL TEMPERATURES BY P.
J. exp. Biol. (1975), 63, 117-130 With 7 figures Printed in Great Britain THE EFFECT OF PROGRESSIVE HYPOXIA ON RESPIRATION IN THE DOGFISH (SCYLIORHINUS CANICULA) AT DIFFERENT SEASONAL TEMPERATURES BY P.
MOTOR ACTIVITY DURING SEARCHING AND WALKING MOVEMENTS OF COCKROACH LEGS
 J. exp. Biol. 133, 111-120 (1987) \\\ Printed in Great Britain The Company of Biologists Limited 1987 MOTOR ACTIVITY DURING SEARCHING AND WALKING MOVEMENTS OF COCKROACH LEGS BY FRED DELCOMYN Department
J. exp. Biol. 133, 111-120 (1987) \\\ Printed in Great Britain The Company of Biologists Limited 1987 MOTOR ACTIVITY DURING SEARCHING AND WALKING MOVEMENTS OF COCKROACH LEGS BY FRED DELCOMYN Department
Respiratory system. Role. Ventilation consists of 4 (5) steps : oxygen delivery and carbon dioxide elimination ph balance sound and voice formation
 Respiratory system Role oxygen delivery and carbon dioxide elimination ph balance sound and voice formation Ventilation consists of 4 (5) steps : 1. pulmonary ventilation gas exchange between lungs and
Respiratory system Role oxygen delivery and carbon dioxide elimination ph balance sound and voice formation Ventilation consists of 4 (5) steps : 1. pulmonary ventilation gas exchange between lungs and
CHAPTER 3: The respiratory system
 CHAPTER 3: The respiratory system Practice questions - text book pages 56-58 1) When the inspiratory muscles contract, which one of the following statements is true? a. the size of the thoracic cavity
CHAPTER 3: The respiratory system Practice questions - text book pages 56-58 1) When the inspiratory muscles contract, which one of the following statements is true? a. the size of the thoracic cavity
bespoke In general health and rehabilitation Breath-by-breath multi-functional respiratory gas analyser In human performance
 Introduction Expired Gas Analysis or indirect calorimetry, can be used to measure ventilation and the fractions of oxygen and carbon dioxide in expired air. From these measurements, the body's oxygen consumption
Introduction Expired Gas Analysis or indirect calorimetry, can be used to measure ventilation and the fractions of oxygen and carbon dioxide in expired air. From these measurements, the body's oxygen consumption
AP Biology. Chapter 42. Gas Exchange. Optimizing gas exchange. Gas exchange. Gas exchange in many forms. Evolution of gas exchange structures
 alveoli Chapter 42. Gas Exchange gills elephant seals Gas exchange & C exchange exchange between environment & cells provides for aerobic cellular respiration need moist membrane need high surface area
alveoli Chapter 42. Gas Exchange gills elephant seals Gas exchange & C exchange exchange between environment & cells provides for aerobic cellular respiration need moist membrane need high surface area
Collin County Community College. Lung Physiology
 Collin County Community College BIOL. 2402 Anatomy & Physiology WEEK 9 Respiratory System 1 Lung Physiology Factors affecting Ventillation 1. Airway resistance Flow = Δ P / R Most resistance is encountered
Collin County Community College BIOL. 2402 Anatomy & Physiology WEEK 9 Respiratory System 1 Lung Physiology Factors affecting Ventillation 1. Airway resistance Flow = Δ P / R Most resistance is encountered
A STUDY OF THE EFFECT OF TEMPERATURE CHANGES ON THE RESPIRATORY PUMPS OF THE RAINBOW TROUT
 J. Exp. Biol. (1970), 5*. 177-192 I77 With 9 text-figures Printed in Great Britain A STUDY OF THE EFFECT OF TEMPERATURE CHANGES ON THE RESPIRATORY PUMPS OF THE RAINBOW TROUT BY G. M. HUGHES AND J. L. ROBERTS*
J. Exp. Biol. (1970), 5*. 177-192 I77 With 9 text-figures Printed in Great Britain A STUDY OF THE EFFECT OF TEMPERATURE CHANGES ON THE RESPIRATORY PUMPS OF THE RAINBOW TROUT BY G. M. HUGHES AND J. L. ROBERTS*
Respiration. Exercise 1A: Breathing in Resting Volunteers Aim: To measure breathing parameters in a resting individual.
 Respiration Background The amount of air that moves in or out of the lungs during any one breathing cycle is called the tidal volume. Above and beyond normal inspiration, it is possible to breathe in additional
Respiration Background The amount of air that moves in or out of the lungs during any one breathing cycle is called the tidal volume. Above and beyond normal inspiration, it is possible to breathe in additional
Measuring Lung Capacity
 Name Class Date Chapter 37 Circulatory and Respiratory Systems Measuring Lung Capacity Introduction The amount of air that you move in and out of your lungs depends on how quickly you are breathing. The
Name Class Date Chapter 37 Circulatory and Respiratory Systems Measuring Lung Capacity Introduction The amount of air that you move in and out of your lungs depends on how quickly you are breathing. The
Cardiovascular Laboratory Biol 106. In this exercise, you will:
 Cardiovascular Laboratory Biol 106 In this exercise, you will: 1) measure an electrical cardiogram of your own heart. 2) Investigate the human dive response 3) Dissect a mammalian heart 4) observe the
Cardiovascular Laboratory Biol 106 In this exercise, you will: 1) measure an electrical cardiogram of your own heart. 2) Investigate the human dive response 3) Dissect a mammalian heart 4) observe the
Exploring the relationship between Heart Rate (HR) and Ventilation Rate (R) in humans.
 Exploring the relationship between Heart Rate (HR) and Ventilation Rate (R) in humans. The Research Question In this investigation I will be considering the following general research question: Does increased
Exploring the relationship between Heart Rate (HR) and Ventilation Rate (R) in humans. The Research Question In this investigation I will be considering the following general research question: Does increased
1) (1.2±0.2 ± S.E.M., N
 The Journal of Experimental Biology 24, 3963 3971 (21) Printed in Great Britain The ompany of Biologists Limited 21 JEB3776 3963 The relative roles of external and internal O 2 versus H + in eliciting
The Journal of Experimental Biology 24, 3963 3971 (21) Printed in Great Britain The ompany of Biologists Limited 21 JEB3776 3963 The relative roles of external and internal O 2 versus H + in eliciting
MEDICAL EQUIPMENT IV MECHANICAL VENTILATORS. Prof. Yasser Mostafa Kadah
 MEDICAL EQUIPMENT IV - 2013 MECHANICAL VENTILATORS Prof. Yasser Mostafa Kadah Mechanical Ventilator A ventilator is a machine, a system of related elements designed to alter, transmit, and direct energy
MEDICAL EQUIPMENT IV - 2013 MECHANICAL VENTILATORS Prof. Yasser Mostafa Kadah Mechanical Ventilator A ventilator is a machine, a system of related elements designed to alter, transmit, and direct energy
Human Respiration Laboratory Experiment By
 Human Respiration Laboratory Experiment By Alison L., Thurow, Brittany Baierlein, Rachel C. Holsinger and Robin L. Cooper Department of Biology, University of Kentucky, Lexington, KY 40506 0225, USA. Purpose:
Human Respiration Laboratory Experiment By Alison L., Thurow, Brittany Baierlein, Rachel C. Holsinger and Robin L. Cooper Department of Biology, University of Kentucky, Lexington, KY 40506 0225, USA. Purpose:
Medical Instruments in the Developing World
 2.2 Ventilators 2.2.1 Clinical Use and Principles of Operation Many patients in an intensive care and the operating room require the mechanical ventilation of their lungs. All thoracic surgery patients,
2.2 Ventilators 2.2.1 Clinical Use and Principles of Operation Many patients in an intensive care and the operating room require the mechanical ventilation of their lungs. All thoracic surgery patients,
CHAPTER 3: The cardio-respiratory system
 : The cardio-respiratory system Exam style questions - text book pages 44-45 1) Describe the structures involved in gaseous exchange in the lungs and explain how gaseous exchange occurs within this tissue.
: The cardio-respiratory system Exam style questions - text book pages 44-45 1) Describe the structures involved in gaseous exchange in the lungs and explain how gaseous exchange occurs within this tissue.
Activity 2: Examining the Effect of Changing Airway Resistance on Respiratory Volumes
 1 BGYC34 PhysioEx Lab 7 Respiratory Systems Mechanics Marking Scheme Part 1 Complete PhysioEx lab #7. Hand-in all of the pages associated with the lab. Note that there are 5 activities to be completed.
1 BGYC34 PhysioEx Lab 7 Respiratory Systems Mechanics Marking Scheme Part 1 Complete PhysioEx lab #7. Hand-in all of the pages associated with the lab. Note that there are 5 activities to be completed.
A Liter a Lung Measuring Lung Capacity
 A Liter a Lung Measuring Lung Capacity OBJECTIVE In this investigation, students will compare the actual and expected vital capacities of their classmates. LEVEL Middle Grades Life Science CONNECTIONS
A Liter a Lung Measuring Lung Capacity OBJECTIVE In this investigation, students will compare the actual and expected vital capacities of their classmates. LEVEL Middle Grades Life Science CONNECTIONS
Essential Skills Course Acute Care Module. Respiratory Day 2 (Arterial Blood Gases) Pre course Workbook
 Essential Skills Course Acute Care Module Respiratory Day 2 (Arterial Blood Gases) Pre course Workbook Acknowledgements This pre course workbook has been complied and updated with reference to the original
Essential Skills Course Acute Care Module Respiratory Day 2 (Arterial Blood Gases) Pre course Workbook Acknowledgements This pre course workbook has been complied and updated with reference to the original
BREATHING AND EXCHANGE OF GASES
 96 BIOLOGY, EXEMPLAR PROBLEMS CHAPTER 17 BREATHING AND EXCHANGE OF GASES MULTIPLE CHOICE QUESTIONS 1. Respiration in insects is called direct because a. The tissues exchange O 2 directly with the air in
96 BIOLOGY, EXEMPLAR PROBLEMS CHAPTER 17 BREATHING AND EXCHANGE OF GASES MULTIPLE CHOICE QUESTIONS 1. Respiration in insects is called direct because a. The tissues exchange O 2 directly with the air in
Respiration. The resspiratory system
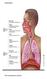 Respiration The resspiratory system The Alveoli The lungs have about 300 million alveoli, with a total crosssec onal area of 50 70 m2.. Each alveolar sac is surrounded by blood capillaries. The walls of
Respiration The resspiratory system The Alveoli The lungs have about 300 million alveoli, with a total crosssec onal area of 50 70 m2.. Each alveolar sac is surrounded by blood capillaries. The walls of
46 healthy human subjects (23 men, 23 women, age range: years)
 Supplementary Methods Tracking General Task 46 healthy human subjects (23 men, 23 women, age range: 18-26 years) participated in this study. All subjects gave informed consent to procedures approved by
Supplementary Methods Tracking General Task 46 healthy human subjects (23 men, 23 women, age range: 18-26 years) participated in this study. All subjects gave informed consent to procedures approved by
