Aseptic processing. the starting material to the finished product, should be controlled by Validation Control of the aseptic process
|
|
|
- Myles Lambert
- 6 years ago
- Views:
Transcription
1 10 Aseptic processing When products are prepared aseptically in unlicensed units there is usually no possibility of quality control of the product before release and use. Consequently quality assurance of the aseptic process is of paramount importance to ensure the quality of the product. This quality assurance includes: validation of the equipment, processes, techniques and staff involved in the preparation control of the process by use of Standard Operating Procedures, monitoring, training, competency assessment, supervision, selfinspection and change control Validation Validation is performed when an aseptic unit is commissioned when any new equipment, process, technique or member of staff is introduced into the process and at defined intervals. The purpose is to show that under simulated conditions aseptic products can be consistently prepared to the required quality using the defined process. Any subsequent changes should be assessed in the same manner to ensure that they do not compromise that quality. Validation methods are described in the appendices but it must be remembered that they only represent the capabilities of the aseptic processing system as tested. To ensure the reproducibility of quality of the product strict adherence to the validated Standard Operating Procedures is essential Control of the aseptic process All key elements and manipulative steps in the aseptic process, from the starting material to the finished product, should be controlled by
2 42 Aseptic processing comprehensive Standard Operating Procedures to ensure that the process consistently produces a product of the requisite quality Key elements of the aseptic process include: entry of personnel and materials into the processing area completing all required documentation for each product including worksheets, labels, etc. maintaining the integrity of the aseptic processing area, and monitoring the workstation and its environment handling and preparation of starting materials, especially disinfection before transfer into the critical zone correct loading and positioning of materials within the critical zone of the controlled workspace good aseptic processing techniques during manipulation of the product, including no-touch of critical surfaces segregation and flow of materials to ensure no inadvertent crosscontamination or substitution of products performance and recording of in-process checks where appropriate removal of product and waste materials from the processing area followed by effective cleaning All aseptic processing must be carried out and supervised by competent staff Staff should be fully conversant with all the relevant Standard Operating Procedures before being deemed competent to work in the aseptic preparation unit Regular updating of staff on the procedures should be undertaken, documented and the extent of knowledge assessed No deviation should be allowed from the Standard Operating Procedures. Should exceptional circumstances necessitate consideration of deviation, this should only be sanctioned by an appropriately experienced and authorised senior member of staff and fully documented. If the changed procedure then becomes normal practice the procedures should be rewritten and revalidated in the usual way to take account of the changed circumstances There should be a formal written procedure for the assessment of any proposed change which may affect product quality and if the change is
3 Some principles and examples of good aseptic technique 43 implemented it must be widely communicated to and acknowledged by all members of the aseptic team Standard Operating Procedures should be written and implemented for all equipment used for aseptic processing. Where appropriate, equipment should be regularly calibrated and the accuracy of volume measuring devices validated All staff working in aseptic processing should be made fully aware of the potential consequences of any deviation from the validated procedure, both to the integrity of the product and to the intended recipient. Regular reminders of the critical nature of the process should be provided Staff involved in aseptic processing should be taught to recognise upper limb disorders or repetitive strain injuries and to use techniques to minimise these conditions wherever possible Some principles and examples of good aseptic technique Before starting a session of work put on a fresh pair of sterile gloves. If the gloves have been used to prepare a previous product during that session of work, check them for damage and if none is found disinfect them using sterile 70% alcohol before commencing preparation of the next product Wherever possible use double-wrapped, sterilised disposable equipment to avoid the need for disinfection of the outer surface before transfer into the critical zone Syringes or needles packed in strips should be separated before transfer into the critical zone to reduce the potential for particle dispersion Where the outer surface of materials has to be disinfected before transfer into the critical zone use the spray and wipe technique with sterile 70% alcohol and a sterile wipe (see 12.3) When transferred into the critical zone allow all materials to dry before proceeding with the preparation Position the product in the critical zone so that there is unobstructed airflow over and around it.
4 44 Aseptic processing Keep the critical zone uncluttered and free from previous waste Avoid reaching over the product to access equipment or dispose of waste Swab the surfaces of bungs that will be penetrated and the necks of ampoules with a fresh sterile 70% alcohol impregnated wipe and allow to dry before proceeding Use a strict no-touch technique to avoid any contact with any surface which will be in contact with the sterile fluid path. 63 Note: Additional procedures to protect the operator may be determined by the hazardous nature of the product, e.g. radiation protection considerations for radiopharmaceuticals Peel open over-wrapped items pointing towards the air stream from the HEPA filter. Do not tear paper wrappers Use closed systems wherever possible Where ampoules have to be used make only one withdrawal immediately after opening and discard any remainder (see Chapter 2, definition 9) When opening glass ampoules cover the neck with a freshly opened sterile 70% alcohol swab before snapping open When withdrawing from glass ampoules use a sterile filter straw or filter needle to remove glass particles. Replace the filter straw or needle with a fresh sterile needle before adding the solution to another container When using vials avoid aerosols by using pressure equalisation within the syringe or venting devices When making additions to infusion bags wherever possible suspend them so that the additive port is in the HEPA-filtered air stream rather than on the work surface.
5 Some principles and examples of good aseptic technique Take care to insert needle through the centre of the additive port, keeping the needle straight to avoid puncturing the bag. Consider the effect of multiple insertions through the additive port and take steps to minimise if necessary Use an appropriate gauge of needle that will minimise damage to rubber bungs whilst still maintaining an acceptable flow rate Ensure that all tubing is clear of fluid and securely clamped before removal from the critical zone Clean up all drips or spillage of product immediately and disinfect the work surface and gloves between products.
Aseptic Techniques. Techniques for Sterile Compounding. Pharmacy Technician Training Systems Passassured, LLC
 Aseptic Techniques Techniques for Sterile Compounding Pharmacy Technician Training Systems Passassured, LLC Aseptic Techniques, Tech for Sterile Compounding PassAssured's Pharmacy Technician Training Program
Aseptic Techniques Techniques for Sterile Compounding Pharmacy Technician Training Systems Passassured, LLC Aseptic Techniques, Tech for Sterile Compounding PassAssured's Pharmacy Technician Training Program
1.0 PURPOSE The purpose of this procedure is to describe the use of basic aseptic technique.
 1.0 PURPOSE The purpose of this procedure is to describe the use of basic aseptic technique. 2.0 RESPONSIBILITY 2.1 It is the responsibility of each employee working in a laboratory setting to follow this
1.0 PURPOSE The purpose of this procedure is to describe the use of basic aseptic technique. 2.0 RESPONSIBILITY 2.1 It is the responsibility of each employee working in a laboratory setting to follow this
Transfer Procedures. By the end of this chapter, you will be able to: The Transfer Process. Chapter 12
 Chapter 12 Transfer Procedures By the end of this chapter, you will be able to: l Explain the term transfer process l Name two types of sanitisation methods often used l Describe the spray and wipe technique
Chapter 12 Transfer Procedures By the end of this chapter, you will be able to: l Explain the term transfer process l Name two types of sanitisation methods often used l Describe the spray and wipe technique
Patients Guide for Self Administering Intravenous Antibiotics General points
 Patients Guide for Self Administering Intravenous Antibiotics General points An intravenous line is an open route for infection therefore you need to prevent this by washing your hands before any procedure
Patients Guide for Self Administering Intravenous Antibiotics General points An intravenous line is an open route for infection therefore you need to prevent this by washing your hands before any procedure
Toolkit for safe Use of Microbiological Safety Cabinets(MSC s)
 Toolkit for safe Use of Microbiological Safety Cabinets(MSC s) What Do I Need To Do? How Do I Manage the Use of MSC s Recommendations 1 What Do I Need To Do Head of Department? Information, induction,
Toolkit for safe Use of Microbiological Safety Cabinets(MSC s) What Do I Need To Do? How Do I Manage the Use of MSC s Recommendations 1 What Do I Need To Do Head of Department? Information, induction,
CTG Certification Protocol -- Version 3.0 SUMMARY
 Date of Test: Test Report Number: SUMMARY Date of Prior Certification: Certified by: Re-certification Due Date: Unit Description: Make : Model: Serial Number: Location: CERTIFIER INFORMATION Name: Client
Date of Test: Test Report Number: SUMMARY Date of Prior Certification: Certified by: Re-certification Due Date: Unit Description: Make : Model: Serial Number: Location: CERTIFIER INFORMATION Name: Client
RECOMBINATE. [Antihemophilic Factor (Recombinant)] (For intravenous use only)
![RECOMBINATE. [Antihemophilic Factor (Recombinant)] (For intravenous use only) RECOMBINATE. [Antihemophilic Factor (Recombinant)] (For intravenous use only)](/thumbs/79/80305643.jpg) Website for more information: http://www.recombinate.com/ INSTRUCTIONS FOR USE RECOMBINATE [Antihemophilic Factor (Recombinant)] (For intravenous use only) Do not attempt to do an infusion to yourself
Website for more information: http://www.recombinate.com/ INSTRUCTIONS FOR USE RECOMBINATE [Antihemophilic Factor (Recombinant)] (For intravenous use only) Do not attempt to do an infusion to yourself
DIAGNOSTIC CARDIOLOGY
 DIAGNOSTIC CARDIOLOGY For Your Procedural Needs Manufactured to the Highest Standards, Customised to Your Specific Needs MEDLINE PROCEDURE PACKS The items contained within this brochure are perfect for
DIAGNOSTIC CARDIOLOGY For Your Procedural Needs Manufactured to the Highest Standards, Customised to Your Specific Needs MEDLINE PROCEDURE PACKS The items contained within this brochure are perfect for
Venepuncture. Clinical Skills. Venepuncture. Dr Brian Jenkins (Clinical Skills Lead) Sian Williams (Clinical Skills Manager)
 Clinical Skills Venepuncture Dr Brian Jenkins (Clinical Skills Lead) Sian Williams (Clinical Skills Manager) Aims and Objectives Aims and Objectives The aim of this module is to facilitate learning regarding
Clinical Skills Venepuncture Dr Brian Jenkins (Clinical Skills Lead) Sian Williams (Clinical Skills Manager) Aims and Objectives Aims and Objectives The aim of this module is to facilitate learning regarding
Air-Mate High Efficiency Powered Air Purifying Respirator (PAPR) Staff Training Module
 Air-Mate High Efficiency Powered Air Purifying Respirator (PAPR) Staff Training Module PRES0389 7/05-1 Powered Air Purifying Respirators (PAPR) Reduce the risk of transmission of infectious agents spread
Air-Mate High Efficiency Powered Air Purifying Respirator (PAPR) Staff Training Module PRES0389 7/05-1 Powered Air Purifying Respirators (PAPR) Reduce the risk of transmission of infectious agents spread
Biological Safety Cabinet Guidelines
 Biological Safety Cabinet Guidelines 2017 Table of Contents 1 Introduction... 2 2 BSC Start-Up Considerations... 3 3 Working in the BSC... 4 4 Completion of Work in the BSC... 5 5 Ultraviolet Lights...
Biological Safety Cabinet Guidelines 2017 Table of Contents 1 Introduction... 2 2 BSC Start-Up Considerations... 3 3 Working in the BSC... 4 4 Completion of Work in the BSC... 5 5 Ultraviolet Lights...
Bachelor of Paramedic Science
 Bachelor of Paramedic Science Practical Workshop Safety Induction Paramedic Teaching Suite and Paramedic Shed Evacuation Evacuation procedures - displayed in the corridors of Paramedic Teaching Suite.
Bachelor of Paramedic Science Practical Workshop Safety Induction Paramedic Teaching Suite and Paramedic Shed Evacuation Evacuation procedures - displayed in the corridors of Paramedic Teaching Suite.
Laboratory Safety Training. Environmental Health and Safety
 Laboratory Safety Training Environmental Health and Safety Why You Need Lab Safety Training Hazardous materials and/or equipment. You need to understand the risks and how to mitigate them. OSU requires
Laboratory Safety Training Environmental Health and Safety Why You Need Lab Safety Training Hazardous materials and/or equipment. You need to understand the risks and how to mitigate them. OSU requires
CBMTG Laboratory Committee-Laboratory Guidance
 Purpose: To confirm, by examination and objective evidence, that a selected cryopreservation bag is compatible with institutional policies and standard operating procedures and functions adequately so
Purpose: To confirm, by examination and objective evidence, that a selected cryopreservation bag is compatible with institutional policies and standard operating procedures and functions adequately so
C O L O G Y. New. g u O N. l o t a C a. The high security connection
 O N g u e C O L O G Y New l o t a C a The high security connection Preparation devices for medications/solutions Bag qimospike B 7218.01 qimospike B is an IV fluid bag access spike fitted with qimo. Aspiration
O N g u e C O L O G Y New l o t a C a The high security connection Preparation devices for medications/solutions Bag qimospike B 7218.01 qimospike B is an IV fluid bag access spike fitted with qimo. Aspiration
Dilution Sequence Protocol
 Dilution Sequence Protocol US Package Insert Multi-Patient (2-5 patients; requires 2 vials of Provocholine) 0.025 0.25 2.5 10 25 Provocholine 100 mg/vial (US NDC 64281-100-06) 1 Provocholine (methacholine
Dilution Sequence Protocol US Package Insert Multi-Patient (2-5 patients; requires 2 vials of Provocholine) 0.025 0.25 2.5 10 25 Provocholine 100 mg/vial (US NDC 64281-100-06) 1 Provocholine (methacholine
Choosing and Installing Microbiological Safety Cabinets
 Choosing and Installing Microbiological Safety Cabinets Guidance for University Departments and Functions First Edition December 2006 Safety Services Office Issued with the approval of the Biological and
Choosing and Installing Microbiological Safety Cabinets Guidance for University Departments and Functions First Edition December 2006 Safety Services Office Issued with the approval of the Biological and
Specimen Collection Procedures
 Specimen Collection Procedures I. Overview The following are the procedures for collecting urine and blood samples from participants in the VADA program. If the precise method of sample collection varies
Specimen Collection Procedures I. Overview The following are the procedures for collecting urine and blood samples from participants in the VADA program. If the precise method of sample collection varies
Summary of BCCA Pharmacy Practice Standards for Hazardous Drugs
 Summary of BCCA Pharmacy Practice Standards for Hazardous Drugs Table of Contents Pharmacy Oncology Certification... 1 Module 1 Safe Handling of Hazardous Drugs... 1 Section A... 1 A.1 Potential Hazards
Summary of BCCA Pharmacy Practice Standards for Hazardous Drugs Table of Contents Pharmacy Oncology Certification... 1 Module 1 Safe Handling of Hazardous Drugs... 1 Section A... 1 A.1 Potential Hazards
Dilution Sequence Protocol
 Dilution Sequence Protocol ATS Short Quadrupling Doses 0.0625 0.25 1 4 16 Provocholine 100 mg/vial (US NDC 64281-100-06) 1 Provocholine (methacholine chloride USP) powder for solution, for inhalation,
Dilution Sequence Protocol ATS Short Quadrupling Doses 0.0625 0.25 1 4 16 Provocholine 100 mg/vial (US NDC 64281-100-06) 1 Provocholine (methacholine chloride USP) powder for solution, for inhalation,
Dilution Sequence Protocol
 Dilution Sequence Protocol ATS Short Quadrupling Doses 0.0625 0.25 1 4 16 Provocholine 1600 mg/vial (US NDC 64281-016-01) 1 Provocholine (methacholine chloride USP) powder for solution, for inhalation,
Dilution Sequence Protocol ATS Short Quadrupling Doses 0.0625 0.25 1 4 16 Provocholine 1600 mg/vial (US NDC 64281-016-01) 1 Provocholine (methacholine chloride USP) powder for solution, for inhalation,
RECONSTITUTION AND ADMINISTRATION INSTRUCTIONS FOR HEALTHCARE PROFESSIONALS
 RECONSTITUTION AND ADMINISTRATION INSTRUCTIONS FOR HEALTHCARE PROFESSIONALS TRUST THE POWER OF PREVENTION WITH CINRYZE Whether your patients are new to treatment or are continuing their journey, remind
RECONSTITUTION AND ADMINISTRATION INSTRUCTIONS FOR HEALTHCARE PROFESSIONALS TRUST THE POWER OF PREVENTION WITH CINRYZE Whether your patients are new to treatment or are continuing their journey, remind
RECONSTITUTION AND ADMINISTRATION INSTRUCTIONS FOR HEALTHCARE PROFESSIONALS. Hereditary Angioedema
 RECONSTITUTION AND ADMINISTRATION INSTRUCTIONS FOR HEALTHCARE PROFESSIONALS Hereditary Angioedema TRUST PROVEN PREVENTION WITH CINRYZE Whether your patients are new to treatment or are continuing their
RECONSTITUTION AND ADMINISTRATION INSTRUCTIONS FOR HEALTHCARE PROFESSIONALS Hereditary Angioedema TRUST PROVEN PREVENTION WITH CINRYZE Whether your patients are new to treatment or are continuing their
CCM Approval: IACUC Approval:
 CCM Approval: IACUC Approval: 1.0 Purpose: The purpose of this SOP is to describe the necessary procedures for the safe administration of potentially hazardous agents 1 to rodents via injection, drinking
CCM Approval: IACUC Approval: 1.0 Purpose: The purpose of this SOP is to describe the necessary procedures for the safe administration of potentially hazardous agents 1 to rodents via injection, drinking
Sharps The Safe Use and Handling of - Safe System of Work
 2017 Sharps The Safe Use and Handling of - Safe System of Work Pendragon PLC 2/8/2017 Sharps The Safe Use and Handling of SSOW This Safe System of Work (SSOW) is designed to communicate group best practice
2017 Sharps The Safe Use and Handling of - Safe System of Work Pendragon PLC 2/8/2017 Sharps The Safe Use and Handling of SSOW This Safe System of Work (SSOW) is designed to communicate group best practice
Webinar Expert Module 1: Garment Logistics and Gowning. Dupont September GOP-Innovations
 Webinar Expert Module 1: Garment Logistics and Gowning Dupont 28-29 September 2017 GOP-Innovations your Partner for Practical Training and e-learning Milenko Pavičić Pharmaceutical microbiologist, trainer,
Webinar Expert Module 1: Garment Logistics and Gowning Dupont 28-29 September 2017 GOP-Innovations your Partner for Practical Training and e-learning Milenko Pavičić Pharmaceutical microbiologist, trainer,
1. The laboratory supervisor must enforce the institutional policies that control access to the laboratory.
 APPENDIX E-3: Biosafety Level 3 (BSL-3) The following is taken from the Biosafety in Microbiological and Biomedical Laboratories (BMBL) 5 th Edition, February 2009 Centers for Disease Control and Prevention
APPENDIX E-3: Biosafety Level 3 (BSL-3) The following is taken from the Biosafety in Microbiological and Biomedical Laboratories (BMBL) 5 th Edition, February 2009 Centers for Disease Control and Prevention
NUS Safety & Health Guidelines Procedure No: OSHE/SOP/GL/08
 Page: Page 1 of 5 Prepared By Approved By Review Date Swapan Kanti Biswas Dr Peck Thian Guan 1.0 OBJECTIVE The objective of this document is to provide guidance on the selection and use of (PPE). This
Page: Page 1 of 5 Prepared By Approved By Review Date Swapan Kanti Biswas Dr Peck Thian Guan 1.0 OBJECTIVE The objective of this document is to provide guidance on the selection and use of (PPE). This
The primary purpose of laboratory fume hoods is to keep toxic or irritating vapors out of the general laboratory working area.
 Introduction Welcome to the Biosafety Cabinets and Fume Hoods Training Course (OHS_BIO304). This training is required for anyone that will be conducting work and/or research that requires the use of a
Introduction Welcome to the Biosafety Cabinets and Fume Hoods Training Course (OHS_BIO304). This training is required for anyone that will be conducting work and/or research that requires the use of a
Radiation Protection Service RADIOISOTOPES : SPECIFIC PROCEDURES AND HAZARD ASSESSMENT
 Radiation Protection Service RADIOISOTOPES : AND Revised November 2008 1 TABLE OF CONTENTS Alpha Emitters... 3 H3, C14 & S35... 3 Ba-133... 4 Ca-45... 4 Cl-36... 5 Co-57... 6 Cr-51... 7 Cu-64... 7 I-125
Radiation Protection Service RADIOISOTOPES : AND Revised November 2008 1 TABLE OF CONTENTS Alpha Emitters... 3 H3, C14 & S35... 3 Ba-133... 4 Ca-45... 4 Cl-36... 5 Co-57... 6 Cr-51... 7 Cu-64... 7 I-125
ML3M Monnier Miniature Compressed Air Lubricator
 5041050/3 IM-P504-04 CH Issue 3 ML3M Monnier Miniature Compressed Air Lubricator Installation and Maintenance Instructions 1. Safety information 2. General product information 3. Installation and commissioning
5041050/3 IM-P504-04 CH Issue 3 ML3M Monnier Miniature Compressed Air Lubricator Installation and Maintenance Instructions 1. Safety information 2. General product information 3. Installation and commissioning
USP <797> LABELING ESTABLISHING BEYOND-USE DATES AND IN-USE TIMES
 USP LABELING ESTABLISHING BEYOND-USE DATES AND IN-USE TIMES Each CSP label must state the date beyond which the preparation cannot be used and must be discarded. A CSP may also be labeled with an
USP LABELING ESTABLISHING BEYOND-USE DATES AND IN-USE TIMES Each CSP label must state the date beyond which the preparation cannot be used and must be discarded. A CSP may also be labeled with an
In summary, the ISPE comments suggest the following additional improvements to the modified Annex 1 document:
 28 April 2006 To: Ms. Sabine Atzor, European Commission, Brussels Mr. David Cockburn, EMEA, London Via e-mail to: sabine.atzor@cec.eu.int david.cockburn@emea.eu.int Doc. Ref: EMEA/INS/GMP/318222/2005,
28 April 2006 To: Ms. Sabine Atzor, European Commission, Brussels Mr. David Cockburn, EMEA, London Via e-mail to: sabine.atzor@cec.eu.int david.cockburn@emea.eu.int Doc. Ref: EMEA/INS/GMP/318222/2005,
Peripheral Venous Catheter Placement Simulator
 Attention Do not let ink from pens, newspapers, this manual or other sources come in contact with the manikin, as they cannot be cleaned the manikin skin. MW9 Peripheral Venous Catheter Placement Simulator
Attention Do not let ink from pens, newspapers, this manual or other sources come in contact with the manikin, as they cannot be cleaned the manikin skin. MW9 Peripheral Venous Catheter Placement Simulator
BIOSAFETY SELF AUDIT PRINCIPAL INVESTIGATOR/PERMIT HOLDER: CONTAINMENT LEVEL: 1 2 (Shaded) RISK GROUP: 1 2 AUDIT TO BE COMPLETED BY (DATE):
 Page 1 of 5 BIOSAFETY PROGRAM BIOSAFETY SELF AUDIT PRINCIPAL INVESTIGATOR/PERMIT HOLDER: SELF AUDIT COMPLETED BY: LABORATORY ROOM NUMBER: CONTAINMENT LEVEL: 1 2 (Shaded) RISK GROUP: 1 2 DATE OF AUDIT:
Page 1 of 5 BIOSAFETY PROGRAM BIOSAFETY SELF AUDIT PRINCIPAL INVESTIGATOR/PERMIT HOLDER: SELF AUDIT COMPLETED BY: LABORATORY ROOM NUMBER: CONTAINMENT LEVEL: 1 2 (Shaded) RISK GROUP: 1 2 DATE OF AUDIT:
PERFORMANCE OF A FINGERSTICK
 PERFORMANCE OF A FINGERSTICK Follow the procedure as outlined above for greeting and identifying the patient. As always, properly fill out appropriate requisition forms, indicating the test(s) ordered.
PERFORMANCE OF A FINGERSTICK Follow the procedure as outlined above for greeting and identifying the patient. As always, properly fill out appropriate requisition forms, indicating the test(s) ordered.
Discussion and guidance on the definition and qualification of porous loads
 SUMMARY Porous Loads requiring Sterilisation are often a Critical Control Point for the manufacture of Sterile products. Particularly Aseptically filled products. Regulatory guidance in this area is not
SUMMARY Porous Loads requiring Sterilisation are often a Critical Control Point for the manufacture of Sterile products. Particularly Aseptically filled products. Regulatory guidance in this area is not
OPERATOR, PRODUCT AND ENVIRONMENTAL PROTECTION
 ISOLATORS OPERATOR, PRODUCT AND ENVIRONMENTAL PROTECTION Aseptic Isolators Extract Technology is a leading supplier of containment and aseptic systems for the pharmaceutical and biotech markets. We have
ISOLATORS OPERATOR, PRODUCT AND ENVIRONMENTAL PROTECTION Aseptic Isolators Extract Technology is a leading supplier of containment and aseptic systems for the pharmaceutical and biotech markets. We have
OPERATOR, PRODUCT AND ENVIRONMENTAL PROTECTION
 ISOLATORS OPERATOR, PRODUCT AND ENVIRONMENTAL PROTECTION Aseptic Isolators Extract Technology is a leading supplier of containment and aseptic systems for the pharmaceutical and biotech markets. We have
ISOLATORS OPERATOR, PRODUCT AND ENVIRONMENTAL PROTECTION Aseptic Isolators Extract Technology is a leading supplier of containment and aseptic systems for the pharmaceutical and biotech markets. We have
Aseptic Vial and Syringe Filling Narlin B. Beaty, Ph.D. Adelaide Nye
 Aseptic Vial and Syringe Filling Narlin B. Beaty, Ph.D. Adelaide Nye Like the preparation of a fine dinner, aseptic filling of vials and syringes is best performed with the highest quality components,
Aseptic Vial and Syringe Filling Narlin B. Beaty, Ph.D. Adelaide Nye Like the preparation of a fine dinner, aseptic filling of vials and syringes is best performed with the highest quality components,
Sole reliance for sterility or other quality aspects must not be placed on finished product test.
 This chapter provides requirements for aseptic production in the manufacture and quality control of veterinary vaccines in accordance with Chapter 1.1.8 Principles of veterinary vaccine production and
This chapter provides requirements for aseptic production in the manufacture and quality control of veterinary vaccines in accordance with Chapter 1.1.8 Principles of veterinary vaccine production and
6900 Maintenance Instruction System Flush
 Equipment Required FA74005 Damper Drain Tube FA16005 Cover Removal Tool FA900005 Beaker 0.25 Litre FA900003 Solvent Cleaning Bottle FA940021 Syringe Polypropylene 50 ml as required FA999045 Gloves Latex
Equipment Required FA74005 Damper Drain Tube FA16005 Cover Removal Tool FA900005 Beaker 0.25 Litre FA900003 Solvent Cleaning Bottle FA940021 Syringe Polypropylene 50 ml as required FA999045 Gloves Latex
Gerard Sheridan Inspector
 Manufacture of Sterile Medicinal Products Areas of Focus for GMP Inspections GMP Information Day; 23rd October 2008. Gerard Sheridan Inspector Date : 23/10/2008 Slide 1 Objectives Highlight areas frequently
Manufacture of Sterile Medicinal Products Areas of Focus for GMP Inspections GMP Information Day; 23rd October 2008. Gerard Sheridan Inspector Date : 23/10/2008 Slide 1 Objectives Highlight areas frequently
THE UNIVERSITY OF NEWCASTLE - DISCIPLINE OF MEDICAL BIOCHEMISTRY
 THE UNIVERSITY OF NEWCASTLE - DISCIPLINE OF MEDICAL BIOCHEMISTRY Page: 1 of 9 1. Risk Assessment: This risk assessment has been prepared using the National Safety Council of Australia Risk Score Calculator
THE UNIVERSITY OF NEWCASTLE - DISCIPLINE OF MEDICAL BIOCHEMISTRY Page: 1 of 9 1. Risk Assessment: This risk assessment has been prepared using the National Safety Council of Australia Risk Score Calculator
Assisting with Insertion. Care of Intraspinal Catheters
 Guidelines included: Assisting with an Insertion Care of Various types of Intraspinal s Care of the Intraspinal Infusion Monitoring Removal of the Short Term Non Assisting with Insertion INR should be
Guidelines included: Assisting with an Insertion Care of Various types of Intraspinal s Care of the Intraspinal Infusion Monitoring Removal of the Short Term Non Assisting with Insertion INR should be
In addition to the responsibility for managing the elements of the respiratory protection program, he/she will be responsible for:
 EBBE (1 of 7) EBBE RESPIRATORY PROTECTION POLICY The Muskogee School District is acknowledged as the employer, and as outlined under current Federal regulations, is assigned the responsibility to provide
EBBE (1 of 7) EBBE RESPIRATORY PROTECTION POLICY The Muskogee School District is acknowledged as the employer, and as outlined under current Federal regulations, is assigned the responsibility to provide
Use of Biosafety II cabinets
 RISK ASSESSMENT FORM Use this form to assist you to complete risk assessments for hazardous activities and processes. Any serious or ongoing hazards should be reported via RiskWare to ensure that appropriate
RISK ASSESSMENT FORM Use this form to assist you to complete risk assessments for hazardous activities and processes. Any serious or ongoing hazards should be reported via RiskWare to ensure that appropriate
TJF-Q180V Cleaning and Disinfection Checklist
 TJF-Q180V Cleaning and Disinfection Checklist TJF-Q180V Cleaning and Disinfection Checklist This checklist is used to evaluate and confirm if cleaning and disinfection of the TJF-Q180V has been performed
TJF-Q180V Cleaning and Disinfection Checklist TJF-Q180V Cleaning and Disinfection Checklist This checklist is used to evaluate and confirm if cleaning and disinfection of the TJF-Q180V has been performed
Exeter Clinical and Health Research
 Exeter Clinical and Health Research Standard Operating Procedure: Blood sample separation and storage SOP Number: NIHRexe/002/GEN Version Number & Date: V1.1 22/02/2017 Review Date: 22/02/2017 Superseded
Exeter Clinical and Health Research Standard Operating Procedure: Blood sample separation and storage SOP Number: NIHRexe/002/GEN Version Number & Date: V1.1 22/02/2017 Review Date: 22/02/2017 Superseded
Environmental Health & Safety Policy Manual
 Environmental Health & Safety Policy Manual Issue Date 8/24/2009 Updated: 5/8/2018 Policy # EHS-400.03 Personal Protective Equipment 1.0 PURPOSE: In order to protect the health of employees and students,
Environmental Health & Safety Policy Manual Issue Date 8/24/2009 Updated: 5/8/2018 Policy # EHS-400.03 Personal Protective Equipment 1.0 PURPOSE: In order to protect the health of employees and students,
Whitepaper Technical Opinion: Grade A - Air Supply and Local Protection
 Whitepaper Technical Opinion: Grade A - Air Supply and Local Protection I N D U S T R Y I N S I G H T S Norman A. Goldschmidt Sr. Principal, President www.geieng.com I N D U S T R Y I N S I G H T S Genesis
Whitepaper Technical Opinion: Grade A - Air Supply and Local Protection I N D U S T R Y I N S I G H T S Norman A. Goldschmidt Sr. Principal, President www.geieng.com I N D U S T R Y I N S I G H T S Genesis
Assembly and Installation Procedures
 Assembly and Installation Procedures for Pall Supracap 100 Capsules 1. Introduction The following procedures must be followed for the installation of Pall Supracap 100 Capsules. The instructions contained
Assembly and Installation Procedures for Pall Supracap 100 Capsules 1. Introduction The following procedures must be followed for the installation of Pall Supracap 100 Capsules. The instructions contained
Exeter Clinical and Health Research
 Exeter Clinical and Health Research Standard Operating Procedure: Centrifuge Operation SOP Number: NIHRexe/019/GEN Version Number & Date: V2 13/06/2017 Review Date: 13/06/2018 Superseded Version Number
Exeter Clinical and Health Research Standard Operating Procedure: Centrifuge Operation SOP Number: NIHRexe/019/GEN Version Number & Date: V2 13/06/2017 Review Date: 13/06/2018 Superseded Version Number
Dri-Line Mk2 Spirax-Monnier Compressed Air Drain Trap
 0509950/2 IM-P050-21 CH Issue 2 Dri-Line Mk2 Spirax-Monnier Compressed Air Drain Trap Installation and Maintenance Instructions 1. Safety information 2. General product information 3. Installation and
0509950/2 IM-P050-21 CH Issue 2 Dri-Line Mk2 Spirax-Monnier Compressed Air Drain Trap Installation and Maintenance Instructions 1. Safety information 2. General product information 3. Installation and
New. The high security connection
 L ON Cata C lo gu e O O G Y New The high security connection Preparation devices for medications/solutions Bag qimospike B 7218.01 qimospike B is an IV fluid bag access spike fitted with qimo. Aspiration
L ON Cata C lo gu e O O G Y New The high security connection Preparation devices for medications/solutions Bag qimospike B 7218.01 qimospike B is an IV fluid bag access spike fitted with qimo. Aspiration
School of Biological Sciences: Zoology Building
 School of Biological Sciences: Zoology Building Ionising Radiation Regulations 1999 LOCAL RULES These rules apply to the following areas:- Zoology rooms 431, 110 Issue date April 2012 Review date April
School of Biological Sciences: Zoology Building Ionising Radiation Regulations 1999 LOCAL RULES These rules apply to the following areas:- Zoology rooms 431, 110 Issue date April 2012 Review date April
PEDIATRIC INJECTABLE TRAINING ARM SIMULATOR LF00958U INSTRUCTION MANUAL
 PEDIATRIC INJECTABLE TRAINING ARM SIMULATOR LF00958U INSTRUCTION MANUAL DO NOT REMOVE FILM FROM TUBING! THIS PRODUCT CONTAINS DRY NATURAL RUBBER! Products by NASCO About the Simulator The Life/form Pediatric
PEDIATRIC INJECTABLE TRAINING ARM SIMULATOR LF00958U INSTRUCTION MANUAL DO NOT REMOVE FILM FROM TUBING! THIS PRODUCT CONTAINS DRY NATURAL RUBBER! Products by NASCO About the Simulator The Life/form Pediatric
Confined Spaces. This procedure applies to all workers of the Diocese of Maitland Newcastle (DoMN).
 Purpose To provide guidance in controlling the risks associated with confined spaces including identifying confined spaces, working in a confined space and issuing confined space entry permits. Scope This
Purpose To provide guidance in controlling the risks associated with confined spaces including identifying confined spaces, working in a confined space and issuing confined space entry permits. Scope This
COMPOUNDING WITH HAZARDOUS DRUGS
 VOLUME 15 NUMBER 2 Secundum Artem Current & Practical Compounding Information for the Pharmacist. An ongoing CE Program provided by a grant from Paddock Laboratories, Inc. COMPOUNDING WITH GOALS AND OBJECTIVES
VOLUME 15 NUMBER 2 Secundum Artem Current & Practical Compounding Information for the Pharmacist. An ongoing CE Program provided by a grant from Paddock Laboratories, Inc. COMPOUNDING WITH GOALS AND OBJECTIVES
A designated area will have been assigned for all blood specimens to be taken. All blood should then be taken in that area only.
 Venepuncture No: 008D 1. Introduction Blood sampling is a necessary requirement to assess the biochemical and haematological profile of subjects who enter clinical research studies. Risks to the subject
Venepuncture No: 008D 1. Introduction Blood sampling is a necessary requirement to assess the biochemical and haematological profile of subjects who enter clinical research studies. Risks to the subject
TEV-TROPIN [somatropin (rdna origin) for injection] 5 mg & 10 mg
![TEV-TROPIN [somatropin (rdna origin) for injection] 5 mg & 10 mg TEV-TROPIN [somatropin (rdna origin) for injection] 5 mg & 10 mg](/thumbs/74/70163471.jpg) TEV-TROPIN [somatropin (rdna origin) for injection] 5 mg & 10 mg TEV-TROPIN (somatropin for injection) Information for the Patient or Parent Read all instructions before proceeding. Do not mix (reconstitute)
TEV-TROPIN [somatropin (rdna origin) for injection] 5 mg & 10 mg TEV-TROPIN (somatropin for injection) Information for the Patient or Parent Read all instructions before proceeding. Do not mix (reconstitute)
Making Life Easier Easypump II
 Making Life Easier Easypump II The elastomeric pump system for short- and long-term infusion therapy Elastomeric Infusion Systems Technical Guide Easypump II Disposable Elastomeric Infusion Pump System
Making Life Easier Easypump II The elastomeric pump system for short- and long-term infusion therapy Elastomeric Infusion Systems Technical Guide Easypump II Disposable Elastomeric Infusion Pump System
STANDARD OPERATING PROCEDURE
 Table of Contents Page 1. Scope 2 2. Objective 2 3. Definitions 2 4. Responsibility 2 5. Procedure 3 6. Precautions 7 7. References 7 8. Appendices 7 9. Revision History 7 Original Authorised Copy for:
Table of Contents Page 1. Scope 2 2. Objective 2 3. Definitions 2 4. Responsibility 2 5. Procedure 3 6. Precautions 7 7. References 7 8. Appendices 7 9. Revision History 7 Original Authorised Copy for:
ISO 7218 INTERNATIONAL STANDARD
 INTERNATIONAL STANDARD ISO 7218 Third edition 2007-08-15 AMENDMENT 1 2013-08-01 Corrected version 2014-04-15 Microbiology of food and animal feeding stuffs General requirements and guidance for microbiological
INTERNATIONAL STANDARD ISO 7218 Third edition 2007-08-15 AMENDMENT 1 2013-08-01 Corrected version 2014-04-15 Microbiology of food and animal feeding stuffs General requirements and guidance for microbiological
PASSENGER SHIP SAFETY. Review of Operational Safety Measures to Enhance the Safety of Passenger Ships. Submitted by ICS SUMMARY
 MARITIME SAFETY COMMITTEE 92 nd session Agenda item 6 MSC 92/6/XX 2013 Original: ENGLISH PASSENGER SHIP SAFETY Review of Operational Safety Measures to Enhance the Safety of Passenger Ships Submitted by
MARITIME SAFETY COMMITTEE 92 nd session Agenda item 6 MSC 92/6/XX 2013 Original: ENGLISH PASSENGER SHIP SAFETY Review of Operational Safety Measures to Enhance the Safety of Passenger Ships Submitted by
Port - Body in a Box User s Manual
 Port - Body in a Box - 5010 User s Manual 308 South Sequoia Parkway Canby, Oregon 97013 USA ph. 503.651.5050 fax 503.651.5052 email info@vatainc.com Thank you for purchasing VATA s Port - Body in a Box
Port - Body in a Box - 5010 User s Manual 308 South Sequoia Parkway Canby, Oregon 97013 USA ph. 503.651.5050 fax 503.651.5052 email info@vatainc.com Thank you for purchasing VATA s Port - Body in a Box
limbsandthings.com Advanced Venepuncture Arm User Guide For more skills training products visit Limbs & Things Ltd.
 User Guide Advanced Venepuncture Arm Product No: 00290 (Light) Product No: 00298 (Brown) For more skills training products visit limbsandthings.com Limbs & Things Ltd. Sussex Street, St Philips Bristol,
User Guide Advanced Venepuncture Arm Product No: 00290 (Light) Product No: 00298 (Brown) For more skills training products visit limbsandthings.com Limbs & Things Ltd. Sussex Street, St Philips Bristol,
SA121, SA122, SA123, SA128 and SA1219 Self-acting Temperature Control Systems (Dial Adjustment)
 3820050/4 IM-P382-01 CH Issue 4 SA121, SA122, SA123, SA128 and SA1219 Self-acting Temperature Control Systems (Dial Adjustment) Installation and Maintenance Instructions 1. Safety information 2. Use 3.
3820050/4 IM-P382-01 CH Issue 4 SA121, SA122, SA123, SA128 and SA1219 Self-acting Temperature Control Systems (Dial Adjustment) Installation and Maintenance Instructions 1. Safety information 2. Use 3.
REB SOP 02 Page 1 of 6. Finger Prick Blood Sampling. Effective Date April Approved by REB April Version Number 1 A.
 REB SOP 02 Page 1 of 6 Short Title Finger Prick Effective Date April 2016 Approved by REB April 2016 Version Number 1 A. PURPOSE Finger prick blood sampling method is used when only a small sample is required
REB SOP 02 Page 1 of 6 Short Title Finger Prick Effective Date April 2016 Approved by REB April 2016 Version Number 1 A. PURPOSE Finger prick blood sampling method is used when only a small sample is required
Specimen Collection. Lead Screen Filter Paper Testing Dried Blood Spots (DBS) Laboratory Services February 2017
 1 Specimen Collection Lead Screen Filter Paper Testing Dried Blood Spots (DBS) Laboratory Services February 2017 2 Agenda Why Use Lead Screen Filter Paper Testing? Before You Begin Collection Drying &
1 Specimen Collection Lead Screen Filter Paper Testing Dried Blood Spots (DBS) Laboratory Services February 2017 2 Agenda Why Use Lead Screen Filter Paper Testing? Before You Begin Collection Drying &
Protocol for Blood Lactate Sampling at 2018 Pan Pacific Para Swimming Championships
 Protocol for Blood Lactate Sampling at 2018 Pan Pacific Para Swimming Championships Note: This testing procedure is only for lactate testing. Blood sampling for any other purpose is not allowed. The following
Protocol for Blood Lactate Sampling at 2018 Pan Pacific Para Swimming Championships Note: This testing procedure is only for lactate testing. Blood sampling for any other purpose is not allowed. The following
Personal Protective Equipment
 TRENT UNIVERSITY Personal Protective Equipment A guide to the use of PPE in the lab TrentEmployee 4/2/2013 [Type the abstract of the document here. The abstract is typically a short summary of the contents
TRENT UNIVERSITY Personal Protective Equipment A guide to the use of PPE in the lab TrentEmployee 4/2/2013 [Type the abstract of the document here. The abstract is typically a short summary of the contents
SA121, SA122, SA123, SA128 and SA1219 Self-acting Temperature Control Systems (Knob Adjustment)
 3810050/5 IM-P381-01 CH Issue 5 SA121, SA122, SA123, SA128 and SA1219 Self-acting Temperature Control Systems (Knob Adjustment) Installation and Maintenance Instructions 1. Safety information 2. Use 3.
3810050/5 IM-P381-01 CH Issue 5 SA121, SA122, SA123, SA128 and SA1219 Self-acting Temperature Control Systems (Knob Adjustment) Installation and Maintenance Instructions 1. Safety information 2. Use 3.
Skills & Scenarios 81
 Skills & Scenarios 81 Psychomotor Skills Checklist Skill Lab Clinical Date Initial Date Initial 1. Change I.V. tubing 2. Change I.V solution 3. Regulate I.V. flow rate 4. Flush peripheral lock 5. Change
Skills & Scenarios 81 Psychomotor Skills Checklist Skill Lab Clinical Date Initial Date Initial 1. Change I.V. tubing 2. Change I.V solution 3. Regulate I.V. flow rate 4. Flush peripheral lock 5. Change
PERFORMANCE CRITERIA
 FPICOR2006A Unit Descriptor Pre-requisite Unit(s) Application of the Unit Competency Field Sector Apply basic first aid This unit specifies the outcomes required to deal with the provision of essential
FPICOR2006A Unit Descriptor Pre-requisite Unit(s) Application of the Unit Competency Field Sector Apply basic first aid This unit specifies the outcomes required to deal with the provision of essential
HEALTH SERVICES CODE A.6 CATEGORY: RN - SNP
 NURSING PROCEDURE TITLE ARTERIAL BLOOD SAMPLING (NEONATAL) A. Umbilical Catheter Blood Collection B. Radial Arterial Line CATEGORY: RN - SNP PURPOSE To obtain blood samples for laboratory analysis NURSING
NURSING PROCEDURE TITLE ARTERIAL BLOOD SAMPLING (NEONATAL) A. Umbilical Catheter Blood Collection B. Radial Arterial Line CATEGORY: RN - SNP PURPOSE To obtain blood samples for laboratory analysis NURSING
Safety Office. Reviewed: 17 July 2012
 Safety Office Reviewed: 17 July 2012 Reviewed: DD Month Decontamination procedures This circular contains expanded guidance on the action which should be taken in the event of an incident involving radioactive
Safety Office Reviewed: 17 July 2012 Reviewed: DD Month Decontamination procedures This circular contains expanded guidance on the action which should be taken in the event of an incident involving radioactive
PREPARATION AND SELF-ADMINISTRATION INSTRUCTIONS FOR PATIENTS
 PREPARATION AND SELF-ADMINISTRATION INSTRUCTIONS FOR PATIENTS Hereditary Angioedema Jenny, a real HAE patient TAKE CONTROL OF YOUR HEREDITARY ANGIOEDEMA (HAE) TREATMENT WITH SELF-ADMINISTRATION Choosing
PREPARATION AND SELF-ADMINISTRATION INSTRUCTIONS FOR PATIENTS Hereditary Angioedema Jenny, a real HAE patient TAKE CONTROL OF YOUR HEREDITARY ANGIOEDEMA (HAE) TREATMENT WITH SELF-ADMINISTRATION Choosing
Safety Services. Introduction
 Safety Services Guidance on the Selection, Installation, Maintenance and Operation of Microbiological Safety Cabinets for use in containment level 2 (CL2) laboratories Introduction 1. Microbiological safety
Safety Services Guidance on the Selection, Installation, Maintenance and Operation of Microbiological Safety Cabinets for use in containment level 2 (CL2) laboratories Introduction 1. Microbiological safety
SOUTHBANK PLACE Asbestos Removal (Brief Guide)
 SOUTHBANK PLACE Asbestos Removal (Brief Guide) The removal of asbestos is regulated by the Control of Asbestos Regulations 2012 and is only undertaken by licensed contractors. The licensing procedures
SOUTHBANK PLACE Asbestos Removal (Brief Guide) The removal of asbestos is regulated by the Control of Asbestos Regulations 2012 and is only undertaken by licensed contractors. The licensing procedures
TEXAS DEPARTMENT OF PUBLIC SAFETY Breath Alcohol Laboratory
 TEXAS DEPARTMENT OF PUBLIC SAFETY Breath Alcohol Laboratory 5805 N LAMAR BLVD BOX 4087 AUSTIN, TEXAS 78773 512/424-5200 STEVEN C. McCRAW DIRECTOR DAVID G. BAKER CHERYL MacBRIDE DEPUTY DIRECTORS January
TEXAS DEPARTMENT OF PUBLIC SAFETY Breath Alcohol Laboratory 5805 N LAMAR BLVD BOX 4087 AUSTIN, TEXAS 78773 512/424-5200 STEVEN C. McCRAW DIRECTOR DAVID G. BAKER CHERYL MacBRIDE DEPUTY DIRECTORS January
Draft. Equipment/Instrument
 Heating Ventilation Air Conditioning (HVAC) Technical Platform Recommended specialist equipment including instruments for those working with Healthcare Ventilation Systems Those working with Healthcare
Heating Ventilation Air Conditioning (HVAC) Technical Platform Recommended specialist equipment including instruments for those working with Healthcare Ventilation Systems Those working with Healthcare
TJF-Q180V Cleaning and Disinfection Checklist
 TJF-Q180V Cleaning and Disinfection Checklist TJF-Q180V Cleaning and Disinfection Checklist This checklist is used to evaluate and confirm if cleaning and disinfection of the TJF-Q180V has been performed
TJF-Q180V Cleaning and Disinfection Checklist TJF-Q180V Cleaning and Disinfection Checklist This checklist is used to evaluate and confirm if cleaning and disinfection of the TJF-Q180V has been performed
Title of Manual: Specimen Collection Document Number: GPA To provide the proper collection protocol for Candida Vaginitis testing.
 Page 1 of 5 I._PURPOSE To provide the proper collection protocol for Candida Vaginitis testing. II. MATERIALS Reagents Supplies Equipment Cervical sampling: CytoBrush & Spatula or Broom in ThinPrep or
Page 1 of 5 I._PURPOSE To provide the proper collection protocol for Candida Vaginitis testing. II. MATERIALS Reagents Supplies Equipment Cervical sampling: CytoBrush & Spatula or Broom in ThinPrep or
MF2M Monnier Miniature Compressed Air Filter
 5040050 / 2 IM-P504-02 CH Issue 2 MF2M Monnier Miniature Compressed Air Filter Installation and Maintenance Instructions 1. Safety information 2. General product information 3. Installation and commissioning
5040050 / 2 IM-P504-02 CH Issue 2 MF2M Monnier Miniature Compressed Air Filter Installation and Maintenance Instructions 1. Safety information 2. General product information 3. Installation and commissioning
Vertebrate Animal Biosafety Level 1 Criteria
 Vertebrate Animal Biosafety Level 1 Criteria Biosafety in Microbiological and Biomedical Laboratories (BMBL) 5 th Edition Section V Animal Biosafety Level 1 (ABSL-1): Animal Biosafety Level 1 is suitable
Vertebrate Animal Biosafety Level 1 Criteria Biosafety in Microbiological and Biomedical Laboratories (BMBL) 5 th Edition Section V Animal Biosafety Level 1 (ABSL-1): Animal Biosafety Level 1 is suitable
Biosafety Level 2 (BSL2) Laboratory Safety Inspection Checklist
 Conducted By: Date of Inspection: Location: Safety and Risk Management Office Biosafety Level 2 (BSL2) Laboratory Safety Inspection Checklist Principal Investigator/Supervisor: General Lab Housekeeping
Conducted By: Date of Inspection: Location: Safety and Risk Management Office Biosafety Level 2 (BSL2) Laboratory Safety Inspection Checklist Principal Investigator/Supervisor: General Lab Housekeeping
WHS PROCEDURE P Confined Spaces. Approved by: Ruth Hampton Review Date: 6 th December 2015
 5 WHS PROCEDURE P21-01 Page 1 of 7 Issue: 2 1. Intent The Salvation Army Australia Eastern Territory (TSA-AUE) is committed to the provision of a safe, healthy and injury free environment for all persons
5 WHS PROCEDURE P21-01 Page 1 of 7 Issue: 2 1. Intent The Salvation Army Australia Eastern Territory (TSA-AUE) is committed to the provision of a safe, healthy and injury free environment for all persons
Users Guide for the Vertical Bunsen Burner Test for Cabin and Cargo Compartment Materials FAA Handbook Chapter 2
 Issue 1.1: May 2010 REF: FAA Chapter 2 Bunsen Burner Test Manual.doc Users Guide for the Vertical Bunsen Burner Test for Cabin and Cargo Compartment Materials FAA Handbook Chapter 2 Issued by: Fire Testing
Issue 1.1: May 2010 REF: FAA Chapter 2 Bunsen Burner Test Manual.doc Users Guide for the Vertical Bunsen Burner Test for Cabin and Cargo Compartment Materials FAA Handbook Chapter 2 Issued by: Fire Testing
Chapter 40. Fluid, Electrolyte, and Acid Base Balance. Procedures Checklist INTRAVENOUS THERAPY. Procedure 40.1: Starting an Intravenous Infusion
 Procedures Checklist INTRAVENOUS THERAPY Chapter 40 Fluid, Electrolyte, and Acid Base Balance Procedure 40.1: Starting an Intravenous Infusion Performed Preparation Yes No Mastered Comments 1. Assess:
Procedures Checklist INTRAVENOUS THERAPY Chapter 40 Fluid, Electrolyte, and Acid Base Balance Procedure 40.1: Starting an Intravenous Infusion Performed Preparation Yes No Mastered Comments 1. Assess:
Blood Pressure Monitoring: Arterial Line
 Approved by: Blood Pressure Monitoring: Arterial Line Gail Cameron Senior Director, Operations, Maternal, Neonatal & Child Health Programs Dr. Ensenat Medical Director, Neonatology Neonatal Nursery Policy
Approved by: Blood Pressure Monitoring: Arterial Line Gail Cameron Senior Director, Operations, Maternal, Neonatal & Child Health Programs Dr. Ensenat Medical Director, Neonatology Neonatal Nursery Policy
HAZCOMM & Lab Safety College of Dental Medicine Research Safety Specialist Environmental Health and Safety
 HAZCOMM & Lab Safety College of Dental Medicine Specialist Environmental Health and Safety Purpose of Training Crucial for a safe work place Policy/procedures may vary Required by Occupational Safety &
HAZCOMM & Lab Safety College of Dental Medicine Specialist Environmental Health and Safety Purpose of Training Crucial for a safe work place Policy/procedures may vary Required by Occupational Safety &
Vacuum Excavation in the vicinity of the Wellington Electricity Network
 Vacuum Excavation in the vicinity of the Network. Document no: ENG-116 Document Rev: 1 Issue Status: Final Issue Date: 14/05/2015 Page 1 of 9 Contents 1. PURPOSE... 3 2. POLICY... 3 3. SCOPE... 3 4. REFERENCES...
Vacuum Excavation in the vicinity of the Network. Document no: ENG-116 Document Rev: 1 Issue Status: Final Issue Date: 14/05/2015 Page 1 of 9 Contents 1. PURPOSE... 3 2. POLICY... 3 3. SCOPE... 3 4. REFERENCES...
WHS PROCEDURE P Plant Isolation. Approved by: Ruth Hampton Review Date: 6 th December 2015
 5 WHS PROCEDURE P29-02 Page 1 of 7 Issue: 2 1. Intent The Salvation Army Australia Eastern Territory (TSA-AUE) is committed to the provision of a safe, healthy and injury free environment within the mission
5 WHS PROCEDURE P29-02 Page 1 of 7 Issue: 2 1. Intent The Salvation Army Australia Eastern Territory (TSA-AUE) is committed to the provision of a safe, healthy and injury free environment within the mission
6800 Maintenance Instruction System Flush Procedure
 Equipment Required FA74005 FA65318 FA900005 FA900003 Damper Drain Tube 6800 Cover Removal Tool Beaker 0.25 Litre Solvent Cleaning Bottle FA940021 Syringe Polypropylene 50 ml as required FA999045 Gloves
Equipment Required FA74005 FA65318 FA900005 FA900003 Damper Drain Tube 6800 Cover Removal Tool Beaker 0.25 Litre Solvent Cleaning Bottle FA940021 Syringe Polypropylene 50 ml as required FA999045 Gloves
POWER LIFT LOW PRESSURE LIFTING BAGS. Instructions for use
 POWER LIFT LOW PRESSURE LIFTING BAGS Instructions for use The system of WEBER-HYDRAULIK low pressure lifting bags of the 0.5 and 1.0 bar series is used particularly for rescuing persons captured in various
POWER LIFT LOW PRESSURE LIFTING BAGS Instructions for use The system of WEBER-HYDRAULIK low pressure lifting bags of the 0.5 and 1.0 bar series is used particularly for rescuing persons captured in various
Parts identification:
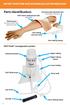 ARTERY PUNCTURE AND INTRAMUSCULAR TRAINING ARM Parts identification: FAST-derm replacement skin (removable) Intramuscular injection pad (removable for cleaning) Training arm (includes one FAST-derm replacement
ARTERY PUNCTURE AND INTRAMUSCULAR TRAINING ARM Parts identification: FAST-derm replacement skin (removable) Intramuscular injection pad (removable for cleaning) Training arm (includes one FAST-derm replacement
Infant IV Leg LF03636U Instruction Manual
 Infant IV Leg LF03636U Instruction Manual Products by Nasco About the Simulator The Life/form Infant IV Leg Simulator is an exciting training aid for practicing and demonstrating intravenous puncture
Infant IV Leg LF03636U Instruction Manual Products by Nasco About the Simulator The Life/form Infant IV Leg Simulator is an exciting training aid for practicing and demonstrating intravenous puncture
TECHNICAL BULLETIN. TriControls. Control Materials for Liquid QC and Calibration Verification
 i-stat TECHNICAL BULLETIN Tris Materials for Liquid QC and OVERVIEW As part of the READi initiative (Responds, Enhances, And Delivers innovations), Abbott Point of Care (APOC) has released a new set of
i-stat TECHNICAL BULLETIN Tris Materials for Liquid QC and OVERVIEW As part of the READi initiative (Responds, Enhances, And Delivers innovations), Abbott Point of Care (APOC) has released a new set of
Contractor Control Procedures. Contractor Control Procedures. Working Together. November Borders College 24/11/2014.
 Working Together Contractor Control Procedures November 2014 Borders College 24/11/2014 1 Working Together 1. Introduction This procedure aims to control the health and safety of contractors working on
Working Together Contractor Control Procedures November 2014 Borders College 24/11/2014 1 Working Together 1. Introduction This procedure aims to control the health and safety of contractors working on
