Submarine Base, Groton, Conn.
|
|
|
- Emery Merritt
- 5 years ago
- Views:
Transcription
1 Submarine Base, Groton, Conn. REPORT NUMBER 554 OBSERVATIONS OF INTRA-OCULAR PRESSURE CHANGES DURING AN FBM PATROL AS MEASURED BY SCHIOTZ TONOMETRY by James G. Geisinger Lieutenant, MC, U. S. Navy Bureau of Medicine and Surgery, Navy Department Research Work Unit MR Released by: Gerald J. Duffnef, CAPT MC USN COMMANDING OFFICER Naval Submarine Medical Center 21 October 1968 This document has been approved for public release and sale; its distribution is unlimited.
2 OBSERVATIONS OF INTRAOCULAR PRESSURE CHANGES DURING AN FBM PATROL AS MEASURED BY SCHIOTZ TONOMETRY by James G. Geisinger Lieutenant, MC, U. S. Navy SUBMARINE MEDICAL RESEARCH LABORATORY NAVAL SUBMARINE MEDICAL CENTER REPORT NUMBER 554 Bureau of Medicine and Surgery, Navy Department Research Work Unit MR Reviewed and Approved by: Reviewed and Approved by: ^/L^e^?- ^^ Charles F. Gell, M.D. Scientific Director SubMedResLab Joseph D. Bloom, CDR, MC, USN Director SubMedResLab Approved and Released by: Gerald J. Duffner CAPTAIN, MC, USN COMMANDING OFFICER, SMC
3 SUMMARY PAGE THE PROBLEM To determine whether a hazardous increase in intra-ocular pressure occurs as a result of elevated carbon dioxide levels in the atmosphere during Fleet Ballistic Missile (FBM) submarine patrols. FINDINGS Baseline ocular tensions were taken on 16 volunteers. Serial determinations were taken at weekly intervals on these same volunteers during an FBM patrol. No significant elevation above the baseline values was found in this group. APPLICATIONS These results can serve to reassure those responsible for the health of submarine crews that increased intra-ocular pressure is not a hazard attendant on prolonged submergence. ADMINISTRATIVE INFORMATION This report was prepared by the author in partial fulfillment of the requirements for qualification as a Submarine Medical Officer. It was approved for publication as Report No. 1 on In-House Research Work Unit MR , under date of 21 October 1968, in order that copies might be available in the School of Submarine Medicine and in the Technical Library, Submarine Medical Center, for study and reference purposes for students of submarine medicine. This document has been approved for public release and sale; its distribution is unlimited. PUBLISHED BY THE NAVAL SUBMARINE MEDICAL CENTER
4 ABSTRACT This paper discusses the method of establishing that no hazard of increased intra-ocular pressure is incurred from the elevated carbon dioxide content of the sealed submarine atmosphere. The reasons for suspecting a possible intra-ocular pressure rise under such conditions are presented. The serial tonometry readings, both under control conditions and at elevated ambient carbon dioxide levels while on patrol are shown. The conclusion from this study is that no elevation of intra-ocular pressure accompanies physiologic adjustments to high carbon dioxide levels.
5
6 TABLE OF CONTENTS Page INTRODUCTION 1 MATERIALS AND METHODS 2 RESULTS 2 DISCUSSION 3 CONCLUSIONS 5 REFERENCES 6 LIST OF FIGURES Figure No. Title 1 Histogram of Baseline Ocular Tensions 4 2 Mean Patrol Ocular Tensions 4 3 Number of Patrols vs Ocular Tension 5 LIST OF TABLES Table Title A Baseline Ocular Tensions 2 B Ocular Tensions During Patrol 2
7 OBSERVATIONS OF INTRAOCULAR PRESSURE CHANGES DURING AN FBM PATROL AS MEASURED BY SCHIÖTZ TONOMETRY INTRODUCTION The development of equipment able to artificially produce oxygen, and remove carbon dioxide and carbon monoxide from the atmosphere enables nuclear submarines to remain submerged for periods far in excess of those previously feasible. The use of this equipment has meant that submariners can and do live in a truly closed atmosphere environment. This has presented a most complex problem to those involved in caring for the health of submarine personnel, i.e., what levels of atmospheric constituents and contaminants, in a closed environment, can be tolerated by the human organism, without serious pathophysiologic effects? Because of the large number of gases and particulate substances involved, and the large number of possible physiologic effects, study and revision of the acceptable limits of atmospheric constituents is a continuous process. The subject of this report is one possible pathophysiologic effect of elevated carbon dioxide concentrations in the submarine atmosphere. Schaefer, et al 1;i have demonstrated that an uncompensated respiratory acidosis occurs in those exposed to an atmosphere with 1.5 percent carbon dioxide continuously for a period of six weeks. It can be concluded from these studies that the ambient level of carbon dioxide has a direct effect on the bicarbonate-carbonic acid plasma buffering system. Therefore, it would seem advisable to study those organ systems whose function is tied up with the bicarbonate-carbonic acid buffering system, for the purpose of determining whether any detrimental changes in their function occur. The formation and absorption of aqueous humor,* in the posterior and anterior chambers of the eye, respectively, are functions *Wherever the word aqueous occurs subsequently in this report it is to be understood that aqueous humor is meant. which appear to be related to the bicarbonatecarbonic acid buffer system. While the mechanism of aqueous formation is not fully understood,"''" it appears that a combination of secretion and nitration is involved. It is known that the anion bicarbonate is present in lower concentrations in aqueous than in plasma," and that the total CO- in aqueous is 63 volumes percent while the level in plasma is 55 to 75 volumes percent. 4 In trying to understand the possible relationship between carbon dioxide and intraocular pressure, it is well to"examine equation 1: (1) a b H,0 + CO^=Jf 2 CO,^H + + HCO:< - Reaction (a) proceeds slowly in the absence of a catalyst, while reaction (b) proceeds very rapidly." However, when the enzyme carbonic anhydrase is added to reaction (a), it will increase the speed of the reaction one hundredfold. Two phenomena of interest occur when an inhibitor of carbonic anhydrase (CA), such as acetazolamide, is administered to a patient. First, reaction (a) is greatly slowed, as in equation 2: (2) H,0 + CO^HXO^H" 1 " + HCOa" Second, intra-ocular pressure is reduced, through decreased aqueous formation. This would lead us to believe either equation 3 or equation 4 contributes to the formation of aqueous, and that interfering with one re- (3) H,0 + CO, CA^H2COI ^H+ _ _ HCO:, - (4) H f + HCOs - > H.COs -24 H CO, action or the other is what causes the decrease in aqueous formation. Proceeding from that point, if we increase *carbonic anhydrase = CA
8 the alveolar pc0 2 we will drive equation 1 to the right, a situation which must be found in carbonic anhydrase containing tissues during the period of adaptation to elevated carbon dioxide levels, such as submarine patrols. It is logical that if reaction 3 is the reaction which aids in the formation of aqueous, an increase in ambient pc0 2 will tend to cause more aqueous to form and the intraocular pressure to increase. If, on the other hand, reaction 4 tends to produce aqueous, an increase in ambient pc0 2 will slow the reaction and decrease both aqueous formation and intra-ocular pressure. To repeat, one of these two must logically be the case from the information already presented. Based on this single factor, the intraocular pressure would seem not to remain the same during a submarine patrol, but would either increase or decrease. The change, however, might be so small that it would not be detectable by ordinary methods, or it might be neutralized or overriden by other factors concurrently affecting intraocular pressure. The magnitude of change must be arrived at empirically. In any case it would appear to be in the interest of submariners to make certain that the elevated levels of carbon dioxide in nuclear submarine atmospheres during prolonged submergence does not elevate intraocular pressure to a dangerous level. The purpose of this paper is to report on Schiotz tonometry done on sixteen volunteers during one patrol aboard the USS ROBERT E. LEE (SSBN 601), with the express purpose of examining for this phenomenon. In order to keep this paper unclassified, no specific percentages of carbon dioxide in the atmosphere will be given. Those for whom this report is primarily intended will be familiar with the atmosphere control equip- ment on board FBM submarines 7 ' 8 and will be aware of the carbon dioxide partial pressures ordinarily encountered. The partial pressures during the patrol in question differed in no major respect from the norm. It is the opinion of this author that revealing day to day and hour to hour carbon dioxide partial pressures would add little to inquiry on this study's primary question. It can, however, be stated that while daily variation did occur in the partial pressure of carbon dioxide, no trend in these values was discernable. MATERIALS AND METHODS A standardized and certified Schiotz tonometer was used for the measurements of intra-ocular pressure. Two drops of 0.5% tetracaine solution provided sufficient anesthesia for the procedure. Care was, of course, taken to disinfect the tonometer between patients. No morbidity arose from the procedure. Sixteen volunteers, ranging in age from 21 to 35 were recruited from among the submarine crew. None of these volunteers had a history of serious eye disease, although two had a family history of glaucoma. R.G.'s father had glaucoma, controlled with the use of miotics. M.W.M.'s father had glaucoma which remained in remission following an iridectomy. No other members of either family have been similarly afflicted. Three, and in some cases four, baseline tonometry readings were taken on each volunteer, while in the normal ambient carbon dioxide level of 0.04%. Subsequently tonometry was done at weekly intervals, corresponding to the end of each week spent in the high carbon dioxide atmosphere. The advantage of this method is that each subject served as his own control. His readings during the experimental period could be compared with his readings during normal carbon dioxide levels. Each eye is considered individually, since each is essentially a separate physiologic unit. RESULTS Three or four control ocular tensions were recorded for each volunteer. The observations were made at weekly intervals. The findings are recorded in millimeters of mercury pressure in Table A. The mean values and standard deviations are significant only for the first, second, and fourth weeks, when all volunteers were observed. These values
9 were 18.6 ± 3.7 mm Hg, 19.8 ± 3.6 mm Hg, and 19.4 ± 3.7 mm Hg for the first, second, and fourth weeks respectively. The mean for all values taken on the three occasions was 19.3 ± 3.6 mm Hg. o.e. TABLE A BASELINE OCULAR TENSIONS Week l Av. L.B. O.D O.S W.B. O.E O.S O.D elevated carbon dioxide levels, the value for week two the end of the second week, etc. The mean values for all observations are as follows: week one 19.0 ± 4.1 mm Hg, week two 18.5 ± 3.4 mm Hg, week three 18.0 ± 3.6 mm Hg, week four 17.6 ± 4.3 mm Hg, week five 17.9 ± 4.0 mm Hg, week six 18.0 ± 4.4 mm H g. The se v alues are g rapm :ally represented in Figure 2. TABLE B OCULAR TENSIONS DURING PATROL O.S Week l R.C. O.D L.B. O.D O.S O.S E.F. O.D W.B. O.D O.S O.S R.G. O.D o.e. O.D O.S O.S L.L. O.D R.C. O.D O.S O.S F.L. O.D E.P. O.D L O.S O.S B.D.M. O.D R.G. O.D O.S O.S B.W.M. O.D L.L. O.D. 20.1! O.S O.S E.P. O.D F.L. O.D O.S O.S R.P. O.D B.D.M. O.D O.S O.S R.S. O.D B.W.M O.D O.S , O.S J.V. O.D E.P. O.D O.S O.S B.N. O.D R.P. O.D J.B. O.S O.S O.D R.S. O.D O.S O.S The mean control intra-ocular pressure for each eye is also recorded in the right hand J.B. O.D column o f Table A. A histo arram of the dis- O.S tribution of the latter values is shown in Figure 1. The distribution is roughly a gaus- sian curve. The mean is coincident with the mean value for the population as a whole. Table B presents the intra-ocular pressures found in the same individuals during a submarine patrol. In this table, the value for week one is the ocular tension observed at the end of the first week of exposure to J.V. B.N. O.D. O.S. O.D. O.S DISCUSSION In the analysis of the data here presented, it is necessary to examine both the group of volunteers as a whole, and each individual. This is necessary in order to ascertain not only that no hazard exists for submarine personnel as a group, but also that no hazard
10 exists to special classifications of submariners. Particularly of interest are those with high normal intra-ocular pressures, and those with a family history of glaucoma mm Hg mm Hg mm Hg mm Hg mm Hg mm Hg mm Hg Figure 1. Distribution of Baseline Intra-Ocular Pressures allow retrospective tracing of trends from individual health records. Such is the case with the sixteen volunteers in this study. One can plot the number of Polaris patrols a submariner has made against his baseline intra-ocular pressure, as in Figure 3. This gives a guide, albeit a crude one, as to the presence or absence of a cumulative effect on ocular tension of repeated prolonged exposures to elevated carbon dioxide levels. The scattergram in Figure 3 demonstrates no tendency to cumulative displacement of ocular tension upward or downward with longer tenure on Polaris submarines. It must be realized that differing natural baseline values add a confusion factor to such a graph. Serially following a group of submarine personnel would be preferable, as already discussed, but is beyond the scope of this study. The drop between the baseline tensions and those of patrol week four is significant at the.01 level, so sampling phenomena are not the cause. 10 The significance of the initial drop in ocular tension is somewhat obscure, and the effect is certainly contrary to the author's a priori expectations. Several possible reasons present themselves. The first In studying the group of volunteers, that is the mean intra-ocular pressure values throughout patrol, it is clear no consistent elevation of ocular tension occurs. In fact, during the period in which one would expect an uncompensated respiratory acidosis 1, the mean ocular tension steadily decreased. Following this, during what would correspond to the period of compensated respiratory acidosis, the mean value fluctuated within the normal range. It never exceeded the highest baseline value, however. This would lead us to believe that for the vast majority of submariners, the submerged environment presents no hazard to eyesight from an ocular tension rise. Some corroboration of this could be derived from serially following the tonometry readings for submariners over a number of patrols. However, tonometry is not included in routine examinations often enough to ocular tension 20 mmhg 17 mm Hg mean mean mean mean mean mean mean baseline wk 1 wk 2 wk 3 wk 4 wk S wk 6 Figure 2 Mean Patrol Ocular Tensions is that this is a result of random sampling of unchanged tensions, and not a true drop in ocular tension at all. This could be better explored by repeating the present study with
11 a larger group of volunteers. A second possibility is that reaction four on page 1 is in fact a reaction which contributes to aqueous formation. A third possibility is that some other factor, either an atmospheric constituent or a feature of life aboard FBM submarines, causes a decrease in ocular tension which overrides any effect of high carbon dioxide. The author cannot, on the basis of this study's design or results, make any definitive choice between these possibilities. Repeating the investigation to see if the results can be duplicated by other workers, may be helpful in reaching an answer. When we scrutinize each volunteer we find only one who appears to have a consistent elevation of ocular tension while on patrol. This is B.W.M., whose family history of glaucoma has already been discussed. R.G., with a similar history, had no elevation while on patrol. This is far too small a sample from which to draw any firm conclusions regarding the safety of carbon dioxide rich cedure would be to seek out similar individuals throughout the submarine force, and obtain comparative measurements ashore and while deployed. CONCLUSIONS In conclusion, there is one inference which can be drawn from the data presented, but three questions are raised. The inference is that exposure to the carbon dioxide levels common on Polaris submarines does not elevate the ocular tension in normal individuals. The questions raised are interrelated. Can the mean decrease in intra-ocular pressure during patrol be reproduced by other observers? What is the cause of this decrease, if indeed, the phenomenon can be confirmed? The third question, and the most pressing from an operational viewpoint, is whether a small population group, with a pre-disposition for glaucoma, is endangered by the submarine atmosphere. ocular tension 28 mm Hg 26 mm Hg^ 24 mm Hg. 22 mm Hg_ 20 mm Hg_ 18 mm Hg_ 16 mm Hg- 14 mm Hg_ 12 mm Hg- x X X X X X 10 patrols Figure 3 Number of Patrols vs Ocular Tension atmospheres for those with a genetic substrate containing a glaucoma factor. However, B.W.M.'s ocular tension changes are enough to raise some doubt. A logical pro- The first and last of these questions can be answered by further careful observation along the lines of this study! To answer the second question will be more challenging, and perhaps ultimately more rewarding.
12 REFERENCES 1. Schaefer, K. E.; Nichols, G., Jr.; and Carey, C. R.: "Acid-Base Balance and Blood and Urine Electrolytes of Man During Acclimatization to Carbon Dioxide"; J. Appl. Physiol.; 19:48; Schaefer, K. E., et al: "Respiratory Acclimatization to Carbon Dioxide"; J. Appl. Physiol.; 18: ; Schaefer, K. E.: "Environmental Physiology of Submarines and Spacecraft (Atmospheric Requirements of Confined Spaces)"; Arch of Environ. Health; 9: ; West, E. S.; and Todd, W. R.: "Textbook of Biochemistry"; Macmillan, New York; Best, C. H.; and Taylor, N. B.: "The Physiological Basis of Medical Practice"; Williams and Wilkins; Baltimore; Adler, F. H.; "Textbook of Ophthalmology"; Saunders; Schillachi, R. F.: "Control of the Chemical Constituents of the Submarine Atmosphere"; U.S. Naval Submarine Medical Center Report No "Submarine Atmosphere Habitability Data Book", NAVSHIPS (Revision 1); September, Goodman, L. S.; and Gilman, A.: "The Pharmacological Basis of Therapeutics"; Macmillan; New York; 1965.
13 UNCLASSIFIED Security Classification DOCUMENT CONTROL DATA -R&D (Security classification of title, body of abstract and indexing annotation must be entered wh/en the overall report Is classified) l. ORIGINATING ACTIVITY (Corporate author) NAVAL SUBMARINE MEDICAL CENTER, Submarine Medical Research Laboratory 2a. REPORT SECURITY CLASSIFICATION UNCLASSIFIED 2*. GROUP 3. REPORT TITLE INTRA-OCULAR PRESSURE CHANGES DURING FBM SUBMARINE PATROLS AS MEASURED BY SCHIOTZ TONOMETRY 4. DESCRIPTIVE NOTES (Type ot report and inclusive dates) Interim report 5. AUTHOR(S) (First name, middle initial, last name) James G. Geisinger, Lieutenant, MC, U. S. Navy 6. REPORT DATE 21 October a. CONTRACT OR GRANT NO. b. PROJECT NO. la. TOTAL NO. OF PAGES 6 9a. ORIGINATOR'S REPORT NUMBER(S) SMRL Report Number 554 lb. NO. OF REFS 9 9b. OTHER REPORT NOISI (Any other numbers that may be assigned this report) 10. DISTRIBUTION STATEMENT This document has been approved for public release and sale; its distribution is unlimited. II. SUPPLEMENTARY NOTES 13. ABSTRACT 12. SPONSORING MILITARY ACTIVITY Naval Submarine Medical Center Box 600 Naval Submarine Base Groton, Connecticut This paper discusses the method of establishing that no hazard of increased intraocular pressure is incurred from the elevated carbon dioxide content of the sealed submarine atmosphere. The reasons for suspecting a possible intra-ocular pressure rise under such conditions are presented. The serial tonometry readings, both under control conditions and at elevated ambient carbon dioxide levels while on patrol are shown. The conclusion from this study is that no elevation of intra-ocular pressure accompanies physiologic adjustments to high carbon dioxide levels. DD FORM I47O I NOV 65 I *t / W S/N (PAGE 1) UNCLASSIFIED Security ClassificaUon 3ND PPSO 13152
14 UNCLASSIFIED Security Classification KEY WO RDS ROLE WT Carbon dioxide, effects of increased Submarine atmosphere Carbon dioxide, adjustment to elevated percentages Intra-ocular pressure measurement in submarines DD JT.A473 (BACK, (PAGE 2) UNCLASSIFIED Security Classification
THE EFFECT OF INTERMITTENT EXPOSURE TO 3% C0 2 ON ACID-BASE BALANCE AND ELECTROLYTE EXCRETION
 THE EFFECT OF INTERMITTENT EXPOSURE TO 3% C0 2 ON ACID-BASE BALANCE AND ELECTROLYTE EXCRETION by K. E. Schaefer C. C. Morgan A. A. Messier and M. J. Jacey SUBMARINE MEDICAL RESEARCH LABORATORY NAVAL SUBMARINE
THE EFFECT OF INTERMITTENT EXPOSURE TO 3% C0 2 ON ACID-BASE BALANCE AND ELECTROLYTE EXCRETION by K. E. Schaefer C. C. Morgan A. A. Messier and M. J. Jacey SUBMARINE MEDICAL RESEARCH LABORATORY NAVAL SUBMARINE
u. %N&tMz2&foi&INE MEQpa*,G» ER Submarine Base, Groton, Conn. REPORT NUMBER 566 EFFECT ON VISION OF REPEATED EXPOSURE TO CARBON DIOXIDE
 J u. %N&tMz2&foi&INE MEQpa*,G» ER Submarine Base, Groton, Conn. REPORT NUMBER 566 EFFECT ON VISION OF REPEATED EXPOSURE TO CARBON DIOXIDE by Donald 0. Weitzman, Jo Ann S. Kinney and S. M. Luria Bureau
J u. %N&tMz2&foi&INE MEQpa*,G» ER Submarine Base, Groton, Conn. REPORT NUMBER 566 EFFECT ON VISION OF REPEATED EXPOSURE TO CARBON DIOXIDE by Donald 0. Weitzman, Jo Ann S. Kinney and S. M. Luria Bureau
Submarine Base, Groton, Conn. REPORT NUMBER 668
 ARINE.W Submarine Base, Groton, Conn. REPORT NUMBER 668 HYPERBARIC AIR AND CORNEAL VASCULARIZATION by Robert J. Kaiser CDRMCUSNR and Donald W. Klopp LCDR MC USN Bureau of Medicine and Surgery, Navy Department
ARINE.W Submarine Base, Groton, Conn. REPORT NUMBER 668 HYPERBARIC AIR AND CORNEAL VASCULARIZATION by Robert J. Kaiser CDRMCUSNR and Donald W. Klopp LCDR MC USN Bureau of Medicine and Surgery, Navy Department
Submarine Base, Groton, Conn.
 r*-! PUBLICATIONS B:. t,,..r f^r-.! j.'fdical C3&7ER L, L>. t-."-- - -» JJ ff? ^ *W -'«--'fr- *" ^ 5 --^--fc-is*^ V»?g ««.»1/ j m : :# V j Jf «r% i ls «"% *Ä\ - ' > /vjv*äs.. /> Submarine Base, Groton,
r*-! PUBLICATIONS B:. t,,..r f^r-.! j.'fdical C3&7ER L, L>. t-."-- - -» JJ ff? ^ *W -'«--'fr- *" ^ 5 --^--fc-is*^ V»?g ««.»1/ j m : :# V j Jf «r% i ls «"% *Ä\ - ' > /vjv*äs.. /> Submarine Base, Groton,
CHAPTER 16 %UHDWKLQJ*DV0L[LQJ3URFHGXUHV
 CHAPTER 16 %UHDWKLQJ*DV0L[LQJ3URFHGXUHV 16-1 INTRODUCTION 16-1.1 Purpose. The purpose of this chapter is to familiarize divers with the techniques used to mix divers breathing gas. 16-1.2 Scope. This chapter
CHAPTER 16 %UHDWKLQJ*DV0L[LQJ3URFHGXUHV 16-1 INTRODUCTION 16-1.1 Purpose. The purpose of this chapter is to familiarize divers with the techniques used to mix divers breathing gas. 16-1.2 Scope. This chapter
 Question 1: Define vital capacity. What is its significance? Vital capacity is the maximum volume of air that can be exhaled after a maximum inspiration. It is about 3.5 4.5 litres in the human body. It
Question 1: Define vital capacity. What is its significance? Vital capacity is the maximum volume of air that can be exhaled after a maximum inspiration. It is about 3.5 4.5 litres in the human body. It
UNDERWATER HEARING IN MAN: II. A Comparison of Temporary Threshold Shifts Induced by 3500 Hertz Tones in Air and Underwater
 I UNDERWATER HEARING IN MAN: II. A Comparison of Temporary Threshold Shifts Induced by 3500 Hertz Tones in Air and Underwater by Paul F. Smith Robert Howard Martin Harris and Day Waterman SUBMARINE MEDICAL
I UNDERWATER HEARING IN MAN: II. A Comparison of Temporary Threshold Shifts Induced by 3500 Hertz Tones in Air and Underwater by Paul F. Smith Robert Howard Martin Harris and Day Waterman SUBMARINE MEDICAL
MEDICAL GENTER. Submarine Base, Groton, Conn. REPORT NUMBER 580 DECOMPRESSION PATTERNS DEVELOPED BY AN INTERDEPENDENT ELECTRIC ANALOG
 MEDICAL GENTER Submarine Base, Groton, Conn. REPORT NUMBER 580 DECOMPRESSION PATTERNS DEVELOPED BY AN INTERDEPENDENT ELECTRIC ANALOG by Gary P. Todd Lieutenant, MC, U. S. Naval Reserve Bureau of Medicine
MEDICAL GENTER Submarine Base, Groton, Conn. REPORT NUMBER 580 DECOMPRESSION PATTERNS DEVELOPED BY AN INTERDEPENDENT ELECTRIC ANALOG by Gary P. Todd Lieutenant, MC, U. S. Naval Reserve Bureau of Medicine
HCO - 3 H 2 CO 3 CO 2 + H H H + Breathing rate is regulated by blood ph and C02. CO2 and Bicarbonate act as a ph Buffer in the blood
 Breathing rate is regulated by blood ph and C02 breathing reduces plasma [CO2]; plasma [CO2] increases breathing. When C02 levels are high, breating rate increases to blow off C02 In low C02 conditions,
Breathing rate is regulated by blood ph and C02 breathing reduces plasma [CO2]; plasma [CO2] increases breathing. When C02 levels are high, breating rate increases to blow off C02 In low C02 conditions,
Submarine Base, Groton, Conn.
 LICAIIONS BRANCH. fjrim MEDICAL CENTER tlle COPY NO. -*- 1 J Submarine Base, Groton, Conn. REPORT NUMBER 697 THE INTELLIGIBILITY OF PROCESSED HELIUM SPEECH AT DIVING DEPTHS GREATER THAN 1000 FEET by Thomas
LICAIIONS BRANCH. fjrim MEDICAL CENTER tlle COPY NO. -*- 1 J Submarine Base, Groton, Conn. REPORT NUMBER 697 THE INTELLIGIBILITY OF PROCESSED HELIUM SPEECH AT DIVING DEPTHS GREATER THAN 1000 FEET by Thomas
Oxygen convulsions are believed by many workers to be caused by an accumulation
 272 J. Physiol. (I949) I09, 272-280 6I2.223.II:6I2.26I THE ROLE OF CARBON DIOXIDE IN OXYGEN POISONING BY H. J. TAYLOR From the Royal Naval Physiological Laboratory, Alverstoke, Hants (Received 26 March
272 J. Physiol. (I949) I09, 272-280 6I2.223.II:6I2.26I THE ROLE OF CARBON DIOXIDE IN OXYGEN POISONING BY H. J. TAYLOR From the Royal Naval Physiological Laboratory, Alverstoke, Hants (Received 26 March
SUBMARINE BASE, GROTON, CONN. REPORT NUMBER 855
 NAVAL RESEA DICAL ÄTORY SUBMARINE BASE, GROTON, CONN. REPORT NUMBER 855 IMPLEMENTING AN EXCHANGE FROM NITROGEN TO HELIUM IN A LARGE HYPERBARIC CHAMBER by. Kenneth R. Bondi, D. Douglas Wray and James E.
NAVAL RESEA DICAL ÄTORY SUBMARINE BASE, GROTON, CONN. REPORT NUMBER 855 IMPLEMENTING AN EXCHANGE FROM NITROGEN TO HELIUM IN A LARGE HYPERBARIC CHAMBER by. Kenneth R. Bondi, D. Douglas Wray and James E.
CALIBRATION OF TONOMETERS*
 Brit. J. Ophthal. (1955) 39, 368. CALIBRATION OF TONOMETERS* BY C. R. S. J-ACKSONt Edinburgh THE steps which led to the current attempt to introduce standardized tonometers into the United Kingdom have
Brit. J. Ophthal. (1955) 39, 368. CALIBRATION OF TONOMETERS* BY C. R. S. J-ACKSONt Edinburgh THE steps which led to the current attempt to introduce standardized tonometers into the United Kingdom have
Section Three Gas transport
 Section Three Gas transport Lecture 6: Oxygen transport in blood. Carbon dioxide in blood. Objectives: i. To describe the carriage of O2 in blood. ii. iii. iv. To explain the oxyhemoglobin dissociation
Section Three Gas transport Lecture 6: Oxygen transport in blood. Carbon dioxide in blood. Objectives: i. To describe the carriage of O2 in blood. ii. iii. iv. To explain the oxyhemoglobin dissociation
The effect of external compression of the eye on intraocular pressure
 -ff r r JL. The effect of external compression of the eye on intraocular pressure I. Its variations with magnitude of compression and with age Mansour F. Armaly and Adrian H. Halasa i $ V' y. A The effect
-ff r r JL. The effect of external compression of the eye on intraocular pressure I. Its variations with magnitude of compression and with age Mansour F. Armaly and Adrian H. Halasa i $ V' y. A The effect
Submarine Base, Groton, Conn.
 Us E Submarine Base, Groton, Conn. REPORT NUMBER DECOMPRESSION PATTERNS DEVELOPED BY AN INTERDEPENDENT ELECTRIC ANALOG by Gary P. Todd Lieutenant, MC, U. S. Naval Reserve Bureau of Medicine and Surgery,
Us E Submarine Base, Groton, Conn. REPORT NUMBER DECOMPRESSION PATTERNS DEVELOPED BY AN INTERDEPENDENT ELECTRIC ANALOG by Gary P. Todd Lieutenant, MC, U. S. Naval Reserve Bureau of Medicine and Surgery,
These two respiratory media (air & water) impose rather different constraints on oxygen uptake:
 Topic 19: OXYGEN UPTAKE AND TRANSPORT (lectures 29-30) OBJECTIVES: 1. Be able to compare air vs. water as a respiratory medium with respect to oxygen content, diffusion coefficient, viscosity and water
Topic 19: OXYGEN UPTAKE AND TRANSPORT (lectures 29-30) OBJECTIVES: 1. Be able to compare air vs. water as a respiratory medium with respect to oxygen content, diffusion coefficient, viscosity and water
Essential Skills Course Acute Care Module. Respiratory Day 2 (Arterial Blood Gases) Pre course Workbook
 Essential Skills Course Acute Care Module Respiratory Day 2 (Arterial Blood Gases) Pre course Workbook Acknowledgements This pre course workbook has been complied and updated with reference to the original
Essential Skills Course Acute Care Module Respiratory Day 2 (Arterial Blood Gases) Pre course Workbook Acknowledgements This pre course workbook has been complied and updated with reference to the original
A COMPRESSION-DECOMPRESSION SCHEDULE FOR PRODUCING DYSBARIC STRESS IN MATURE RATS. Michael J. Jacey Donald V. Tappan and John J.
 A COMPRESSION-DECOMPRESSION SCHEDULE FOR PRODUCING DYSBARIC STRESS IN MATURE RATS by Michael J. Jacey Donald V. Tappan and John J. Wojtowicz NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY REPORT NUMBER 749
A COMPRESSION-DECOMPRESSION SCHEDULE FOR PRODUCING DYSBARIC STRESS IN MATURE RATS by Michael J. Jacey Donald V. Tappan and John J. Wojtowicz NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY REPORT NUMBER 749
The physiological functions of respiration and circulation. Mechanics. exercise 7. Respiratory Volumes. Objectives
 exercise 7 Respiratory System Mechanics Objectives 1. To explain how the respiratory and circulatory systems work together to enable gas exchange among the lungs, blood, and body tissues 2. To define respiration,
exercise 7 Respiratory System Mechanics Objectives 1. To explain how the respiratory and circulatory systems work together to enable gas exchange among the lungs, blood, and body tissues 2. To define respiration,
2. State the volume of air remaining in the lungs after a normal breathing.
 CLASS XI BIOLOGY Breathing And Exchange of Gases 1. Define vital capacity. What is its significance? Answer: Vital Capacity (VC): The maximum volume of air a person can breathe in after a forced expiration.
CLASS XI BIOLOGY Breathing And Exchange of Gases 1. Define vital capacity. What is its significance? Answer: Vital Capacity (VC): The maximum volume of air a person can breathe in after a forced expiration.
NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY
 NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY SUBMARINE BASE, GROTON, CONN. REPORT NO. 1102 EFFECTS OF HYPERBARIC CONDITIONS ON CORNEAL PHYSIOLOGY WITH HYDROGEL CONTACT LENSES by Joseph F. Molinari and James
NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY SUBMARINE BASE, GROTON, CONN. REPORT NO. 1102 EFFECTS OF HYPERBARIC CONDITIONS ON CORNEAL PHYSIOLOGY WITH HYDROGEL CONTACT LENSES by Joseph F. Molinari and James
660 mm Hg (normal, 100 mm Hg, room air) Paco, (arterial Pc02) 36 mm Hg (normal, 40 mm Hg) % saturation 50% (normal, 95%-100%)
 148 PHYSIOLOGY CASES AND PROBLEMS Case 26 Carbon Monoxide Poisoning Herman Neiswander is a 65-year-old retired landscape architect in northern Wisconsin. One cold January morning, he decided to warm his
148 PHYSIOLOGY CASES AND PROBLEMS Case 26 Carbon Monoxide Poisoning Herman Neiswander is a 65-year-old retired landscape architect in northern Wisconsin. One cold January morning, he decided to warm his
Retinal vascular response to breathing increased carbon dioxide and oxygen concentrations. Regina Frayser and John B. Hickam
 Retinal vascular response to breathing increased carbon dioxide and oxygen concentrations Regina Frayser and John B. Hickam The retina has a high rate of oxygen consumption, and the retinal vessels are
Retinal vascular response to breathing increased carbon dioxide and oxygen concentrations Regina Frayser and John B. Hickam The retina has a high rate of oxygen consumption, and the retinal vessels are
Scope: This plan applies to all personnel, including contractors, who enter or work in confined spaces, or supervise such activities.
 11/13/1995 4 5/20/2013 1 of 10 Authority and Scope Regulation: 29 CFR 1910.146 Scope: This plan applies to all personnel, including contractors, who enter or work in confined spaces, or supervise such
11/13/1995 4 5/20/2013 1 of 10 Authority and Scope Regulation: 29 CFR 1910.146 Scope: This plan applies to all personnel, including contractors, who enter or work in confined spaces, or supervise such
Respiratory System Physiology. Dr. Vedat Evren
 Respiratory System Physiology Dr. Vedat Evren Respiration Processes involved in oxygen transport from the atmosphere to the body tissues and the release and transportation of carbon dioxide produced in
Respiratory System Physiology Dr. Vedat Evren Respiration Processes involved in oxygen transport from the atmosphere to the body tissues and the release and transportation of carbon dioxide produced in
HUMAN Sample Experiment High Altitude Simulation (a one variable experiment) (version 5/04/06)
 HUMAN Sample Experiment High Altitude Simulation (a one variable experiment) (version 5/04/06) Students wish to simulate an ascent to the summit of Mount Mckinley (12,500 ft.), perhaps to compare the simulated
HUMAN Sample Experiment High Altitude Simulation (a one variable experiment) (version 5/04/06) Students wish to simulate an ascent to the summit of Mount Mckinley (12,500 ft.), perhaps to compare the simulated
Chapter 17 The Respiratory System: Gas Exchange and Regulation of Breathing
 Chapter 17 The Respiratory System: Gas Exchange and Regulation of Breathing Overview of Pulmonary Circulation o Diffusion of Gases o Exchange of Oxygen and Carbon Dioxide o Transport of Gases in the Blood
Chapter 17 The Respiratory System: Gas Exchange and Regulation of Breathing Overview of Pulmonary Circulation o Diffusion of Gases o Exchange of Oxygen and Carbon Dioxide o Transport of Gases in the Blood
Life Support Team Mission Day Instructions
 Life Support Team Mission Day Instructions Overview The Environmental Control and Life Support System (ECLS) on the space station maintains the astronauts' environment. It makes sure that the atmospheric
Life Support Team Mission Day Instructions Overview The Environmental Control and Life Support System (ECLS) on the space station maintains the astronauts' environment. It makes sure that the atmospheric
Gas Exchange ACTIVITY OVERVIEW SUMMARY KEY CONCEPTS AND PROCESS SKILLS KEY VOCABULARY. Teacher s Guide B-75 L A B O R ATO R Y
 Gas Exchange 17 40- to 2 50-minute sessions ACTIVITY OVERVIEW L A B O R ATO R Y SUMMARY This activity explores the role of the respiratory system in the regulation of gases in the blood. Students investigate
Gas Exchange 17 40- to 2 50-minute sessions ACTIVITY OVERVIEW L A B O R ATO R Y SUMMARY This activity explores the role of the respiratory system in the regulation of gases in the blood. Students investigate
Respiratory Physiology Gaseous Exchange
 Respiratory Physiology Gaseous Exchange Session Objectives. What you will cover Basic anatomy of the lung including airways Breathing movements Lung volumes and capacities Compliance and Resistance in
Respiratory Physiology Gaseous Exchange Session Objectives. What you will cover Basic anatomy of the lung including airways Breathing movements Lung volumes and capacities Compliance and Resistance in
Lung Volumes and Capacities
 Lung Volumes and Capacities Normally the volume of air entering the lungs during a single inspiration is approximately equal to the volume leaving on the subsequent expiration and is called the tidal volume.
Lung Volumes and Capacities Normally the volume of air entering the lungs during a single inspiration is approximately equal to the volume leaving on the subsequent expiration and is called the tidal volume.
Barbara A. Heavers, Ph.D. A. A. Messier, B.S. R. Montanti, HM2 and K. E. Schaefer, M.D. NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY REPORT NUMBER 742
 C0 2 EFFECTS ON ACID-BASE BALANCE IN AIR SATURATION DIVES by Barbara A. Heavers, Ph.D. A. A. Messier, B.S. R. Montanti, HM2 and K. E. Schaefer, M.D. NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY REPORT NUMBER
C0 2 EFFECTS ON ACID-BASE BALANCE IN AIR SATURATION DIVES by Barbara A. Heavers, Ph.D. A. A. Messier, B.S. R. Montanti, HM2 and K. E. Schaefer, M.D. NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY REPORT NUMBER
Leak Checking Large Vacuum Chambers
 Leak Checking Large Vacuum Chambers Technical Overview Vacuum Technologies Introduction Understanding the pump-down characteristics of a large vacuum vessel is critical for determining whether the vacuum
Leak Checking Large Vacuum Chambers Technical Overview Vacuum Technologies Introduction Understanding the pump-down characteristics of a large vacuum vessel is critical for determining whether the vacuum
Challenges in Undersea Medicine
 Challenges in Undersea Medicine CAPT Fred Yeo, MD Commanding Officer Naval Submarine Medical Research Laboratory April 2016 DISTRIBUTION STATEMENT A. Approved for public release. UNCLASSIFIED 1 Panel Members
Challenges in Undersea Medicine CAPT Fred Yeo, MD Commanding Officer Naval Submarine Medical Research Laboratory April 2016 DISTRIBUTION STATEMENT A. Approved for public release. UNCLASSIFIED 1 Panel Members
Temperature Influence on the Flammability Limits of Heat Treating Atmospheres
 Temperature Influence on the Flammability Limits of Heat Treating Atmospheres John Dwyer, Jr., James G. Hansel, Tom Philips Air Products and Chemicals, Inc., Allentown, PA Abstract The flammability limits
Temperature Influence on the Flammability Limits of Heat Treating Atmospheres John Dwyer, Jr., James G. Hansel, Tom Philips Air Products and Chemicals, Inc., Allentown, PA Abstract The flammability limits
Inquiry Investigation: Factors Affecting Photosynthesis
 Inquiry Investigation: Factors Affecting Photosynthesis Background Photosynthesis fuels ecosystems and replenishes the Earth's atmosphere with oxygen. Like all enzyme-driven reactions, the rate of photosynthesis
Inquiry Investigation: Factors Affecting Photosynthesis Background Photosynthesis fuels ecosystems and replenishes the Earth's atmosphere with oxygen. Like all enzyme-driven reactions, the rate of photosynthesis
Specifications...Easy CLIA Classification: Sample Type: Blood gas analyzer operation has never been simpler Sample Size: Measured Parameters PCO2
 medicacorp.com easy inside and out With EasyBloodGas, Medica has redefined blood gas analyzer design The sophistication of traditional blood gas analyzers has been packaged in a new compact format with
medicacorp.com easy inside and out With EasyBloodGas, Medica has redefined blood gas analyzer design The sophistication of traditional blood gas analyzers has been packaged in a new compact format with
^- ; asefehs. a»s.^r,--. - "SSSES. v^yj:-- MEDIGÄL. -^j~ ^ z<_^
 -^«gg, ^- ; Pr v^yj:-- MEDIGÄL "SSSES -^j~ ^ z
-^«gg, ^- ; Pr v^yj:-- MEDIGÄL "SSSES -^j~ ^ z
Alien Gases: Investigating Photosynthesis and Respiration through Inquiry
 Alien Gases: Investigating Photosynthesis and Respiration through Inquiry Overview: Students perform an inquiry based lab investigation using bromothymol blue solution and an aquatic plant. The goal is
Alien Gases: Investigating Photosynthesis and Respiration through Inquiry Overview: Students perform an inquiry based lab investigation using bromothymol blue solution and an aquatic plant. The goal is
Introduction. U.S. Navy Diving Manual (Mar-1970)
 DECOMPRESSION SICKNESS AND OXYGEN TOXICITY IN U.S. NAVY SURFACE-SUPPLIED HE-O2 DIVING Wayne A. Gerth U.S. Navy Experimental Diving Unit 321 Bullfinch Road Panama City, FLORIDA 3247 U.S.A. Introduction
DECOMPRESSION SICKNESS AND OXYGEN TOXICITY IN U.S. NAVY SURFACE-SUPPLIED HE-O2 DIVING Wayne A. Gerth U.S. Navy Experimental Diving Unit 321 Bullfinch Road Panama City, FLORIDA 3247 U.S.A. Introduction
U.S. NAVAL SUBMARINE- MEDICAL CENTER
 U.S. NAVAL SUBMARINE- MEDICAL CENTER Submarine Base, Groton, Conn. MFMORANDUM REPORT NO. 65-14 LIGHTING SURVEY OF THE U.S.S. DOGFISH (SS-350) by S. M. Luria and Donald 0. Weitzman Bureau of Medicine and
U.S. NAVAL SUBMARINE- MEDICAL CENTER Submarine Base, Groton, Conn. MFMORANDUM REPORT NO. 65-14 LIGHTING SURVEY OF THE U.S.S. DOGFISH (SS-350) by S. M. Luria and Donald 0. Weitzman Bureau of Medicine and
OPESAVER Super Irrigation System
 OPESAVER Super Irrigation System Makoto Kishimoto, MD Dokkyo University School of Medicine Kishimoto Eye Clinic Moriyama City Shiga, Japan n ABSTRACT OPESERVER reduces the surge phenomenon during ultrasonic
OPESAVER Super Irrigation System Makoto Kishimoto, MD Dokkyo University School of Medicine Kishimoto Eye Clinic Moriyama City Shiga, Japan n ABSTRACT OPESERVER reduces the surge phenomenon during ultrasonic
Kinetic-Molecular Theory
 GASES Chapter Eleven Kinetic-Molecular Theory! Recall that our only previous description of gases stated that gases completely fill and take the shape of their containers.! The Kinetic-Molecular Theory
GASES Chapter Eleven Kinetic-Molecular Theory! Recall that our only previous description of gases stated that gases completely fill and take the shape of their containers.! The Kinetic-Molecular Theory
EVALUATION OF INTRAVENOUS THERAPY DEVICES IN THE HYPERBARIC CHAMBER
 Navy Experimental Diving Unit NEDU TR 03-21 321 Bullfinch Rd. December 2003 Panama City, FL 32407-7015 EVALUATION OF INTRAVENOUS THERAPY DEVICES IN THE HYPERBARIC CHAMBER Navy Experimental Diving Unit
Navy Experimental Diving Unit NEDU TR 03-21 321 Bullfinch Rd. December 2003 Panama City, FL 32407-7015 EVALUATION OF INTRAVENOUS THERAPY DEVICES IN THE HYPERBARIC CHAMBER Navy Experimental Diving Unit
Physiology Unit 4 RESPIRATORY PHYSIOLOGY
 Physiology Unit 4 RESPIRATORY PHYSIOLOGY In Physiology Today Respiration External respiration ventilation gas exchange Internal respiration cellular respiration gas exchange Respiratory Cycle Inspiration
Physiology Unit 4 RESPIRATORY PHYSIOLOGY In Physiology Today Respiration External respiration ventilation gas exchange Internal respiration cellular respiration gas exchange Respiratory Cycle Inspiration
How and why Eurotrol s CueSee Hypoxic works
 How and why Eurotrol s CueSee Hypoxic works Thank you for taking the time to learn some of the advantages and principles behind Eurotrol s CueSee Hypoxic for the validation of low po2 blood gas measurements.
How and why Eurotrol s CueSee Hypoxic works Thank you for taking the time to learn some of the advantages and principles behind Eurotrol s CueSee Hypoxic for the validation of low po2 blood gas measurements.
TPM TIP. Oil Viscosity
 TPM TIP Oil Viscosity TIP #1 Classification according to viscosity is the most prevalent method of describing oils, and the most common classification systems are those of the SAE, AGMA, and ISO. Each
TPM TIP Oil Viscosity TIP #1 Classification according to viscosity is the most prevalent method of describing oils, and the most common classification systems are those of the SAE, AGMA, and ISO. Each
World of Chemistry Notes for Students [Chapter 13, page 1] Chapter 13 Gases
![World of Chemistry Notes for Students [Chapter 13, page 1] Chapter 13 Gases World of Chemistry Notes for Students [Chapter 13, page 1] Chapter 13 Gases](/thumbs/96/127526823.jpg) World of Chemistry Notes for Students [Chapter 3, page ] Chapter 3 Gases ) Sec 3.8 Kinetic Theory of Gases and the Nature of Gases The Kinetic Theory of Matter says that the tiny particles in all forms
World of Chemistry Notes for Students [Chapter 3, page ] Chapter 3 Gases ) Sec 3.8 Kinetic Theory of Gases and the Nature of Gases The Kinetic Theory of Matter says that the tiny particles in all forms
Power Transformer Maintenance. Maintenance Practices & Procedures for Substation Class Distribution Power Transformers
 Maintenance Practices & Procedures for Substation Class Distribution Power Transformers Why should special maintenance practices be applied to power transformers? Power transformers utilize active cooling
Maintenance Practices & Procedures for Substation Class Distribution Power Transformers Why should special maintenance practices be applied to power transformers? Power transformers utilize active cooling
8. Now plot on the following grid the values of T (K) and V from the table above, and connect the points.
 Charles s Law According to Charles s law, the volume of a fixed mass of gas varies directly with its Kelvin temperature if its pressure is constant. The following table contains Celsius temperature and
Charles s Law According to Charles s law, the volume of a fixed mass of gas varies directly with its Kelvin temperature if its pressure is constant. The following table contains Celsius temperature and
Douglas and Haldane(2) has shown that the oxygen determinations. since it forms the basis of the "Coefficient of Utilisation" (Krrogh) and
 THE MEASUREMENT OF THE OXYGEN CONTENT OF THE MIXED VENOUS BLOOD, AND OF THE VOLUME OF BLOOD CIRCULATING PER MINUTE. BY J. BARCROFT, F. J. W. ROUGHTON AND R. SHOJI. (From the Physiological Laboratory, Cambridge.)
THE MEASUREMENT OF THE OXYGEN CONTENT OF THE MIXED VENOUS BLOOD, AND OF THE VOLUME OF BLOOD CIRCULATING PER MINUTE. BY J. BARCROFT, F. J. W. ROUGHTON AND R. SHOJI. (From the Physiological Laboratory, Cambridge.)
(Received 16 January 1946)
 186 J. Physiol. (I946) I05, I86-I90 6I2.2I5.9 THE ABSORPTION OF FLUIDS FROM THE LUNGS BY F. C. COURTICE AND P. J. PHIPPS From the Experimental Station, Porton and the Laboratory of Physiology, Oxford (Received
186 J. Physiol. (I946) I05, I86-I90 6I2.2I5.9 THE ABSORPTION OF FLUIDS FROM THE LUNGS BY F. C. COURTICE AND P. J. PHIPPS From the Experimental Station, Porton and the Laboratory of Physiology, Oxford (Received
The over-ventilated cat shows a similar adjustment to diminished. being over-ventilated, and he considered that on that account there was
 6I2.235:6I2.26I THE SOURCE OF COa EXPIRED AND THE SITE OF ITS RETENTION. BY LAURENCE IRVING, J. K. W. FERGUSON AND F. B. PLEWES. (From the Department of Physiology, University of Toronto.) AFTER evisceration
6I2.235:6I2.26I THE SOURCE OF COa EXPIRED AND THE SITE OF ITS RETENTION. BY LAURENCE IRVING, J. K. W. FERGUSON AND F. B. PLEWES. (From the Department of Physiology, University of Toronto.) AFTER evisceration
You should be able to: Describe Equipment Barometer Manometer. 5.1 Pressure Read and outline 5.1 Define Barometer
 A P CHEMISTRY - Unit 5: Gases Unit 5: Gases Gases are distinguished from other forms of matter, not only by their power of indefinite expansion so as to fill any vessel, however large, and by the great
A P CHEMISTRY - Unit 5: Gases Unit 5: Gases Gases are distinguished from other forms of matter, not only by their power of indefinite expansion so as to fill any vessel, however large, and by the great
I Physical Principles of Gas Exchange
 Respiratory Gases Exchange Dr Badri Paudel, M.D. 2 I Physical Principles of Gas Exchange 3 Partial pressure The pressure exerted by each type of gas in a mixture Diffusion of gases through liquids Concentration
Respiratory Gases Exchange Dr Badri Paudel, M.D. 2 I Physical Principles of Gas Exchange 3 Partial pressure The pressure exerted by each type of gas in a mixture Diffusion of gases through liquids Concentration
TOXIC GAS DETECTORS IN THE WORKPLACE UK REGULATIONS AND STANDARDS
 SUMMARY TOXIC GAS DETECTORS IN THE WORKPLACE UK REGULATIONS AND STANDARDS In recent articles supplied by the Council of Gas Detection and Environmental Monitoring (CoGDEM) for inclusion in International
SUMMARY TOXIC GAS DETECTORS IN THE WORKPLACE UK REGULATIONS AND STANDARDS In recent articles supplied by the Council of Gas Detection and Environmental Monitoring (CoGDEM) for inclusion in International
PICU Resident Self-Study Tutorial The Basic Physics of Oxygen Transport. I was told that there would be no math!
 Physiology of Oxygen Transport PICU Resident Self-Study Tutorial I was told that there would be no math! INTRODUCTION Christopher Carroll, MD Although cells rely on oxygen for aerobic metabolism and viability,
Physiology of Oxygen Transport PICU Resident Self-Study Tutorial I was told that there would be no math! INTRODUCTION Christopher Carroll, MD Although cells rely on oxygen for aerobic metabolism and viability,
Gases and Respiration. Respiration Overview I
 Respiration Overview I Respiration Overview II Gas Laws Equation of State: PV = nrt Same volumes of different gases have same # of molecules BTPS: body temp, atmospheric pressure, saturated ATPS: ambient
Respiration Overview I Respiration Overview II Gas Laws Equation of State: PV = nrt Same volumes of different gases have same # of molecules BTPS: body temp, atmospheric pressure, saturated ATPS: ambient
PURE SUBSTANCE. Nitrogen and gaseous air are pure substances.
 CLASS Third Units PURE SUBSTANCE Pure substance: A substance that has a fixed chemical composition throughout. Air is a mixture of several gases, but it is considered to be a pure substance. Nitrogen and
CLASS Third Units PURE SUBSTANCE Pure substance: A substance that has a fixed chemical composition throughout. Air is a mixture of several gases, but it is considered to be a pure substance. Nitrogen and
Levels of CO2 in Arterial Blood of Carp under Carbon Dioxide Anesthesia
 J. Nutr. Sci. Vitaminol., 28, 35-39, 1982 Levels of CO2 in Arterial Blood of Carp under Carbon Dioxide Anesthesia Hisateru MITSUDA, Saburo UENO, Hiroshi MIZUNO, Tadashi UEDA, Hiromi FUJIKAWA, Tomoko NOHARA,
J. Nutr. Sci. Vitaminol., 28, 35-39, 1982 Levels of CO2 in Arterial Blood of Carp under Carbon Dioxide Anesthesia Hisateru MITSUDA, Saburo UENO, Hiroshi MIZUNO, Tadashi UEDA, Hiromi FUJIKAWA, Tomoko NOHARA,
Gas Laws. Directions: Describe what contribution each of the Scientist below made to the Gas Laws and include there gas law equation.
 Gas Laws Name Date Block Introduction One of the most amazing things about gases is that, despite wide differences in chemical properties, all the gases more or less obey the gas laws. The gas laws deal
Gas Laws Name Date Block Introduction One of the most amazing things about gases is that, despite wide differences in chemical properties, all the gases more or less obey the gas laws. The gas laws deal
Physical Chemistry of Gases: Gas Exchange Linda Costanzo, Ph.D.
 Physical Chemistry of Gases: Gas Exchange Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. Application of the gas laws to pulmonary physiology. 2. How to
Physical Chemistry of Gases: Gas Exchange Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. Application of the gas laws to pulmonary physiology. 2. How to
Respiratory Protection
 Respiratory Protection Purpose The purpose is to establish a set of guidelines for the selection of respiratory protection equipment, situations for its use, and training in the use of the respirator required.
Respiratory Protection Purpose The purpose is to establish a set of guidelines for the selection of respiratory protection equipment, situations for its use, and training in the use of the respirator required.
Respiratory System Review
 KEY THIS TEST WILL BE COMPLETED IN ONE CLASS PERIOD MONDAY, MARCH 10. 2014 Respiratory System Review Name A. Directions: Fill in the blank with the appropriate vocabulary word or words (several examples
KEY THIS TEST WILL BE COMPLETED IN ONE CLASS PERIOD MONDAY, MARCH 10. 2014 Respiratory System Review Name A. Directions: Fill in the blank with the appropriate vocabulary word or words (several examples
PLANT OPERATIONS AND TECHNOLOGY I & II: REAL TIME FURNACE AIR IN- LEAKAGE MONITORING ON COAL, OIL AND/OR GAS FIRED BOILERS
 Electric Power Conference 2010 May 19th, 2010, Baltimore, MD PLANT OPERATIONS AND TECHNOLOGY I & II: REAL TIME FURNACE AIR IN- LEAKAGE MONITORING ON COAL, OIL AND/OR GAS FIRED BOILERS Stephen Hall Senior
Electric Power Conference 2010 May 19th, 2010, Baltimore, MD PLANT OPERATIONS AND TECHNOLOGY I & II: REAL TIME FURNACE AIR IN- LEAKAGE MONITORING ON COAL, OIL AND/OR GAS FIRED BOILERS Stephen Hall Senior
THE literature on this subject, which was reviewed recently (CAMPBELL, doses of amytal, and in addition received A.C.E. mixture during the
 -~~ -v GAS TENSIONS IN THE MUCOUS MEMBRANE OF THE STOMACH AND SMALL INTESTINE. By J. ARGYLL CAMPBELL. From the National Institute for Medical Research, Hampstead. (With six figures in the text.) (Received
-~~ -v GAS TENSIONS IN THE MUCOUS MEMBRANE OF THE STOMACH AND SMALL INTESTINE. By J. ARGYLL CAMPBELL. From the National Institute for Medical Research, Hampstead. (With six figures in the text.) (Received
NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY
 NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY SUBMARINE BASE, GROTON, CONN. REPORT NUMBER 1029 LEAKAGE INTO THE NAVY OXYGEN BREATHING APPARATUS WHEN WORN OVER SPECTACLES " ' by S. M. Luria " and J. H, Dougherty,
NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY SUBMARINE BASE, GROTON, CONN. REPORT NUMBER 1029 LEAKAGE INTO THE NAVY OXYGEN BREATHING APPARATUS WHEN WORN OVER SPECTACLES " ' by S. M. Luria " and J. H, Dougherty,
1. Scope: This document describes the procedure for verifying the performance of vaccine vial monitors.
 PQS Independent type-testing protocol WHO/PQS/E06/IN05.VP.2 Original: English Distribution: General TITLE: Vaccine Vial Monitor Product verification protocol: E06/IN05.VP.2 Applies to specification ref(s):
PQS Independent type-testing protocol WHO/PQS/E06/IN05.VP.2 Original: English Distribution: General TITLE: Vaccine Vial Monitor Product verification protocol: E06/IN05.VP.2 Applies to specification ref(s):
13.1!"#$#%"&'%()$*+%,+-.$+/*$#
 343%%%%%%%%%5)"./$+%67%%%%%!"#$# 13.1!"#$#%"&'%()$*+%,+-.$+/*$#!"#$%&'($)*!"#$%&'($)+ If you want to understand how gases behave such as why fresh air rushes into your lungs when certain chest muscles
343%%%%%%%%%5)"./$+%67%%%%%!"#$# 13.1!"#$#%"&'%()$*+%,+-.$+/*$#!"#$%&'($)*!"#$%&'($)+ If you want to understand how gases behave such as why fresh air rushes into your lungs when certain chest muscles
6A.004 SCBA MAINTENANCE. Policy Number. Page 1 of 5 SCOPE
 SCBA MAINTENANCE SCOPE Stoney Point Fire Department Self-contained breathing apparatus provides respiratory protection under conditions of oxygen deficiency or in concentrations of toxic gases immediately
SCBA MAINTENANCE SCOPE Stoney Point Fire Department Self-contained breathing apparatus provides respiratory protection under conditions of oxygen deficiency or in concentrations of toxic gases immediately
Diving at Altitude. Part One: Starting with Residual Nitrogen. By John Adsit January 30, 2018
 Diving at Altitude By John Adsit January 30, 2018 Diving at altitude demands adjustments to the procedures used at sea level. With moderate increases in altitude and with shallower dives, the differences
Diving at Altitude By John Adsit January 30, 2018 Diving at altitude demands adjustments to the procedures used at sea level. With moderate increases in altitude and with shallower dives, the differences
6I2.2I6:6I alveolar pressure. It follows that the evident alteration in the respiratory rhythm is an alteration in amplitude.
 6I2.2I6:6I2.223.11 SOME EFFECTS OF CARBONIC ACID ON THE CHARACTER OF HUMAN RESPIRATION. BY J. BARCROFT AND R. MARGARIA' (Turin). (From the Physiological Laboratory, Cambridge.) THE following facts concerning
6I2.2I6:6I2.223.11 SOME EFFECTS OF CARBONIC ACID ON THE CHARACTER OF HUMAN RESPIRATION. BY J. BARCROFT AND R. MARGARIA' (Turin). (From the Physiological Laboratory, Cambridge.) THE following facts concerning
Respiratory Physiology. Adeyomoye O.I
 Respiratory Physiology By Adeyomoye O.I Outline Introduction Hypoxia Dyspnea Control of breathing Ventilation/perfusion ratios Respiratory/barometric changes in exercise Intra-pulmonary & intra-pleural
Respiratory Physiology By Adeyomoye O.I Outline Introduction Hypoxia Dyspnea Control of breathing Ventilation/perfusion ratios Respiratory/barometric changes in exercise Intra-pulmonary & intra-pleural
analyses, it seemed that the pressure of alveolar carbon dioxide was less
 VARIATIONS IN ALVEOLAR CARBON DIOXIDE PRESSURE IN RELATION TO MEALS1. By (From the Bland-Sutton Institute qf Pathology, Middlesex Hospital.) THE investigation described below arose in the following manner.
VARIATIONS IN ALVEOLAR CARBON DIOXIDE PRESSURE IN RELATION TO MEALS1. By (From the Bland-Sutton Institute qf Pathology, Middlesex Hospital.) THE investigation described below arose in the following manner.
Experiment B-3 Respiration
 1 Experiment B-3 Respiration Objectives To study the diffusion process of oxygen and carbon dioxide between the alveoli and pulmonary capillaries. To determine the percentage of oxygen in exhaled air while
1 Experiment B-3 Respiration Objectives To study the diffusion process of oxygen and carbon dioxide between the alveoli and pulmonary capillaries. To determine the percentage of oxygen in exhaled air while
VOLUNTARY BREATHHOLDING. I. PULMONARY GAS
 VOLUNTARY BREATHHOLDING. I. PULMONARY GAS EXCHANGE DURING BREATHHOLDING'1 By CHARLES D. STEVENS, EUGENE B. FERRIS, JOSEPH P. WEBB, GEORGE L. ENGEL, AND MYRTLE LOGAN (From the Departments of Internal Medicine
VOLUNTARY BREATHHOLDING. I. PULMONARY GAS EXCHANGE DURING BREATHHOLDING'1 By CHARLES D. STEVENS, EUGENE B. FERRIS, JOSEPH P. WEBB, GEORGE L. ENGEL, AND MYRTLE LOGAN (From the Departments of Internal Medicine
Pressure and Flow Characteristics of Ocular Viscoelastic and Silicone Oil Through Glaucoma Drainage Devices
 Pressure and Flow Characteristics of Ocular Viscoelastic and Silicone Oil Through Glaucoma Drainage Devices Reid Mollman, MD Table Rock Regional Roundup 9/29/2017 Disclosures None of the participants in
Pressure and Flow Characteristics of Ocular Viscoelastic and Silicone Oil Through Glaucoma Drainage Devices Reid Mollman, MD Table Rock Regional Roundup 9/29/2017 Disclosures None of the participants in
Water Quality Guidelines for Total Gas Pressure: First Update
 Water Quality Water Quality Guidelines for Total Gas Pressure: First Update Overview Report Prepared pursuant to Section 2(e) of the Environment Management Act, 1981 Approved: Date Assistant Deputy Minister
Water Quality Water Quality Guidelines for Total Gas Pressure: First Update Overview Report Prepared pursuant to Section 2(e) of the Environment Management Act, 1981 Approved: Date Assistant Deputy Minister
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Lung test gas, CO/C 2 H 2 /CH 4 AGA 0.3%, 0.3%, 0.3% medicinal gas, compressed 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Carbon monoxide
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Lung test gas, CO/C 2 H 2 /CH 4 AGA 0.3%, 0.3%, 0.3% medicinal gas, compressed 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Carbon monoxide
ANSI / ASSE Z88.2 American National Standard Practices for Respiratory Protection (1992 rev. versus 2015 rev.)
 ANSI / ASSE Z88.2 BACKGROUND: A substantial body of research has been published since the 1992 revision of Z88.2. This information related to the proper use and performance of respiratory protection in
ANSI / ASSE Z88.2 BACKGROUND: A substantial body of research has been published since the 1992 revision of Z88.2. This information related to the proper use and performance of respiratory protection in
Subj: HYPERBARIC OXYGEN TREATMENT IN NAVY RECOMPRESSION CHAMBERS
 DEPARTMENT OF THE NAVY BUREAU OF MEDICINE AND SURGERY 7700 ARLINGTON BOULEVARD FALLS CHURCH, VA 22042 IN REPLY REFER TO BUMEDINST 6320.38C BUMED-M95 BUMED INSTRUCTION 6320.38C From: Chief, Bureau of Medicine
DEPARTMENT OF THE NAVY BUREAU OF MEDICINE AND SURGERY 7700 ARLINGTON BOULEVARD FALLS CHURCH, VA 22042 IN REPLY REFER TO BUMEDINST 6320.38C BUMED-M95 BUMED INSTRUCTION 6320.38C From: Chief, Bureau of Medicine
Annex 9 Processes Quality Control. Introduction
 Annex 9 Processes Quality Control Introduction The eradication of screwworms depends on the release and dispersal into the environment of sterile screwworm flies in optimum biological condition providing
Annex 9 Processes Quality Control Introduction The eradication of screwworms depends on the release and dispersal into the environment of sterile screwworm flies in optimum biological condition providing
The Following information is based on material prepared by the American Lung Association
 The Following information is based on material prepared by the American Lung Association RESPIRATORY PROTECTION FROM INHALATION HAZARDS The inhalation of harmful dusts, toxic gases and vapors at fire and
The Following information is based on material prepared by the American Lung Association RESPIRATORY PROTECTION FROM INHALATION HAZARDS The inhalation of harmful dusts, toxic gases and vapors at fire and
Respiration - Human 1
 Respiration - Human 1 At the end of the lectures on respiration you should be able to, 1. Describe events in the respiratory processes 2. Discuss the mechanism of lung ventilation in human 3. Discuss the
Respiration - Human 1 At the end of the lectures on respiration you should be able to, 1. Describe events in the respiratory processes 2. Discuss the mechanism of lung ventilation in human 3. Discuss the
ENVIRONMENTAL AND POLLUTANTS GAS ANALYZERS
 XIX IMEKO World Congress Fundamental and Applied Metrology September 6, 9, Lisbon, Portugal ENVIRONMENTAL AND POLLUTANTS GAS ANALYZERS Ana Madeira, Florbela A. Dias and Eduarda Filipe 3 Instituto Português
XIX IMEKO World Congress Fundamental and Applied Metrology September 6, 9, Lisbon, Portugal ENVIRONMENTAL AND POLLUTANTS GAS ANALYZERS Ana Madeira, Florbela A. Dias and Eduarda Filipe 3 Instituto Português
Humidity Therapy. Terms to know:
 RC-170 Humidity Therapy Terms to know: Humidity: Water in a vapor state of matter. Absolute Humidity (mg/liter) actual amount of water in a vapor form. Relative Humidity (% up to 100) absolute compared
RC-170 Humidity Therapy Terms to know: Humidity: Water in a vapor state of matter. Absolute Humidity (mg/liter) actual amount of water in a vapor form. Relative Humidity (% up to 100) absolute compared
Gases and Pressure. Main Ideas
 Gases and Pressure Key Terms pressure millimeters of mercury partial pressure newton atmosphere of pressure Dalton s law of partial pressures barometer pascal In the chapter States of Matter, you read
Gases and Pressure Key Terms pressure millimeters of mercury partial pressure newton atmosphere of pressure Dalton s law of partial pressures barometer pascal In the chapter States of Matter, you read
BP OIL -- TOLEDO REFINERY
 Document Type: Procedure BP OIL -- TOLEDO REFINERY Refinery Wide Procedure No.: SAF 086 Effective Date: May 9, 2016 Use of Nitrogen (N 2 ) Rev. No.: 9 Owner: Emily Stewart Auth. By: Chris Conley (Signature
Document Type: Procedure BP OIL -- TOLEDO REFINERY Refinery Wide Procedure No.: SAF 086 Effective Date: May 9, 2016 Use of Nitrogen (N 2 ) Rev. No.: 9 Owner: Emily Stewart Auth. By: Chris Conley (Signature
Submarine Base, Groton, Conn. REPORT NUMBER 690. RESPONSE VARIATIONS OF THREE TYPES OF MICROPHONES PRESSURIZED TO 19 Ata
 'd-jl^ c^/^ * Submarine Base, Groton, Conn. REPORT NUMBER 690 RESPONSE VARIATIONS OF THREE TYPES OF MICROPHONES PRESSURIZED TO 19 Ata by Thomas Murry and Russell L. Sergeant Bureau of Medicine and Surgery,
'd-jl^ c^/^ * Submarine Base, Groton, Conn. REPORT NUMBER 690 RESPONSE VARIATIONS OF THREE TYPES OF MICROPHONES PRESSURIZED TO 19 Ata by Thomas Murry and Russell L. Sergeant Bureau of Medicine and Surgery,
Let s talk about Capnography
 Let s talk about Capnography This is one of a series of articles by Keith Simpson BVSc MRCVS MIET (Electronics) discussing the practical aspects of some common monitoring techniques. Capnometry is the
Let s talk about Capnography This is one of a series of articles by Keith Simpson BVSc MRCVS MIET (Electronics) discussing the practical aspects of some common monitoring techniques. Capnometry is the
CHAPTER 9 $LU'HFRPSUHVVLRQ
 CHAPTER 9 $LU'HFRPSUHVVLRQ 9-1 INTRODUCTION 9-1.1 Purpose. This chapter discusses decompression requirements for air diving operations. 9-1.2 Scope. This chapter discusses five different tables, each with
CHAPTER 9 $LU'HFRPSUHVVLRQ 9-1 INTRODUCTION 9-1.1 Purpose. This chapter discusses decompression requirements for air diving operations. 9-1.2 Scope. This chapter discusses five different tables, each with
Chapter 1: Respiration
 Chapter 1: Respiration Respiration Human Breathing Mechanism Transport of oxygen Importance of a healthy respiratory system Respiratory system lungs inhalation exhalation Diffusion of oxygen by blood Transport
Chapter 1: Respiration Respiration Human Breathing Mechanism Transport of oxygen Importance of a healthy respiratory system Respiratory system lungs inhalation exhalation Diffusion of oxygen by blood Transport
29th Monitoring Research Review: Ground-Based Nuclear Explosion Monitoring Technologies
 IMPROVED CALIBRATION AND OPERATION OF A STABLE-GAS QUANTIFICATION MANIFOLD AS PART OF A RADIOXENON COLLECTION SYSTEM ABSTRACT Richard M. Williams, James C. Hayes, and Randy R. Kirkham Pacific Northwest
IMPROVED CALIBRATION AND OPERATION OF A STABLE-GAS QUANTIFICATION MANIFOLD AS PART OF A RADIOXENON COLLECTION SYSTEM ABSTRACT Richard M. Williams, James C. Hayes, and Randy R. Kirkham Pacific Northwest
B14 gas exchange note, reading and questions.notebook April 13, 2011
 Review All organisms (plants and animals) need energy to carry out their life processes. The chemi reaction which provides this energy is called. In larger animals it is sometimes called respiration because
Review All organisms (plants and animals) need energy to carry out their life processes. The chemi reaction which provides this energy is called. In larger animals it is sometimes called respiration because
Respiration. The resspiratory system
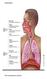 Respiration The resspiratory system The Alveoli The lungs have about 300 million alveoli, with a total crosssec onal area of 50 70 m2.. Each alveolar sac is surrounded by blood capillaries. The walls of
Respiration The resspiratory system The Alveoli The lungs have about 300 million alveoli, with a total crosssec onal area of 50 70 m2.. Each alveolar sac is surrounded by blood capillaries. The walls of
partial pressure is to be applied to the dissociation curve of fully oxygenated
 6I2. I27. I THE DETERMINATION OF THE CARBON DIOXIDE CONTENT OF THE MIXED VENOUS BLOOD. Part I. The effect of oxygenation and the critical oxygen tension. BY M. C. G. ISRAELS (Platt Physiological Scholar)
6I2. I27. I THE DETERMINATION OF THE CARBON DIOXIDE CONTENT OF THE MIXED VENOUS BLOOD. Part I. The effect of oxygenation and the critical oxygen tension. BY M. C. G. ISRAELS (Platt Physiological Scholar)
A. What are the three states of matter chemists work with?
 Chapter 10 and 12 The Behavior of Gases Chapter 10 The States of Matter A. What are the three states of matter chemists work with? Section 10.1 Pg 267 B. We will explain the behavior of gases using the
Chapter 10 and 12 The Behavior of Gases Chapter 10 The States of Matter A. What are the three states of matter chemists work with? Section 10.1 Pg 267 B. We will explain the behavior of gases using the
breathing oxygen-rich gas mixtures" was made in 1911 by Benedict the London Hospital.
 THE; EFFECT OF INHALATION OF OXYGEN ON THE RATE OF THE PULSE IN HEALTH. BY JOHN PARKINSON, M.D., M.R.C.P., Medical Registrar to the London Hospital. NONE of the early workers on the effect of oxygen on
THE; EFFECT OF INHALATION OF OXYGEN ON THE RATE OF THE PULSE IN HEALTH. BY JOHN PARKINSON, M.D., M.R.C.P., Medical Registrar to the London Hospital. NONE of the early workers on the effect of oxygen on
(a) (i) Describe how a large difference in oxygen concentration is maintained between a fish gill and the surrounding water.
 1. Answers should be written in continuous prose. Credit will be given for biological accuracy, the organisation and presentation of information and the way in which an answer is expressed. Fick s law
1. Answers should be written in continuous prose. Credit will be given for biological accuracy, the organisation and presentation of information and the way in which an answer is expressed. Fick s law
