Barbara A. Heavers, Ph.D. A. A. Messier, B.S. R. Montanti, HM2 and K. E. Schaefer, M.D. NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY REPORT NUMBER 742
|
|
|
- Arnold Joseph
- 5 years ago
- Views:
Transcription
1 C0 2 EFFECTS ON ACID-BASE BALANCE IN AIR SATURATION DIVES by Barbara A. Heavers, Ph.D. A. A. Messier, B.S. R. Montanti, HM2 and K. E. Schaefer, M.D. NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY REPORT NUMBER 742 Bureau of Medicine and Surgery, Navy Department Research Work Unit MR BOKL.09 Transmitted by: K. E. Schaefer, M.D. Head, Environmental Physiology Branch NAVSUBMEDRSCHLAB Reviewed and Approved by: Approved and Released by Charles F. Gell, M.D., D.Sc.(Med) SCIENTIFIC DIRECTOR NavSubMedRschLab J. H. Baker, CAPT MC USN OFFICER IN CHARGE NavSubMedRschLab Approved for public release; distribution unlimited.
2 SUMMARY PAGE THE PROBLEM To evaluate the effects of low levels of ambient CO2, frequently found in diving operations due to inadequate CO2 scrubbing, on acid-base parameters in air saturation dives to depth equivalents of 50 and 60 feet. FINDINGS In two saturation dives, rats of the Sprague-Dawley strain were exposed to increased air pressures equivalent to 50 and 60 feet of sea water for varying periods of time up to 60 days. The average ambient CO2 concentrations (surface equivalent) were 0.41% CO2 during the 60 day-50 foot saturation dive and 0.33% CO2 during the 35 day-60 foot saturation dive. In both experiments, consistent increases in arterial CO2 tensions to mm Hg, and decreases in ph were found. Plasma bicarbonate was elevated 5-7 meq and plasma chloride correspondingly decreased. These findings indicate the existence of a pronounced respiratory acidosis which was thought to be caused by the low levels of ambient C0 2 in conjunction with increased oxygen and nitrogen levels. Neither the elevated nitrogen tension or oxygen tension alone could have produced such changes in acid-base parameters. APPLICATION A systematic study of the effects of increased ambient CO2 levels, frequently found in diving operations, on acid-base parameters during saturation dives on air had, to our knowledge, not been made. The findings indicate an approximate 10-fold increase in the effectiveness of ambient CO2 under increased air pressures on acid-base parameters as compared to ambient CO2 effects under normal atmospheric conditions. This could contribute significantly to accidents in diving operations. It is recommended that greater attention be given to elimination of ambient CO2 by installing efficient CO2 scrubbing systems in diving operations. ADMINISTRATIVE INFORMATION This investigation was conducted as part of Bureau of Medicine and Surgery Research Work Unit MR BOKL. The present report is Number 9 on this work unit. It was submitted for review on 6 March 1973, approved for publication on 11 April 1973 and designated as NavSubMedRschLab Report No PUBLISHED BY THE NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY 11
3 ABSTRACT Blood gas tensions, ph, plasma chloride, sodium and potassium, blood urea, ammonia and amino acid nitrogen were measured in mature Sprague-Dawley rats at various intervals during and after exposure to pressures equivalent to depth at 50 feet and 60 feet, lasting for 60 and 35 days, respectively. In both experiments, a consistent decrease in ph to values of about 7.30 and a rise in COg tensions to mm Hg in the arterial blood were found. Plasma bicarbonate was elevated 5-7 meq and chloride correspondingly decreased. Plasma potassium was consistently increased in both experiments. These findings indicate the existence of a pronounced respiratory acidosis during the saturation dives to 50 and 60 feet. There is no evidence in the literature which would indicate that, elevated pressures of nitrogen or oxygen, per se; in the ranges observed in these experiments could cause significant acid-base alterations. It must, therefore, be concluded that the increased ambient CC"2 levels in conjunction with the effects of increased oxygen and nitrogen is responsible for the observed changes. When compared with data obtained in rats during acute and chronic hypercapnia, the observed PaCC>2, ph, and bicarbonate values correspond to the range of values obtained by exposure to 3% and 5% CO2. iii
4
5 C0 2 EFFECTS ON ACID-BASE BALANCE IN AIR SATURATION DIVES INTRODUCTION There is a lack of knowledge concerning the effects of increased levels of ambient C0 2 at increased pressures during short and chronic exposures to different gas mixtures. In previous studies involving saturation dives by men to pressures equivalent to 200, 800 and 1,000 feet of seawater, differences in ambient CO2 resulted in marked differences in the response characteristics of respiration, acid-base balance and electrolyte excretion '. In the first experiment, in which three subjects were exposed for 12 days to a Helium- Oxygen-Nitrogen atmosphere at 200 feet, the average ambient CO2 level amounted to 1.17% sea level equivalent. Alveolar carbon dioxide tension measured under these conditions corresponded with alveolar C0 2 tensions measured during inhalation of 5.5% CO2 at normal atmospheric pressure. Electrolyte excretion in the urine showed patterns similar to those observed during exposure of dogs and man to 3% C0 2. None of these findings were observed during saturation excursion dives to much greater depths at 800 and 1000 feet, breathing helium-oxygen under conditions, in which ambient C0 2 was below measurable levels. The alveolar C0 2 tensions were found to be normal and the pattern of urine electrolyte excretion did not show any evidence of C0 2 retention. These findings indicated that increased pressure in a helium-oxygennitrogen atmosphere caused an amplifi- cation of ambient C0 2 effects by a factor of 5. The present report deals with a study of acid-base characteristics during air saturation dives at 50 and 60 feet in rats. Due to external circumstances, the life-support system used in the chamber during the dives could not reduce the ambient C0 2 levels below % C0 2 (surface equivalent). The results indicate that the increased air-pressure produced an amplification of ambient C0 2 effects by a factor of 10 as compared with the effects of C0 2 exposure at normal atmospheric conditions. METHODS AND MATERIALS In two separate dives, male albino rats of the Sprague-Dawley strain, weighing 250 to 450 grams were exposed to hyperbaric conditions equivalent to 50 and 60 ft. of seawater, respectively, for varying periods of time up to 60 days. At the 50 ft. depth the chamber was maintained at 51.06% 0 2 and 0.42% C0 2 (surface equivalent), and a temperature of 79.98F. At the 60 ft. depth the chamber was maintained at 56.8% 0 2 and 0.33% C0 2 (surface equivalent), a temperature of 82.6Fand a relative humidity of 73.9%. The following blood sampling procedure was utilized by trained divers at both depths. Groups of five animals were anesthesized with sodium pentobarbital (0.1 cc/l00 g). The abdominal aorta was exposed and approximately 10 cc of blood drawn with a heparinized glass
6 syringe. The anaerobic blood samples were cooled in ice and safely decompressed at a rate of 8 ft/min. In the 60-ft. dive a group of five animals at the surface were used as controls, at each time exposure, using the same sampling procedure. A 6-7 ml aliquot of each sample was drawn off for plasma and red cell determination. The remaining anaerobic whole blood was used immediately for blood gas analysis and bicarbonate (HCO3) determinations. The Pa02, PaCC»2 and ph were determined for each sample using the Instrumentation Laboratory (IL) ph/gas Analyzer Model 113. The Pa02 was adjusted by a temperature correction factor of 1.02 for this instrument. The actual HCO3 was calculated from an IL nomogram based on ph and PaC02» The standard HCO3 was determined by equilibrating each blood sample with high C0 2 (7.94%) and low C0 2 (4.64%), using the Astrup Microtonometer AMT 1. Equilibration time was 3 minutes and each sample was measured twice. Actual and standard HCO3 concentrations are expressed in mequivalents/l blood. Hematocrits were determined by drawing each sample into a 100 yx heparinized capillary tube which was then spun for 10 min. at 10,000 rpm and read on an International Circular Microcapillary reader. Plasma was obtained by centrifuging 5-6 cc whole blood at 2000 rpm for 10 min. The plasma was drawn off and refrigerated immediately. Plasma ammonia was measured within 2 hours of sampling with an Orion Ammonia Gas Sensing Electrode Model The electrode was adapted for measurement of plasma volumes of less than 0.2 cc by addition of a special flowthru cap obtained from Orion Research Incorporated and a Technicon Auto Analyzer Proportioning Pump Model 1. Plasma sodium (Na) and potassium (K) were measured with an IL Flamephotometer Model 343 and plasma chloride (Cl~) with a Buchler-Cotlove Chloridometer. Plasma urea was measured using a Hyland Laboratories UN Test Kit. Plasma amino acid nitrogen was measured according to Russell (1944) 7. RESULTS The 50 ft. dive data on chamber ambient CO2, whole blood ph, PaC02, HCO3 and Cl~ are shown in Figure 1. The CO2 scrubbing system limitations resulted in a slightly elevated, mean ambient CO2 of 3.1 mm Hg through the 60-day period. The whole blood ph is acidotic during the first three days, shifts toward alkalotic at two weeks and then returns an acidotic state during the remainder of the experiment. Animals safely decompressed continue to show a slight acidosis after 5 days at the surface. The early phase of acidosis is accompanied by a rapid rise in PaC02 and HCOg and a sharp decline in plasma Cl". The PaC0 2 and HCO3 slowly recover to normal at two weeks and then
7 60 DAYS AT 50 FT DEPTH IN AIR- C0 Z AMBIENT ucmn «-SMFACE-H IN All «OWTROL i CONTROL DAYS EXPOSURE SDoyt Fig. 1. Ambient CÖ2 levels, acid-base parameters (ph, PaCÖ2, and HCO 3) and plasma chloride during 60 days of exposure to air pressure equivalent to 50 feet of seawater and during 5 days of subsequent recovery at atmospheric pressure. o control; statistically significant difference from controls at the 5% level and better
8 rise again after three weeks. The Cl, on the other hand, remains low after the initial drop and returns to normal only after five days at the surface. The plasma Na and K values are shown in Table 1. There is a dramatic increase in K levels during the 50 ft. exposure, but only intermittent changes in the Na levels. The plasma urea and amino-acid nitrogen (N) values are shown in Table 2. Plasma urea values remain nearly normal, while amino acid N shows periodic fluctuations during the exposure. The 60-ft. dive data on chamber ambient CO2, whole blood ph, PaO and PaCC>2 are shown in Figure 2. In this dive, improvement of the CO2 scrubbing system resulting in a mean ambient CO2 of 2.5 mm Hg throughout the 35- day period. Again the whole blood ph is acidotic during the first week of exposure, then gradually returns to normal within four weeks. There is a marked alkalosis on the first day of recovery following safe decompression and a return to normal by the sixth day. Pa0 2 is significantly elevated during the entire exposure, but rapidly returns to normal following safe decompression. As in the 50 ft. dive the PaC0 2 is elevated during the entire dive. There is a significant decline in PaCC>2 during the first day after safe decompression followed by a slow return to normal by the eighth day. Figure 3 shows the changes in plasma Cl~, actual HCO3, standard HCOg and hematocrit for the 60 foot dive. As before, plasma Cl~ values exhibit a sharp decline on the first day and a significantly low Cl~ level persists throughout the dive. The recovery animals exhibit a return to normal Cl~ levels on the first day after decompression followed by a significant increase through the sixth day and a return to normal by the eighth day. Actual HCOg values again show a dramatic rise on the first day followed by a slow decline during the first week. There is a return to normal immediately following decompression. The standard HCO3 shows a similar rapid rise in the first week of exposure and a rapid return to normal after decompression. There is a very little change in hematocrit levels. A brief decline occurs during the first and second days following decompression. The changes in plasma ammonia levels are shown in Figure 4. There is a sharp decrease during the first week of exposure, a return to near normal after ten days and then another rapid decline after four weeks. Following decompression there is a general return to normal, with some fluctuation at six days and two weeks. The Na and K plasma levels are shown in Table 3. K shows a dramatic increase during the first five days and a return to normal by one week, while Na levels remain normal throughout the duration. The plasma urea and amino acid N remain nearly normal as seen in Table 4.
9 TABLE 1 60 DAYS AT 50 FT. DEPTH IN AIR DAY Na (meq./l plasma) K (meq. /L plasma) mean S.E.M. "t" mean S.E.M. "t" CONTROL * * * * * * * * * 4.15 OS 5.32* * * * RECOVERY 3 hrs * 5 days *Statistically significant from controls at the 5% level.
10 TABLE 2 60 DAYS AT 50 FT. DEPTH IN AIR DAY AMINOACID N (mg/dlplasma) UREA (mg/dl plasma) mean S.E.M. "t" mean S.E.M. "t" CONTROL _ * * * * * * RECOVERY 3hrs days *Statistieally significant from controls at the 5% level.
11 -35 DAYS AT 60 FT DEPTH IN AIR- C0 Z AMBIENT RECOVERY SURFACE - IN AIR DAYS EXPOSURE IO DAYS Fig. 2. Ambient CÖ2 levels and acid-base parameters (ph, PäÖ2 and PaC02) during 35 days of exposure to air pressure equivalent to 60 feet of seawater, and 2 weeks of subsequent recovery at atmospheric pressure. o control; statistically significant difference from controls at the 5% level and better
12 35 DAYS AT 60FT DEPTH IN AIR- CIS RECOVERY * SURFACE H IN MR -Hi > o i H 1 1 Standard HC0' DAYS EXPOSURE Fig. 3. Plasma chloride, actual and standard bicarbonate and hematocrit during 35 days of exposure to air pressure equivalent to 60 feet of seawater and during 2 weeks of subsequent recovery at atmospheric pressure. o control; statistically significant difference from controls at the 5% level and better
13 DAYS AT 60FT DEPTH IN AIR RECOVERY SURFACE H IN AIR DAYS EXPOSURE 14 Days Fig. 4. Plasma ammonia levels during 35 days of exposure to air pressure equivalent to 60 feet of seawater and during 2 weeks of subsequent recovery at atmospheric pressure. o control; statistically significant difference from controls at the 5% level and better DISCUSSION The remarkable increases of arterial PaCC>2 observed during the first experiments (saturation air dive at 50 feet) led us to look for a possible artifact due to narcosis at increased pressure or some other incidental effect. However, the corresponding decreases in plasma chloride levels support the findings of a respiratory acidosis obtained by blood gas analysis. The recovery values, showing a decrease in bicarbonate and increase in chloride, give further credence to the conclusion that a real respiratory acidosis existed during the 50-foot saturation dive on air.
14 TABLE 3 35 DAYS AT 60 FT. DEPTH IN AIR DAY Na (meq. /L plasma) K (meq./l plasma) mean S.E.M. "t" mean S.E.M. "t" CONTROL ,71* * * RECOVERY , * * , * Statistically significant from controls at the 5% level. 10
15 TABLE 4 35 DAYS AT 60 FT. DEPTH IN AIR DAY AMINOACID N (mg/dl plasma) UREA (mg/dl plasma) mean S.E.M. "t" mean S.E.M. "t" CONTROL * RECOVERY ^Statistically significant from controls at the 5% level. During the second air saturation dive at 60 feet, measurements of experimental animals exposed to pressure were in each instance carried out simultaneously with measurements of the same number of control animals. Again, the findings revealed a severe respiratory acidosis in the experimental animals, but not in the con- trol animals. Moreover, measurements during an extended recovery period reflect a response in the acidbase parameters; in particular, the decreases in bicarbonate and increases in chloride, which could have been produced only by a persistent respiratory acidosis during the exposure period. 11
16 TABLE 5 BICARBONATE-CHLORIDE EXCHANGE IN PLASMA OF RATS AT DEPTHS OF 50 and 60 FEET + H Nanamols/L. A PC0 2 mm Hg A HC0 3 meq./l. A Cl~ meq./l. A CONTROL Feet 1 Day Days Days Days CONTROL Feet 1 Day ' Days SO Days Days 93-5 RECOVERY AIR IDay Days Days Days
17 These findings are considered to provide sufficient evidence for the existence of a severe respiratory acidosis during the air saturation dives to 50 and 60 feet. Three factors could have contributed to the observed changes in acid-base parameters: the increased pressures of nitrogen, oxygen and COo. Based on the findings of Lambertsen^on acidbase characteristics in human subjects during 14 days of exposure to an increased atmospheric density, increased nitrogen pressure equivalent to 100 feet of seawater, and normal inspired oxygen tension, any effects of nitrogen per se at increased pressures on acid-base parameters seemed to be very unlikely. According to this report, PaCC>2 showed only slight elevations from sea level control of 41.5 mm Hg to 44.6 mm Hg on Day 7-9 returning to control levels at Days or 41.7 mm Hg. The ph values did not change at all. Increased oxygen levels of 51-57% in the ambient atmosphere could only contribute to a very small rise in PaCC>2 due to interference with the CO2 transport of hemoglobin. However, as has been shown by Lambertsen et or this increase in PaC02 results in a hyperventilation and subsequent decrease in PaCC>2. Breathing air at 3.5 atm. which corresponds to an ambient oxygen concentration of 73.5% surface equivalent, did not significantly alter the arterial CO2 tension or the ph. It must, therefore, be concluded that the increased oxygen levels existing in our experiments could certainly not cause an increase of arterial CO2 tensions amounting to mm Hg. Thus, the increased ambient CO2 level is seen as the only other possible factor which could contribute to such increases in arterial PaCC>2 changes. However, on the basis of the available data it is not possible to state how this CO2 effect is accomplished. If the PaCC>2 and ph data obtained in 50 and 60 foot air saturation dives are compared with PaCC>2 and ph data collected under conditions of acute and chronic hypercapnia in rats^, they are found to lie in the range of values measured during exposure of rats to 3% and 5%C0 2. This means that under increased air pressure, through possibly potentiating effects of increased oxygen and nitrogen, the C0 2 effects, as far as ph and PaC0 2 are concerned, are amplified by a factor of 10. Adaptation to hypercapnia involves an increase of bicarbonate accompanied by a nearly equal decrease in plasma chloride. It has been shown by Polak etafi for the dog and Levitin etar for the rat, that the magnitude of the urinary chloride excretion during chronic hypercapnia is sufficient to account for the decline in plasma chloride. The increased chloride excretion in chronic hypercapnia represents an important defense mechanism for the maintenance of ph in addition to the increased bicarbonate reabsorption in the kidney. In both saturation dives at 50 and 60 feet the increases in plasma bicarbonate and the corresponding decreases in plasma chloride indicate the existence of a chronic hypercapnia, showing the same pattern of 13
18 acid-base changes as seen in hypercapnia under normal atmospheric conditions. Increased plasma potassium levels have also been observed in chronic hypercapnia induced by exposure of guinea pigs to 15% CCv^0 and in patients with respiratory acidosis (Kilburn 2 ). These findings on potassium changes are therefore additional indicators of a chronic respiratory acidosis. An increased urinary ammonia excretion has been found in rats during chronic hypercapnia^' 2 which could be reflected in a lowered blood ammonia level in line with observations made in this study. The stress of rather severe respiratory acidosis imposed by increased ambient CO2 levels during the air saturation dives did not result in any mortality of the rats maintained on a normal diet. This is in agreement with the high resistance to damage from carbon dioxide observed in rats at normal atmospheric pressure. Rats exposed for prolonged periods to 20% CO2 did not show any mortality 1 '. Exploratory histopathological investigations were carried out in 30 rats exposed for various time intervals to pressures equivalent to 50 and 60 feet of seawater and subsequently these animals were decompressed. Light microscopy studies did not show clearly pronounced signs of pulmonary damage in any of the animals. This does not exclude, however, the possibility that systematic studies of a larger number of animals might have revealed slight histopathological changes in the lung, i.e., perivascular and alveolar edema as observed in rats during prolonged exposure to 3% CO2 (Niemoeller and Schaefer 6 ). The ph values measured in the present series of saturation dives correspond with the ph values obtained in rats exposed tercc>2 concentrations of 3% and above. REFERENCES 1. Carter, N. W., D. W. Seldin, and H. C. Teng, Tissue and Renal Response to Chronic Respiratory Acidosis. J. Clin. Invest. 38:949, Kilburn, K. H. Movements of Potassium During Acute Respiratory Acidosis and Recovery. J. Appl. Physiol. 21: , Lambertsen, C. J., J. H. Ewing, R. H. Kough, R. Gould, and M. W. Stroud 3rd. Oxygen Toxicity. Arterial and Internal Jugular Blood Gas Composition in Man During Inhalation of Air, 100% CO2 and 2% CO2 in O2 at 3.5 Atmospheres Ambient Pressure. J. Appl. Physiol. 8: , Lambertsen, C. J. Tektite 2. Scientists in the Sea. Eds. J. W. Miller, J. G. Van Derwalker, and R. A. Waller. U. S. Dept. of the Interior, Aug Levitin, H., W. Branscome, F. H. Epstein. The Pathogenesis of Hypochloremia in Respiratory Acidosis. J. Clin. Invest. 37:1667,
19 6. Niemoeller, H. and K. E. Sehaefer. Development of Hyaline Membranes and Atelectasis in Experimental Chronic Respiratory Acidosis. Proc. Soc. Exp. Biol. & Med. 110: , Sullivan, W. J. and P. J. Dorman The Renal Response to Chronic Respiratory Acidosis. J. Clin. Invest. 34:268, Polak, A., G. D. Haynie, R. M. Hays, and W. B. Schwartz. Effects of Chronic Hypercapnia on Electrolyte and Acid-Base Equilibrium. I. Adaptation. J. Clin. Invest. 40: , Russell, Jane. Note on the Coroimetric Determination of Amino Nitrogen. J. Biol. Chem. 156: 467, Schaefer, K. E., G. F. Bond, W. F. Mazzone, C. R. Carey and J. H. Dougherty. Carbon Dioxide Retention and Metabolic Changes During Prolonged Exposure to High Pressure Environment'. Aerospace Med. 39: , Schaefer, K. E., C. R. Carey, and J. H. Dougherty. Pulmonary Gas Exchange and Urinary Electrolyte Excretion During Saturation-Excursion Diving to Pressure Equivalent to 800 and Feet of Seawater. Aerospace Med. 41: , Schaefer, K. E., H. Niemoeller, A. A. Messier, E. Heyder, and J. Spencer. Chronic CO2 Toxicity: Species Difference in Physiological and Histopathological Effects. NSMRL Report No. 656,
20
21 UNCLASSIFIED Security Classification DOCUMENT CONTROL DATA -R&D (Security classification of title, body of abstract and indexing annotation must be entered when the overall report Is classified) ORIGINATING ACTIVITY (Corporals author) NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY, Naval Submarine Medical Center 3. REPORT TITLE CO EFFECTS ON ACID-BASE BALANCE IN AIR SATURATION DIVES Jfl. REPORT SECURITY CLASSIFICATION Unclassified 2b. GROUP N/A 4. DESCRIPTIVE NOTES (Type of report and inclusive dates) Interim report 5- AUTHOR(S) (First name, middle initial, las< name) Barbara A. HEAVERS, Arthur A. MESSIER, R. MONTANTI and Karl E. SCHAEFER 6 REPOR TDATE 11 April a. CONTRACT OR GRANT NO. b. PROJ EC T NO. MR BOKL.09 7fl. TOTAL NO. OF PAGES 15 9a. ORIGINATOR'S REPORT NUMBERfS) 7b. NO. OF REFS 11 NSMRL Report Number 742 9b. OTHER REPORT No(S} (Any other numbers that may be assigned this report) 10. DISTRIBUTION STATEMENT Approved for public release; distribution unlimited 11- SUPPLEMENTARY NOTES 12. SPONSORING MILI TARY ACTIVITY Naval Submarine Medical Center Box 600 Naval Submarine Base Groton, Connecticut ABSTRACT Blood gas tensions, ph, plasma chloride, sodium and potassium, blood urea, ammonia and amino-acid nitrogen were measured in mature Sprague-Dawley rats at various intervals during and after exposure to pressures equivalent to depth at 50 and 60 feet, lasting for 60 and 35 days, respectively. In both experiments, a consistent decrease in ph values to about 7.30 and a rise in CO tensions to mm Hg in the arterial blood were found. Plasma bicarbonate was elevated 5-7 meq and chloride correspondingly decreased. Plasma potassium was consistently increased in both experiments. These findings indicate the existence of a pronounced respiratory acidosis during the saturation dives to 50 and 60 feet. There is no evidence in the literature which would indicate that as individual agents, elevated pressures ;of nitrogen or oxygen in the ranges observed in these experiments could cause significant acid-base alterations. It must, therefore, be concluded that the increased ambient CO levels in conjunction with the effects of increased oxygen and nitrogen is responsible for the observed changes. When compared with data obtained in rats during acute and chronic hypercapnia the observed PaCO, ph and bicarbonate values correspond to the range of values obtained by exposure to 3% and 5% CO. DD F0RM 1 NO V I *T / W S/N (PAGE 1} UNCLASSIFIED Security Classification
22 UNCLASSIFIED Security Classification KEY WORDS Acid-base balance Respiratory acidosis Air-Saturation Chronic hypercapnia DD, F N o R v M. s 1473 «BACK) (PAGE 2) UNCLASSIFIED Security Classification
THE EFFECT OF INTERMITTENT EXPOSURE TO 3% C0 2 ON ACID-BASE BALANCE AND ELECTROLYTE EXCRETION
 THE EFFECT OF INTERMITTENT EXPOSURE TO 3% C0 2 ON ACID-BASE BALANCE AND ELECTROLYTE EXCRETION by K. E. Schaefer C. C. Morgan A. A. Messier and M. J. Jacey SUBMARINE MEDICAL RESEARCH LABORATORY NAVAL SUBMARINE
THE EFFECT OF INTERMITTENT EXPOSURE TO 3% C0 2 ON ACID-BASE BALANCE AND ELECTROLYTE EXCRETION by K. E. Schaefer C. C. Morgan A. A. Messier and M. J. Jacey SUBMARINE MEDICAL RESEARCH LABORATORY NAVAL SUBMARINE
- How do the carotid bodies sense arterial blood gases? o The carotid bodies weigh 25mg, yet they have their own artery. This means that they have
 - How do the carotid bodies sense arterial blood gases? o The carotid bodies weigh 25mg, yet they have their own artery. This means that they have the highest blood flow of all organs, which makes them
- How do the carotid bodies sense arterial blood gases? o The carotid bodies weigh 25mg, yet they have their own artery. This means that they have the highest blood flow of all organs, which makes them
A COMPRESSION-DECOMPRESSION SCHEDULE FOR PRODUCING DYSBARIC STRESS IN MATURE RATS. Michael J. Jacey Donald V. Tappan and John J.
 A COMPRESSION-DECOMPRESSION SCHEDULE FOR PRODUCING DYSBARIC STRESS IN MATURE RATS by Michael J. Jacey Donald V. Tappan and John J. Wojtowicz NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY REPORT NUMBER 749
A COMPRESSION-DECOMPRESSION SCHEDULE FOR PRODUCING DYSBARIC STRESS IN MATURE RATS by Michael J. Jacey Donald V. Tappan and John J. Wojtowicz NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY REPORT NUMBER 749
Essential Skills Course Acute Care Module. Respiratory Day 2 (Arterial Blood Gases) Pre course Workbook
 Essential Skills Course Acute Care Module Respiratory Day 2 (Arterial Blood Gases) Pre course Workbook Acknowledgements This pre course workbook has been complied and updated with reference to the original
Essential Skills Course Acute Care Module Respiratory Day 2 (Arterial Blood Gases) Pre course Workbook Acknowledgements This pre course workbook has been complied and updated with reference to the original
SUBMARINE BASE, GROTON, CONN. REPORT NUMBER 855
 NAVAL RESEA DICAL ÄTORY SUBMARINE BASE, GROTON, CONN. REPORT NUMBER 855 IMPLEMENTING AN EXCHANGE FROM NITROGEN TO HELIUM IN A LARGE HYPERBARIC CHAMBER by. Kenneth R. Bondi, D. Douglas Wray and James E.
NAVAL RESEA DICAL ÄTORY SUBMARINE BASE, GROTON, CONN. REPORT NUMBER 855 IMPLEMENTING AN EXCHANGE FROM NITROGEN TO HELIUM IN A LARGE HYPERBARIC CHAMBER by. Kenneth R. Bondi, D. Douglas Wray and James E.
 Question 1: Define vital capacity. What is its significance? Vital capacity is the maximum volume of air that can be exhaled after a maximum inspiration. It is about 3.5 4.5 litres in the human body. It
Question 1: Define vital capacity. What is its significance? Vital capacity is the maximum volume of air that can be exhaled after a maximum inspiration. It is about 3.5 4.5 litres in the human body. It
Chapter 17 The Respiratory System: Gas Exchange and Regulation of Breathing
 Chapter 17 The Respiratory System: Gas Exchange and Regulation of Breathing Overview of Pulmonary Circulation o Diffusion of Gases o Exchange of Oxygen and Carbon Dioxide o Transport of Gases in the Blood
Chapter 17 The Respiratory System: Gas Exchange and Regulation of Breathing Overview of Pulmonary Circulation o Diffusion of Gases o Exchange of Oxygen and Carbon Dioxide o Transport of Gases in the Blood
Respiratory physiology II.
 Respiratory physiology II. Learning objectives: 29. Pulmonary gas exchange. 30. Oxygen transport in the blood. 31. Carbon-dioxide transport in the blood. 1 Pulmonary gas exchange The transport mechanism
Respiratory physiology II. Learning objectives: 29. Pulmonary gas exchange. 30. Oxygen transport in the blood. 31. Carbon-dioxide transport in the blood. 1 Pulmonary gas exchange The transport mechanism
Oxygen convulsions are believed by many workers to be caused by an accumulation
 272 J. Physiol. (I949) I09, 272-280 6I2.223.II:6I2.26I THE ROLE OF CARBON DIOXIDE IN OXYGEN POISONING BY H. J. TAYLOR From the Royal Naval Physiological Laboratory, Alverstoke, Hants (Received 26 March
272 J. Physiol. (I949) I09, 272-280 6I2.223.II:6I2.26I THE ROLE OF CARBON DIOXIDE IN OXYGEN POISONING BY H. J. TAYLOR From the Royal Naval Physiological Laboratory, Alverstoke, Hants (Received 26 March
Chapter 4: Ventilation Test Bank MULTIPLE CHOICE
 Instant download and all chapters Test Bank Respiratory Care Anatomy and Physiology Foundations for Clinical Practice 3rd Edition Will Beachey https://testbanklab.com/download/test-bank-respiratory-care-anatomy-physiologyfoundations-clinical-practice-3rd-edition-will-beachey/
Instant download and all chapters Test Bank Respiratory Care Anatomy and Physiology Foundations for Clinical Practice 3rd Edition Will Beachey https://testbanklab.com/download/test-bank-respiratory-care-anatomy-physiologyfoundations-clinical-practice-3rd-edition-will-beachey/
Submarine Base, Groton, Conn. REPORT NUMBER 668
 ARINE.W Submarine Base, Groton, Conn. REPORT NUMBER 668 HYPERBARIC AIR AND CORNEAL VASCULARIZATION by Robert J. Kaiser CDRMCUSNR and Donald W. Klopp LCDR MC USN Bureau of Medicine and Surgery, Navy Department
ARINE.W Submarine Base, Groton, Conn. REPORT NUMBER 668 HYPERBARIC AIR AND CORNEAL VASCULARIZATION by Robert J. Kaiser CDRMCUSNR and Donald W. Klopp LCDR MC USN Bureau of Medicine and Surgery, Navy Department
Lung Volumes and Capacities
 Lung Volumes and Capacities Normally the volume of air entering the lungs during a single inspiration is approximately equal to the volume leaving on the subsequent expiration and is called the tidal volume.
Lung Volumes and Capacities Normally the volume of air entering the lungs during a single inspiration is approximately equal to the volume leaving on the subsequent expiration and is called the tidal volume.
(Received 16 January 1946)
 186 J. Physiol. (I946) I05, I86-I90 6I2.2I5.9 THE ABSORPTION OF FLUIDS FROM THE LUNGS BY F. C. COURTICE AND P. J. PHIPPS From the Experimental Station, Porton and the Laboratory of Physiology, Oxford (Received
186 J. Physiol. (I946) I05, I86-I90 6I2.2I5.9 THE ABSORPTION OF FLUIDS FROM THE LUNGS BY F. C. COURTICE AND P. J. PHIPPS From the Experimental Station, Porton and the Laboratory of Physiology, Oxford (Received
Exam Key. NROSCI/BIOSC 1070 and MSNBIO 2070 Exam # 2 October 28, 2016 Total POINTS: % of grade in class
 NROSCI/BIOSC 1070 and MSNBIO 2070 Exam # 2 October 28, 2016 Total POINTS: 100 20% of grade in class 1) An arterial blood sample for a patient at sea level is obtained, and the following physiological values
NROSCI/BIOSC 1070 and MSNBIO 2070 Exam # 2 October 28, 2016 Total POINTS: 100 20% of grade in class 1) An arterial blood sample for a patient at sea level is obtained, and the following physiological values
NROSCI/BIOSC 1070 and MSNBIO 2070 Exam # 2 October 28, 2016 Total POINTS: % of grade in class
 NROSCI/BIOSC 1070 and MSNBIO 2070 Exam # 2 October 28, 2016 Total POINTS: 100 20% of grade in class 1) An arterial blood sample for a patient at sea level is obtained, and the following physiological values
NROSCI/BIOSC 1070 and MSNBIO 2070 Exam # 2 October 28, 2016 Total POINTS: 100 20% of grade in class 1) An arterial blood sample for a patient at sea level is obtained, and the following physiological values
The physiological functions of respiration and circulation. Mechanics. exercise 7. Respiratory Volumes. Objectives
 exercise 7 Respiratory System Mechanics Objectives 1. To explain how the respiratory and circulatory systems work together to enable gas exchange among the lungs, blood, and body tissues 2. To define respiration,
exercise 7 Respiratory System Mechanics Objectives 1. To explain how the respiratory and circulatory systems work together to enable gas exchange among the lungs, blood, and body tissues 2. To define respiration,
Physiology Unit 4 RESPIRATORY PHYSIOLOGY
 Physiology Unit 4 RESPIRATORY PHYSIOLOGY In Physiology Today Respiration External respiration ventilation gas exchange Internal respiration cellular respiration gas exchange Respiratory Cycle Inspiration
Physiology Unit 4 RESPIRATORY PHYSIOLOGY In Physiology Today Respiration External respiration ventilation gas exchange Internal respiration cellular respiration gas exchange Respiratory Cycle Inspiration
Capnography in the Veterinary Technician Toolbox. Katie Pinner BS, LVT Bush Advanced Veterinary Imaging Richmond, VA
 Capnography in the Veterinary Technician Toolbox Katie Pinner BS, LVT Bush Advanced Veterinary Imaging Richmond, VA What are Respiration and Ventilation? Respiration includes all those chemical and physical
Capnography in the Veterinary Technician Toolbox Katie Pinner BS, LVT Bush Advanced Veterinary Imaging Richmond, VA What are Respiration and Ventilation? Respiration includes all those chemical and physical
TECHNICAL BULLETIN. TriControls. Control Materials for Liquid QC and Calibration Verification
 i-stat TECHNICAL BULLETIN Tris Materials for Liquid QC and OVERVIEW As part of the READi initiative (Responds, Enhances, And Delivers innovations), Abbott Point of Care (APOC) has released a new set of
i-stat TECHNICAL BULLETIN Tris Materials for Liquid QC and OVERVIEW As part of the READi initiative (Responds, Enhances, And Delivers innovations), Abbott Point of Care (APOC) has released a new set of
HCO - 3 H 2 CO 3 CO 2 + H H H + Breathing rate is regulated by blood ph and C02. CO2 and Bicarbonate act as a ph Buffer in the blood
 Breathing rate is regulated by blood ph and C02 breathing reduces plasma [CO2]; plasma [CO2] increases breathing. When C02 levels are high, breating rate increases to blow off C02 In low C02 conditions,
Breathing rate is regulated by blood ph and C02 breathing reduces plasma [CO2]; plasma [CO2] increases breathing. When C02 levels are high, breating rate increases to blow off C02 In low C02 conditions,
RESPIRATORY REGULATION DURING EXERCISE
 RESPIRATORY REGULATION DURING EXERCISE Respiration Respiration delivery of oxygen to and removal of carbon dioxide from the tissue External respiration ventilation and exchange of gases in the lung Internal
RESPIRATORY REGULATION DURING EXERCISE Respiration Respiration delivery of oxygen to and removal of carbon dioxide from the tissue External respiration ventilation and exchange of gases in the lung Internal
P215 Respiratory System, Part 2
 P15 Respiratory System, Part Gas Exchange Oxygen and Carbon Dioxide constant need for oxygen constant production of carbon dioxide exchange (and movement) lung alveoli pulmonary arteries pulmonary capillaries
P15 Respiratory System, Part Gas Exchange Oxygen and Carbon Dioxide constant need for oxygen constant production of carbon dioxide exchange (and movement) lung alveoli pulmonary arteries pulmonary capillaries
Respiration - Human 1
 Respiration - Human 1 At the end of the lectures on respiration you should be able to, 1. Describe events in the respiratory processes 2. Discuss the mechanism of lung ventilation in human 3. Discuss the
Respiration - Human 1 At the end of the lectures on respiration you should be able to, 1. Describe events in the respiratory processes 2. Discuss the mechanism of lung ventilation in human 3. Discuss the
Collin County Community College. Lung Physiology
 Collin County Community College BIOL. 2402 Anatomy & Physiology WEEK 9 Respiratory System 1 Lung Physiology Factors affecting Ventillation 1. Airway resistance Flow = Δ P / R Most resistance is encountered
Collin County Community College BIOL. 2402 Anatomy & Physiology WEEK 9 Respiratory System 1 Lung Physiology Factors affecting Ventillation 1. Airway resistance Flow = Δ P / R Most resistance is encountered
J. Physiol. (I941) I00, I98-21I 6I :6I2.825
 198 J. Physiol. (I941) I00, I9821I 6I2.22.02:6I2.825 THE EFFECT OF OXYGEN LACK ON THE CEREBRAL CIRCULATION BY F. C. COURTICE From the Departments of Physiology and of Surgery, Oxford (Received 24 March
198 J. Physiol. (I941) I00, I9821I 6I2.22.02:6I2.825 THE EFFECT OF OXYGEN LACK ON THE CEREBRAL CIRCULATION BY F. C. COURTICE From the Departments of Physiology and of Surgery, Oxford (Received 24 March
Respiratory Pulmonary Ventilation
 Respiratory Pulmonary Ventilation Pulmonary Ventilation Pulmonary ventilation is the act of breathing and the first step in the respiratory process. Pulmonary ventilation brings in air with a new supply
Respiratory Pulmonary Ventilation Pulmonary Ventilation Pulmonary ventilation is the act of breathing and the first step in the respiratory process. Pulmonary ventilation brings in air with a new supply
Levels of CO2 in Arterial Blood of Carp under Carbon Dioxide Anesthesia
 J. Nutr. Sci. Vitaminol., 28, 35-39, 1982 Levels of CO2 in Arterial Blood of Carp under Carbon Dioxide Anesthesia Hisateru MITSUDA, Saburo UENO, Hiroshi MIZUNO, Tadashi UEDA, Hiromi FUJIKAWA, Tomoko NOHARA,
J. Nutr. Sci. Vitaminol., 28, 35-39, 1982 Levels of CO2 in Arterial Blood of Carp under Carbon Dioxide Anesthesia Hisateru MITSUDA, Saburo UENO, Hiroshi MIZUNO, Tadashi UEDA, Hiromi FUJIKAWA, Tomoko NOHARA,
Oxygen and Carbon dioxide Transport. Dr. Laila Al-Dokhi
 Oxygen and Carbon dioxide Transport Dr. Laila Al-Dokhi Objectives 1. Understand the forms of oxygen transport in the blood, the importance of each. 2. Differentiate between O2 capacity, O2 content and
Oxygen and Carbon dioxide Transport Dr. Laila Al-Dokhi Objectives 1. Understand the forms of oxygen transport in the blood, the importance of each. 2. Differentiate between O2 capacity, O2 content and
Section Three Gas transport
 Section Three Gas transport Lecture 6: Oxygen transport in blood. Carbon dioxide in blood. Objectives: i. To describe the carriage of O2 in blood. ii. iii. iv. To explain the oxyhemoglobin dissociation
Section Three Gas transport Lecture 6: Oxygen transport in blood. Carbon dioxide in blood. Objectives: i. To describe the carriage of O2 in blood. ii. iii. iv. To explain the oxyhemoglobin dissociation
PICU Resident Self-Study Tutorial The Basic Physics of Oxygen Transport. I was told that there would be no math!
 Physiology of Oxygen Transport PICU Resident Self-Study Tutorial I was told that there would be no math! INTRODUCTION Christopher Carroll, MD Although cells rely on oxygen for aerobic metabolism and viability,
Physiology of Oxygen Transport PICU Resident Self-Study Tutorial I was told that there would be no math! INTRODUCTION Christopher Carroll, MD Although cells rely on oxygen for aerobic metabolism and viability,
Respiratory System Study Guide, Chapter 16
 Part I. Clinical Applications Name: Respiratory System Study Guide, Chapter 16 Lab Day/Time: 1. A person with ketoacidosis may hyperventilate. Explain why this occurs, and explain why this hyperventilation
Part I. Clinical Applications Name: Respiratory System Study Guide, Chapter 16 Lab Day/Time: 1. A person with ketoacidosis may hyperventilate. Explain why this occurs, and explain why this hyperventilation
CARBON DIOXIDE EFFECTS UNDER CONDITIONS OF RAISED ENVIRONMENTAL PRESSURE. K. E. Schaefer, M.D.
 CARBON DIOXIDE EFFECTS UNDER CONDITIONS OF RAISED ENVIRONMENTAL PRESSURE by K. E. Schaefer, M.D. NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY REPORT NUMBER 804 Bureau of Medicine and Surgery, Navy Department
CARBON DIOXIDE EFFECTS UNDER CONDITIONS OF RAISED ENVIRONMENTAL PRESSURE by K. E. Schaefer, M.D. NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY REPORT NUMBER 804 Bureau of Medicine and Surgery, Navy Department
VOLUNTARY BREATHHOLDING. I. PULMONARY GAS
 VOLUNTARY BREATHHOLDING. I. PULMONARY GAS EXCHANGE DURING BREATHHOLDING'1 By CHARLES D. STEVENS, EUGENE B. FERRIS, JOSEPH P. WEBB, GEORGE L. ENGEL, AND MYRTLE LOGAN (From the Departments of Internal Medicine
VOLUNTARY BREATHHOLDING. I. PULMONARY GAS EXCHANGE DURING BREATHHOLDING'1 By CHARLES D. STEVENS, EUGENE B. FERRIS, JOSEPH P. WEBB, GEORGE L. ENGEL, AND MYRTLE LOGAN (From the Departments of Internal Medicine
The over-ventilated cat shows a similar adjustment to diminished. being over-ventilated, and he considered that on that account there was
 6I2.235:6I2.26I THE SOURCE OF COa EXPIRED AND THE SITE OF ITS RETENTION. BY LAURENCE IRVING, J. K. W. FERGUSON AND F. B. PLEWES. (From the Department of Physiology, University of Toronto.) AFTER evisceration
6I2.235:6I2.26I THE SOURCE OF COa EXPIRED AND THE SITE OF ITS RETENTION. BY LAURENCE IRVING, J. K. W. FERGUSON AND F. B. PLEWES. (From the Department of Physiology, University of Toronto.) AFTER evisceration
EMERGENCY MEDICINE AND THE LABORATORY. Ehsan Bolvardi(MD)
 EMERGENCY MEDICINE AND THE LABORATORY Ehsan Bolvardi(MD) bolvardie@mums.ac.ir Objective ED and laboratory Inappropriate use of laboratory ABG sampling ABG sampling error Introduction The laboratory is
EMERGENCY MEDICINE AND THE LABORATORY Ehsan Bolvardi(MD) bolvardie@mums.ac.ir Objective ED and laboratory Inappropriate use of laboratory ABG sampling ABG sampling error Introduction The laboratory is
Chapter 11: Respiratory System Review Assignment
 Name: Date: Mark: / 45 Chapter 11: Respiratory System Review Assignment Multiple Choice = 45 Marks Identify the choice that best completes the statement or answers the question. 1. Which of the following
Name: Date: Mark: / 45 Chapter 11: Respiratory System Review Assignment Multiple Choice = 45 Marks Identify the choice that best completes the statement or answers the question. 1. Which of the following
CHAPTER 3: The cardio-respiratory system
 : The cardio-respiratory system Exam style questions - text book pages 44-45 1) Describe the structures involved in gaseous exchange in the lungs and explain how gaseous exchange occurs within this tissue.
: The cardio-respiratory system Exam style questions - text book pages 44-45 1) Describe the structures involved in gaseous exchange in the lungs and explain how gaseous exchange occurs within this tissue.
Circulatory And Respiration
 Circulatory And Respiration Composition Of Blood Blood Heart 200mmHg 120mmHg Aorta Artery Arteriole 50mmHg Capillary Bed Venule Vein Vena Cava Heart Differences Between Arteries and Veins Veins transport
Circulatory And Respiration Composition Of Blood Blood Heart 200mmHg 120mmHg Aorta Artery Arteriole 50mmHg Capillary Bed Venule Vein Vena Cava Heart Differences Between Arteries and Veins Veins transport
Chapter 23. Gas Exchange and Transportation
 Chapter 23 Gas Exchange and Transportation What is air? Mixture of gasses 78.6 % nitrogen 20.9% oxygen 0.04% carbon dioxide 0 4% water vapor depending on temperature and humidity and minor gases argon,
Chapter 23 Gas Exchange and Transportation What is air? Mixture of gasses 78.6 % nitrogen 20.9% oxygen 0.04% carbon dioxide 0 4% water vapor depending on temperature and humidity and minor gases argon,
Chapter 23. Gas Exchange and Transportation
 Chapter 23 Gas Exchange and Transportation What is air? Mixture of gasses 78.6 % nitrogen 20.9% oxygen 0.04% carbon dioxide 0 4% water vapor depending on temperature and humidity other minor gases argon,
Chapter 23 Gas Exchange and Transportation What is air? Mixture of gasses 78.6 % nitrogen 20.9% oxygen 0.04% carbon dioxide 0 4% water vapor depending on temperature and humidity other minor gases argon,
Section Two Diffusion of gases
 Section Two Diffusion of gases Lecture 5: Partial pressure and the composition of gasses in air. Factors affecting diffusion of gases. Ventilation perfusion ratio effect on alveolar gas concentration.
Section Two Diffusion of gases Lecture 5: Partial pressure and the composition of gasses in air. Factors affecting diffusion of gases. Ventilation perfusion ratio effect on alveolar gas concentration.
I Physical Principles of Gas Exchange
 Respiratory Gases Exchange Dr Badri Paudel, M.D. 2 I Physical Principles of Gas Exchange 3 Partial pressure The pressure exerted by each type of gas in a mixture Diffusion of gases through liquids Concentration
Respiratory Gases Exchange Dr Badri Paudel, M.D. 2 I Physical Principles of Gas Exchange 3 Partial pressure The pressure exerted by each type of gas in a mixture Diffusion of gases through liquids Concentration
Submarine Base, Groton, Conn.
 I UG NE Submarine Base, Groton, Conn. REPORT NUMBER 52 CARBON DIOXIDE RETENTION DURING PROLONGED EXPOSURE TO HIGH PRESSURE ENVIRONMENT by Karl E. Schaefer, M.D., George F. Bond, CAPT MC USN, Walter F.
I UG NE Submarine Base, Groton, Conn. REPORT NUMBER 52 CARBON DIOXIDE RETENTION DURING PROLONGED EXPOSURE TO HIGH PRESSURE ENVIRONMENT by Karl E. Schaefer, M.D., George F. Bond, CAPT MC USN, Walter F.
660 mm Hg (normal, 100 mm Hg, room air) Paco, (arterial Pc02) 36 mm Hg (normal, 40 mm Hg) % saturation 50% (normal, 95%-100%)
 148 PHYSIOLOGY CASES AND PROBLEMS Case 26 Carbon Monoxide Poisoning Herman Neiswander is a 65-year-old retired landscape architect in northern Wisconsin. One cold January morning, he decided to warm his
148 PHYSIOLOGY CASES AND PROBLEMS Case 26 Carbon Monoxide Poisoning Herman Neiswander is a 65-year-old retired landscape architect in northern Wisconsin. One cold January morning, he decided to warm his
Human Biology Respiratory System
 Human Biology Respiratory System Respiratory System Responsible for process of breathing Works in cooperation with Circulatory system Three types: 1. Internal Respiration 2. External Respiration 3. Cellular
Human Biology Respiratory System Respiratory System Responsible for process of breathing Works in cooperation with Circulatory system Three types: 1. Internal Respiration 2. External Respiration 3. Cellular
Gases and Respiration. Respiration Overview I
 Respiration Overview I Respiration Overview II Gas Laws Equation of State: PV = nrt Same volumes of different gases have same # of molecules BTPS: body temp, atmospheric pressure, saturated ATPS: ambient
Respiration Overview I Respiration Overview II Gas Laws Equation of State: PV = nrt Same volumes of different gases have same # of molecules BTPS: body temp, atmospheric pressure, saturated ATPS: ambient
AIIMS, New Delhi. Dr. K. K. Deepak, Prof. & HOD, Physiology AIIMS, New Delhi Dr. Geetanjali Bade, Asst. Professor AIIMS, New Delhi
 Course : PG Pathshala-Biophysics Paper 13 : Physiological Biophysics Module 17 : Gas transport and pulmonary circulation Principal Investigator: Co-Principal Investigator: Paper Coordinator: Content Writer:
Course : PG Pathshala-Biophysics Paper 13 : Physiological Biophysics Module 17 : Gas transport and pulmonary circulation Principal Investigator: Co-Principal Investigator: Paper Coordinator: Content Writer:
Respiratory System Physiology. Dr. Vedat Evren
 Respiratory System Physiology Dr. Vedat Evren Respiration Processes involved in oxygen transport from the atmosphere to the body tissues and the release and transportation of carbon dioxide produced in
Respiratory System Physiology Dr. Vedat Evren Respiration Processes involved in oxygen transport from the atmosphere to the body tissues and the release and transportation of carbon dioxide produced in
Submarine Base, Groton, Conn.
 r*-! PUBLICATIONS B:. t,,..r f^r-.! j.'fdical C3&7ER L, L>. t-."-- - -» JJ ff? ^ *W -'«--'fr- *" ^ 5 --^--fc-is*^ V»?g ««.»1/ j m : :# V j Jf «r% i ls «"% *Ä\ - ' > /vjv*äs.. /> Submarine Base, Groton,
r*-! PUBLICATIONS B:. t,,..r f^r-.! j.'fdical C3&7ER L, L>. t-."-- - -» JJ ff? ^ *W -'«--'fr- *" ^ 5 --^--fc-is*^ V»?g ««.»1/ j m : :# V j Jf «r% i ls «"% *Ä\ - ' > /vjv*äs.. /> Submarine Base, Groton,
plethysmographic methods that when the subject was pinched on the upper
 24 J. Physiol. (I95I) II2, 24-2I 6I2.I5.6II.976 THE DECREASE IN HAND BLOOD FLOW FOLLOWING INFLATION OF AN ARTERIAL OCCLUSION CUFF ON THE OPPOSITE ARM BY IAN C. RODDIE From the Department of Physiology,
24 J. Physiol. (I95I) II2, 24-2I 6I2.I5.6II.976 THE DECREASE IN HAND BLOOD FLOW FOLLOWING INFLATION OF AN ARTERIAL OCCLUSION CUFF ON THE OPPOSITE ARM BY IAN C. RODDIE From the Department of Physiology,
Gas Exchange ACTIVITY OVERVIEW SUMMARY KEY CONCEPTS AND PROCESS SKILLS KEY VOCABULARY. Teacher s Guide B-75 L A B O R ATO R Y
 Gas Exchange 17 40- to 2 50-minute sessions ACTIVITY OVERVIEW L A B O R ATO R Y SUMMARY This activity explores the role of the respiratory system in the regulation of gases in the blood. Students investigate
Gas Exchange 17 40- to 2 50-minute sessions ACTIVITY OVERVIEW L A B O R ATO R Y SUMMARY This activity explores the role of the respiratory system in the regulation of gases in the blood. Students investigate
Submarine Base, Groton, Conn.
 Submarine Base, Groton, Conn. REPORT NUMBER 554 OBSERVATIONS OF INTRA-OCULAR PRESSURE CHANGES DURING AN FBM PATROL AS MEASURED BY SCHIOTZ TONOMETRY by James G. Geisinger Lieutenant, MC, U. S. Navy Bureau
Submarine Base, Groton, Conn. REPORT NUMBER 554 OBSERVATIONS OF INTRA-OCULAR PRESSURE CHANGES DURING AN FBM PATROL AS MEASURED BY SCHIOTZ TONOMETRY by James G. Geisinger Lieutenant, MC, U. S. Navy Bureau
Respiration. The resspiratory system
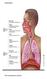 Respiration The resspiratory system The Alveoli The lungs have about 300 million alveoli, with a total crosssec onal area of 50 70 m2.. Each alveolar sac is surrounded by blood capillaries. The walls of
Respiration The resspiratory system The Alveoli The lungs have about 300 million alveoli, with a total crosssec onal area of 50 70 m2.. Each alveolar sac is surrounded by blood capillaries. The walls of
CDI Blood Parameter Monitoring System 500 A New Tool for the Clinical Perfusionist
 Original Article Blood Parameter Monitoring System 500 A New Tool for the Clinical Perfusionist David W. Fried, MS Ed, CCP; Joseph J. Leo, BS, CCP; Gabriel J. Mattioni, BS, CCP; Hasratt Mohamed, CCP; Raymond
Original Article Blood Parameter Monitoring System 500 A New Tool for the Clinical Perfusionist David W. Fried, MS Ed, CCP; Joseph J. Leo, BS, CCP; Gabriel J. Mattioni, BS, CCP; Hasratt Mohamed, CCP; Raymond
4/18/2012. Northern Fur Seal Three Foraging Patterns. Thermocline. Diving Physiology and Behavior
 Northern Fur Seal Three Foraging Patterns Thermocline Diving Physiology and Behavior 1 Fundamental Constraint on Foraging Behavior Return to Surface to Breathe 2 Studies of Dive Behavior Dive depths from
Northern Fur Seal Three Foraging Patterns Thermocline Diving Physiology and Behavior 1 Fundamental Constraint on Foraging Behavior Return to Surface to Breathe 2 Studies of Dive Behavior Dive depths from
Evaluating the Effects of Stressors on Immune Function during Simulated Dives in Marine Mammals
 DISTRIBUTION STATEMENT A. Approved for public release; distribution is unlimited. Evaluating the Effects of Stressors on Immune Function during Simulated Dives in Marine Mammals Tracy Romano and Laura
DISTRIBUTION STATEMENT A. Approved for public release; distribution is unlimited. Evaluating the Effects of Stressors on Immune Function during Simulated Dives in Marine Mammals Tracy Romano and Laura
NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY
 NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY SUBMARINE BASE, GROTON, CONN. REPORT NO. 1102 EFFECTS OF HYPERBARIC CONDITIONS ON CORNEAL PHYSIOLOGY WITH HYDROGEL CONTACT LENSES by Joseph F. Molinari and James
NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY SUBMARINE BASE, GROTON, CONN. REPORT NO. 1102 EFFECTS OF HYPERBARIC CONDITIONS ON CORNEAL PHYSIOLOGY WITH HYDROGEL CONTACT LENSES by Joseph F. Molinari and James
What does the % represent on the beakers?
 DISSOLVED OXYGEN VIDEO FAQs What does the % represent on the beakers? What are the glass tubes to beakers for? How is the temperature being kept the same (at 5 o then 35 o )? What is salinity in parts
DISSOLVED OXYGEN VIDEO FAQs What does the % represent on the beakers? What are the glass tubes to beakers for? How is the temperature being kept the same (at 5 o then 35 o )? What is salinity in parts
Point-of-Care Testing: A Cardiovascular Perfusionist s Perspective
 Point-of-Care Testing: A Cardiovascular Perfusionist s Perspective Cory M. Alwardt, PhD, CCP Chief Perfusionist/ECMO Coordinator Assistant Professor of Surgery Mayo Clinic Hospital, Phoenix alwardt.cory@mayo.edu
Point-of-Care Testing: A Cardiovascular Perfusionist s Perspective Cory M. Alwardt, PhD, CCP Chief Perfusionist/ECMO Coordinator Assistant Professor of Surgery Mayo Clinic Hospital, Phoenix alwardt.cory@mayo.edu
Respiratory Physiology Gaseous Exchange
 Respiratory Physiology Gaseous Exchange Session Objectives. What you will cover Basic anatomy of the lung including airways Breathing movements Lung volumes and capacities Compliance and Resistance in
Respiratory Physiology Gaseous Exchange Session Objectives. What you will cover Basic anatomy of the lung including airways Breathing movements Lung volumes and capacities Compliance and Resistance in
Some Clinical Aspects on the Blood Gas Physiology
 Special Article* Some Clinical Aspects on the Blood Gas Physiology Hiroshi Sasamoto Professor and Chairman, Department of Medicine, School of Medicine Keio University, Shinjuku, Tokyo Recent trends on
Special Article* Some Clinical Aspects on the Blood Gas Physiology Hiroshi Sasamoto Professor and Chairman, Department of Medicine, School of Medicine Keio University, Shinjuku, Tokyo Recent trends on
Monday, ! Today: Respiratory system! 5/20/14! Transport of Blood! What we ve been covering! Circulatory system! Parts of blood! Heart! tubing!
 Monday, 5.19.14! What we ve been covering! Circulatory system! Parts of blood! Heart! tubing! Transport of Blood! What is transported! Nutrients! Oxygen! Carbon Dioxide! Hormones! Antibodies! What it is/does!
Monday, 5.19.14! What we ve been covering! Circulatory system! Parts of blood! Heart! tubing! Transport of Blood! What is transported! Nutrients! Oxygen! Carbon Dioxide! Hormones! Antibodies! What it is/does!
Physical Chemistry of Gases: Gas Exchange Linda Costanzo, Ph.D.
 Physical Chemistry of Gases: Gas Exchange Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. Application of the gas laws to pulmonary physiology. 2. How to
Physical Chemistry of Gases: Gas Exchange Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. Application of the gas laws to pulmonary physiology. 2. How to
Unit II Problem 4 Physiology: Diffusion of Gases and Pulmonary Circulation
 Unit II Problem 4 Physiology: Diffusion of Gases and Pulmonary Circulation - Physical principles of gases: Pressure of a gas is caused by the movement of its molecules against a surface (more concentration
Unit II Problem 4 Physiology: Diffusion of Gases and Pulmonary Circulation - Physical principles of gases: Pressure of a gas is caused by the movement of its molecules against a surface (more concentration
Retinal vascular response to breathing increased carbon dioxide and oxygen concentrations. Regina Frayser and John B. Hickam
 Retinal vascular response to breathing increased carbon dioxide and oxygen concentrations Regina Frayser and John B. Hickam The retina has a high rate of oxygen consumption, and the retinal vessels are
Retinal vascular response to breathing increased carbon dioxide and oxygen concentrations Regina Frayser and John B. Hickam The retina has a high rate of oxygen consumption, and the retinal vessels are
Douglas and Haldane(2) has shown that the oxygen determinations. since it forms the basis of the "Coefficient of Utilisation" (Krrogh) and
 THE MEASUREMENT OF THE OXYGEN CONTENT OF THE MIXED VENOUS BLOOD, AND OF THE VOLUME OF BLOOD CIRCULATING PER MINUTE. BY J. BARCROFT, F. J. W. ROUGHTON AND R. SHOJI. (From the Physiological Laboratory, Cambridge.)
THE MEASUREMENT OF THE OXYGEN CONTENT OF THE MIXED VENOUS BLOOD, AND OF THE VOLUME OF BLOOD CIRCULATING PER MINUTE. BY J. BARCROFT, F. J. W. ROUGHTON AND R. SHOJI. (From the Physiological Laboratory, Cambridge.)
49 Arterial Blood Gases
 49 Arterial Blood Gases E. P. TRULOCK, III Definition Arterial blood gases (ABGs) is a collective term applied to three separate measurements-ph, Pco 2, and Poe -generally made together to evaluate acid-base
49 Arterial Blood Gases E. P. TRULOCK, III Definition Arterial blood gases (ABGs) is a collective term applied to three separate measurements-ph, Pco 2, and Poe -generally made together to evaluate acid-base
Blood Parameter Monitoring System 550
 Technical Compendium CDI Blood Parameter Monitoring System 550 An overview of the CDI System 550 and its industry leading technology. CDI System 550 Measures or Calculates 12 Critical Blood Parameters
Technical Compendium CDI Blood Parameter Monitoring System 550 An overview of the CDI System 550 and its industry leading technology. CDI System 550 Measures or Calculates 12 Critical Blood Parameters
IV. FROM AQUATIC TO ATMOSPHERIC BREATHING: THE TRACHEA & THE LUNG
 GAS EXCHANGE AND TRANSPORT I. INTRODUCTION: Heterotrophs oxidize carbon cmpds using O 2 to generate CO 2 & H 2 O. This is cellular respiration II. HOW GAS ENTERS A CELL A. The composition of air: 79% N
GAS EXCHANGE AND TRANSPORT I. INTRODUCTION: Heterotrophs oxidize carbon cmpds using O 2 to generate CO 2 & H 2 O. This is cellular respiration II. HOW GAS ENTERS A CELL A. The composition of air: 79% N
COMPARISON OF BIOCHEMICAL RESPONSES BETWEEN SINGLE AND REPEATED EXPOSURES TO AIR AT 6.7 ATA. E. Heyder, M. J. Jacey, and D. V.
 COMPARISON OF BIOCHEMICAL RESPONSES BETWEEN SINGLE AND REPEATED EXPOSURES TO AIR AT 6.7 ATA by E. Heyder, M. J. Jacey, and D. V. Tappan NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY REPORT NUMBER 810 Bureau
COMPARISON OF BIOCHEMICAL RESPONSES BETWEEN SINGLE AND REPEATED EXPOSURES TO AIR AT 6.7 ATA by E. Heyder, M. J. Jacey, and D. V. Tappan NAVAL SUBMARINE MEDICAL RESEARCH LABORATORY REPORT NUMBER 810 Bureau
technique which have led to increased precision
 THE ACCURACY OF DIRECT DETERMINATIONS OF OXYGEN AND CARBON DIOXIDE TENSIONS IN HUMAN BLOOD IN VITRO 1 By GILES F. FILLEY, ESTHER GAY, AND GEORGE W. WRIGHT (From the Department of Physiology, the Edwtvard
THE ACCURACY OF DIRECT DETERMINATIONS OF OXYGEN AND CARBON DIOXIDE TENSIONS IN HUMAN BLOOD IN VITRO 1 By GILES F. FILLEY, ESTHER GAY, AND GEORGE W. WRIGHT (From the Department of Physiology, the Edwtvard
Submarine Base, Groton, Conn.
 LICAIIONS BRANCH. fjrim MEDICAL CENTER tlle COPY NO. -*- 1 J Submarine Base, Groton, Conn. REPORT NUMBER 697 THE INTELLIGIBILITY OF PROCESSED HELIUM SPEECH AT DIVING DEPTHS GREATER THAN 1000 FEET by Thomas
LICAIIONS BRANCH. fjrim MEDICAL CENTER tlle COPY NO. -*- 1 J Submarine Base, Groton, Conn. REPORT NUMBER 697 THE INTELLIGIBILITY OF PROCESSED HELIUM SPEECH AT DIVING DEPTHS GREATER THAN 1000 FEET by Thomas
RESPIRATION III SEMESTER BOTANY MODULE II
 III SEMESTER BOTANY MODULE II RESPIRATION Lung Capacities and Volumes Tidal volume (TV) air that moves into and out of the lungs with each breath (approximately 500 ml) Inspiratory reserve volume (IRV)
III SEMESTER BOTANY MODULE II RESPIRATION Lung Capacities and Volumes Tidal volume (TV) air that moves into and out of the lungs with each breath (approximately 500 ml) Inspiratory reserve volume (IRV)
Challenges in Undersea Medicine
 Challenges in Undersea Medicine CAPT Fred Yeo, MD Commanding Officer Naval Submarine Medical Research Laboratory April 2016 DISTRIBUTION STATEMENT A. Approved for public release. UNCLASSIFIED 1 Panel Members
Challenges in Undersea Medicine CAPT Fred Yeo, MD Commanding Officer Naval Submarine Medical Research Laboratory April 2016 DISTRIBUTION STATEMENT A. Approved for public release. UNCLASSIFIED 1 Panel Members
200602q PULMONARY FUNCTION AFTER OXYGEN-ACCELERATED DECOMPRESSIONS FROM REPETITIVE SUB-SATURATION AIR DIVES
 Navy Experimental Diving Unit TA 01-10 321 Bullfinch Rd. NEDU TR 05-05 Panama City, FL 32407-7015 April 2005 PULMONARY FUNCTION AFTER OXYGEN-ACCELERATED DECOMPRESSIONS FROM REPETITIVE SUB-SATURATION AIR
Navy Experimental Diving Unit TA 01-10 321 Bullfinch Rd. NEDU TR 05-05 Panama City, FL 32407-7015 April 2005 PULMONARY FUNCTION AFTER OXYGEN-ACCELERATED DECOMPRESSIONS FROM REPETITIVE SUB-SATURATION AIR
transients' of large amplitude can be imposed on the arterial, cardiac and Since both coughing and the Valsalva manoeuvre raise intrathoracic pressure
 351 J. Physiol. (I953) I22, 35I-357 EFFECTS OF COUGHING ON INTRATHORACIC PRESSURE, ARTERIAL PRESSURE AND PERIPHERAL BLOOD FLOW BY E. P. SHARPEY-SCHAFER From the Department of Medicine, St Thomas's Hospital
351 J. Physiol. (I953) I22, 35I-357 EFFECTS OF COUGHING ON INTRATHORACIC PRESSURE, ARTERIAL PRESSURE AND PERIPHERAL BLOOD FLOW BY E. P. SHARPEY-SCHAFER From the Department of Medicine, St Thomas's Hospital
Physiology of Respiration
 Physiology of Respiration External Respiration = pulmonary ventilation breathing involves 2 processes: inspiration expiration Inspiration an active process involves contraction of diaphragm innervated
Physiology of Respiration External Respiration = pulmonary ventilation breathing involves 2 processes: inspiration expiration Inspiration an active process involves contraction of diaphragm innervated
Respiratory Lecture Test Questions Set 3
 Respiratory Lecture Test Questions Set 3 1. The pressure of a gas: a. is inversely proportional to its volume b. is unaffected by temperature changes c. is directly proportional to its volume d. does not
Respiratory Lecture Test Questions Set 3 1. The pressure of a gas: a. is inversely proportional to its volume b. is unaffected by temperature changes c. is directly proportional to its volume d. does not
Department of Biology Work Sheet Respiratory system,9 class
 I. Name the following : Department of Biology Work Sheet Respiratory system,9 class 1. A muscular sheet separating the thoracic and abdominal cavities. 2. A respiratory tube supported by cartilaginous
I. Name the following : Department of Biology Work Sheet Respiratory system,9 class 1. A muscular sheet separating the thoracic and abdominal cavities. 2. A respiratory tube supported by cartilaginous
Gas Exchange Respiratory Systems
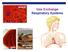 alveoli gills Gas Exchange Respiratory Systems elephant seals 2008-2009 Why do we need a respiratory system? respiration for respiration Need O 2 in for aerobic cellular respiration make ATP Need CO 2
alveoli gills Gas Exchange Respiratory Systems elephant seals 2008-2009 Why do we need a respiratory system? respiration for respiration Need O 2 in for aerobic cellular respiration make ATP Need CO 2
Specifications...Easy CLIA Classification: Sample Type: Blood gas analyzer operation has never been simpler Sample Size: Measured Parameters PCO2
 medicacorp.com easy inside and out With EasyBloodGas, Medica has redefined blood gas analyzer design The sophistication of traditional blood gas analyzers has been packaged in a new compact format with
medicacorp.com easy inside and out With EasyBloodGas, Medica has redefined blood gas analyzer design The sophistication of traditional blood gas analyzers has been packaged in a new compact format with
Respiratory System. Part 2
 Respiratory System Part 2 Respiration Exchange of gases between air and body cells Three steps 1. Ventilation 2. External respiration 3. Internal respiration Ventilation Pulmonary ventilation consists
Respiratory System Part 2 Respiration Exchange of gases between air and body cells Three steps 1. Ventilation 2. External respiration 3. Internal respiration Ventilation Pulmonary ventilation consists
RESPIRATORY GAS EXCHANGE
 RESPIRATORY GAS EXCHANGE Alveolar PO 2 = 105 mmhg; Pulmonary artery PO 2 = 40 mmhg PO 2 gradient across respiratory membrane 65 mmhg (105 mmhg 40 mmhg) Results in pulmonary vein PO 2 ~100 mmhg Partial
RESPIRATORY GAS EXCHANGE Alveolar PO 2 = 105 mmhg; Pulmonary artery PO 2 = 40 mmhg PO 2 gradient across respiratory membrane 65 mmhg (105 mmhg 40 mmhg) Results in pulmonary vein PO 2 ~100 mmhg Partial
Alveolus and Respiratory Membrane
 Alveolus and Respiratory Membrane thin membrane where gas exchange occurs in the lungs, simple squamous epithelium (Squamous cells have the appearance of thin, flat plates. They fit closely together in
Alveolus and Respiratory Membrane thin membrane where gas exchange occurs in the lungs, simple squamous epithelium (Squamous cells have the appearance of thin, flat plates. They fit closely together in
Hypoxia Following Rapid Decompression to 18,288 m (60,000 ft) Attributable to Alveolar Hypoventilation
 Hypoxia Following Rapid Decompression to 18,288 m (60,000 ft) Attributable to Alveolar Hypoventilation Desmond M Connolly PhD QinetiQ Aircrew Systems Senior Medical Officer Timothy J D Oyly BSc Amanda
Hypoxia Following Rapid Decompression to 18,288 m (60,000 ft) Attributable to Alveolar Hypoventilation Desmond M Connolly PhD QinetiQ Aircrew Systems Senior Medical Officer Timothy J D Oyly BSc Amanda
Some major points on the Effects of Hypoxia
 Some major points on the Effects of Hypoxia Source: Kings College London http://www.kcl.ac.uk/teares/gktvc/vc/dental/year1/lectures/rbmsmajorpoints/effectsofhypoxia.htm Cells obtain their energy from oxygen.
Some major points on the Effects of Hypoxia Source: Kings College London http://www.kcl.ac.uk/teares/gktvc/vc/dental/year1/lectures/rbmsmajorpoints/effectsofhypoxia.htm Cells obtain their energy from oxygen.
Submarine Base, Groton, Conn. REPORT NUMBER 690. RESPONSE VARIATIONS OF THREE TYPES OF MICROPHONES PRESSURIZED TO 19 Ata
 'd-jl^ c^/^ * Submarine Base, Groton, Conn. REPORT NUMBER 690 RESPONSE VARIATIONS OF THREE TYPES OF MICROPHONES PRESSURIZED TO 19 Ata by Thomas Murry and Russell L. Sergeant Bureau of Medicine and Surgery,
'd-jl^ c^/^ * Submarine Base, Groton, Conn. REPORT NUMBER 690 RESPONSE VARIATIONS OF THREE TYPES OF MICROPHONES PRESSURIZED TO 19 Ata by Thomas Murry and Russell L. Sergeant Bureau of Medicine and Surgery,
Human gas exchange. Question Paper. Save My Exams! The Home of Revision. Cambridge International Examinations. 56 minutes. Time Allowed: Score: /46
 Human gas exchange Question Paper Level Subject Exam oard Topic Sub Topic ooklet O Level iology ambridge International Examinations Respiration Human gas exchange Question Paper Time llowed: 56 minutes
Human gas exchange Question Paper Level Subject Exam oard Topic Sub Topic ooklet O Level iology ambridge International Examinations Respiration Human gas exchange Question Paper Time llowed: 56 minutes
2. State the volume of air remaining in the lungs after a normal breathing.
 CLASS XI BIOLOGY Breathing And Exchange of Gases 1. Define vital capacity. What is its significance? Answer: Vital Capacity (VC): The maximum volume of air a person can breathe in after a forced expiration.
CLASS XI BIOLOGY Breathing And Exchange of Gases 1. Define vital capacity. What is its significance? Answer: Vital Capacity (VC): The maximum volume of air a person can breathe in after a forced expiration.
2.1.1 List the principal structures of the
 physiology 2.1.1 List the principal structures of the The principle structures of the respiratory are: Nose/Mouth used for inhalation of oxygen-rich air and expelling carbon dioxide rich air Pharynx -
physiology 2.1.1 List the principal structures of the The principle structures of the respiratory are: Nose/Mouth used for inhalation of oxygen-rich air and expelling carbon dioxide rich air Pharynx -
PHTY 300 Wk 1 Lectures
 PHTY 300 Wk 1 Lectures Arterial Blood Gas Components The test provides information on - Acid base balance - Oxygenation - Hemoglobin levels - Electrolyte blood glucose, lactate, renal function When initially
PHTY 300 Wk 1 Lectures Arterial Blood Gas Components The test provides information on - Acid base balance - Oxygenation - Hemoglobin levels - Electrolyte blood glucose, lactate, renal function When initially
Outline - Respiratory System. Function of the respiratory system Parts of the respiratory system Mechanics of breathing Regulation of breathing
 Respiratory system Function Outline - Respiratory System I. II. III. IV. Respiratory System The function of the respiratory system is to bring in oxygen to the body and remove carbon dioxide. Function
Respiratory system Function Outline - Respiratory System I. II. III. IV. Respiratory System The function of the respiratory system is to bring in oxygen to the body and remove carbon dioxide. Function
These two respiratory media (air & water) impose rather different constraints on oxygen uptake:
 Topic 19: OXYGEN UPTAKE AND TRANSPORT (lectures 29-30) OBJECTIVES: 1. Be able to compare air vs. water as a respiratory medium with respect to oxygen content, diffusion coefficient, viscosity and water
Topic 19: OXYGEN UPTAKE AND TRANSPORT (lectures 29-30) OBJECTIVES: 1. Be able to compare air vs. water as a respiratory medium with respect to oxygen content, diffusion coefficient, viscosity and water
Working safely under pressure: Field experience in occupational medicine
 Working safely under pressure: Field experience in occupational medicine Dr. med. Claudia Pletscher Suva, Department of Occupational Medicine Head of Preventative Occupational Medicine Sector Agenda Introduction
Working safely under pressure: Field experience in occupational medicine Dr. med. Claudia Pletscher Suva, Department of Occupational Medicine Head of Preventative Occupational Medicine Sector Agenda Introduction
Respiration (revised 2006) Pulmonary Mechanics
 Respiration (revised 2006) Pulmonary Mechanics PUL 1. Diagram how pleural pressure, alveolar pressure, airflow, and lung volume change during a normal quiet breathing cycle. Identify on the figure the
Respiration (revised 2006) Pulmonary Mechanics PUL 1. Diagram how pleural pressure, alveolar pressure, airflow, and lung volume change during a normal quiet breathing cycle. Identify on the figure the
Chapter 13 The Respiratory System
 Chapter 13 The Respiratory System by Dr. Jay M. Templin Brooks/Cole - Thomson Learning Atmosphere Tissue cell External respiration Alveoli of lungs 1 Ventilation or gas exchange between the atmosphere
Chapter 13 The Respiratory System by Dr. Jay M. Templin Brooks/Cole - Thomson Learning Atmosphere Tissue cell External respiration Alveoli of lungs 1 Ventilation or gas exchange between the atmosphere
CHAPTER 6. Oxygen Transport. Copyright 2008 Thomson Delmar Learning
 CHAPTER 6 Oxygen Transport Normal Blood Gas Value Ranges Table 6-1 OXYGEN TRANSPORT Oxygen Dissolved in the Blood Plasma Dissolve means that the gas maintains its precise molecular structure About.003
CHAPTER 6 Oxygen Transport Normal Blood Gas Value Ranges Table 6-1 OXYGEN TRANSPORT Oxygen Dissolved in the Blood Plasma Dissolve means that the gas maintains its precise molecular structure About.003
UNDERSTANDING A DIVE COMPUTER. by S. Angelini, Ph.D. Mares S.p.A.
 UNDERSTANDING A DIVE COMPUTER by S. Angelini, Ph.D. Mares S.p.A. Dive Computer UNDERSTANDING A DIVE COMPUTER The decompression algorithm in a dive computer is an attempt to replicate the effects of a dive
UNDERSTANDING A DIVE COMPUTER by S. Angelini, Ph.D. Mares S.p.A. Dive Computer UNDERSTANDING A DIVE COMPUTER The decompression algorithm in a dive computer is an attempt to replicate the effects of a dive
I. Gas Exchange Respiratory Surfaces Respiratory Surface:
 I. Gas Exchange Respiratory Surfaces Respiratory Surface: Common characteristics of respiratory surfaces: a) Moist: allows for the RAPID diffusion of dissolved gasses across its surface. Whereas the respiratory
I. Gas Exchange Respiratory Surfaces Respiratory Surface: Common characteristics of respiratory surfaces: a) Moist: allows for the RAPID diffusion of dissolved gasses across its surface. Whereas the respiratory
evidence, too, that such slowing was accompanied by a fall in ph of the
 506 J. Phyaiol. (1960), 152, pp. 506-514 With 3 text-figures Printed sn Great Britain CHANGES IN CARDIAC AND RESPIRATORY FUNCTION, AND IN BLOOD CARBON DIOXIDE PRESSURE AND ph, IN CATS EXPOSED TO OXYGEN
506 J. Phyaiol. (1960), 152, pp. 506-514 With 3 text-figures Printed sn Great Britain CHANGES IN CARDIAC AND RESPIRATORY FUNCTION, AND IN BLOOD CARBON DIOXIDE PRESSURE AND ph, IN CATS EXPOSED TO OXYGEN
