Answer the following questions :
|
|
|
- Oscar Hawkins
- 5 years ago
- Views:
Transcription
1 1
2 Model Answer Exam 1-Cairo Governorate ( ) 1)A) Complete the following statements : 1-oxygen 2-carbon dioxide 3-constant-changeable. 4-brain spinal cord. B) Give reasons for: 1-To keep the nervous system healthy. 2-To prevent return back of mercury to mercury bulb quickly. 2) [A] Choose the correct answer : 1. 1/5 2. wood kg. 4. nitrogen. 5. maintaining the balance of the body. [B] Mention one function for each of the following: 1-To protect the heart and two lungs. 2-To control the involuntary movement such as heart beats. 3-To help the body to move. 3- [A] Write the scientific term. 1. Heat conductors. 2. Thermometer. 3. Nitrogen. 4. Joints. 5.Nerve cell or neuron [B] Mention one use for each of the following : I. plants use carbon dioxide to make food by photosynthesis. 2. To measure the weight of the body. 3.In making thermometer. 4- [A] Put ( ) or( ): [B] Mention one example for each of the following : 1-Skull. 2-Secreting saliva on seeing or smelling good food. Model exam 2-Giza Governorate ( ) 1)A) Complete the following statements : 2
3 1-kilogram- Newton Celisus three oxygen B) Give reasons for: 1- To produce carbon dioxide which expands by heating to make the bread porous and tasty. 2) [A] Choose the correct answer : 1-100grams. 2- nitrogen 3- cerebellum 4- elbow 5- copper 6- oxygen B) Give reasons for: 1- Because it is a liquid metal that can be seen. It is a good conductor of heat and doesn't stick to the walls of the capillary. 3- [A] Put ( ) or( ): [B] Mention one function for each of the following: 1- To protect the heart and two lungs. 2- It is used to measure the weight of any object. 4- [A] Write the scientific term. 1-Heat conductor. 2-spinal cord 3-nerve cell or neuron. 4-Temperature 5-Tendons 6-Nitrogen [B] Put the following labels instead of numbers: 1- Hydrochloric acid. 2- tap. 3-carbon dioxide gas 4 calcium carbonate powder. Model exam 3-Qaliubiya Governorate ( ) 1)A) Complete the following statements : 1-Handle of cooking pots-handle of kettles 2-31 pairs- 12 pairs. 3- Balance- spring. 4- Hydrogen peroxide-manganese dioxide. 2- [A] Write the scientific term: 1-mass 2-joints 3-carbon dioxide 4-Brain. 2-B) Give reasons for: 1- To produce carbon dioxide gas that makes the bread porous and tasty. 2- Because it is good conductor of heat. 3-Because it has constant volume and doesn t change volume by changing temperature. 4-Because it regulates the involuntary processes as heart beats and respiration. 3- [A] What happens to each of the following : 1-The weight decreases. 2-The color doesn t change. 3
4 3-The Earth s temperature increases and harms the climate. 4-It will be harmed. 3- [B] What is meant by : 1- It's the degree of hotness or coldness of a body. 2- It is the involuntary response that the body makes by the nervous system when it is subjected to an external stimulus such as light, heat, etc. 3-The object s mass = 100 grams. 4- [A] Compare between each of the following : 1-Elbow joint is Slightly movable joints and wrist joint is Freely movable joints : 2-The trunk muscle is Voluntary muscles and the gastrointestinal tract muscle is Involuntary muscles: 3-Carbon dioxide gas easily dissolves in water and nitrogen gas is scarcely dissolves in water. 4-Stainless steel is a good conductor of heat and plastic is a bad conductor of heat 4- [A] Choose the correct answer : heavier than air 3- pelvic Model exam 4-Menoufia Governorate ( ) 1)A) Complete the following statements : 1-carbon dioxide 2-weight 3-axial 4-nitrogen. 1)[B] Mention one function for each of the following: 1-It is used to measure mass. 2-Avoid mercury from returning back quickly to the bulb in order to read the temperature. 3- It maintains the balance of the body during movement. 2- [A] Write the scientific term: 1-heat conductors 2-manganses dioxide 3-spinal cord 4-ozone 2- [B] What happens to each of the following : 1-no life on the Earth s surface. 2-The shoulder joints move in one direction. 3- Nail rusts. 3- [A] Choose the correct answer : 1- from hand to ice 2-10pairs 3- all the previous 4- fatty 3-B) Give reasons for: 1- To protect them. 2- Because moon s mass is less than the Earth s mass. 3- To connect the muscle with the bones. 4- [A] Put ( ) or( ):
5 4- [B] I-Compare between : Voluntary muscles They are the muscles that can move willingly and can be controlled. involuntary muscles They are the muscles that can move automatically and you can't control or even be aware of their movements. Examples: Limb muscles Trunk muscles Face muscles Abdominal wall muscles Examples : Gastrointestinal tract Bladder muscles Blood vessels 2- Carbon dioxide gas easily dissolves in water and nitrogen gas is scarcely dissolves in water. II-weight on Earth= mass x 10= 6 x 10 = 60 Newton. Weight on moon =weight on Earth x 1/6 = 60 x 1/6 = 10 Newton. Complete: 1-spring scale. 3-wood and plastic Model Exam(5) 2-manganese dioxide 4-two oxygen-ozone. 5-iron oxide 6-expand-contract. 7-netural. 2-a-Write scientific term: Kilogram. 2-heat conductors 3-constriction. 4-dust and smoke 5-mass. 3-a- Give reason for 1-Because it is a liquid metal that can be seen. It is a good conductor of heat and doesn't stick to the walls of the capillary. 2-Due to photosynthesis process. 3-b- Correct the underlined words: 1-Alcohol 2-Atmosphere 3-c-Mention the use of: Cutting and welding of metals. 4-a-1-Weight on Earth=mass x 10= 3 x 10= 30 Newton Weight on moon=mass x 1/6x10= 3 x 1/6x10= 5 Newton 5
6 5- mass on the moon= 3kg 4-b- Choose: 1-b 2-a 3-c 4-c- Compare: Scale of Celsius thermometer Scale of medical thermometer 0oC to100oc 35oC to42oc Example of burning Burning of cleansing wire Example of oxidation Iron rusting Model exam 6-El-Gharbia Governorate Answer the following questions: 1) [A] Complete the following statements : 1- of the air 2-12 pairs-31 pairs. 3- Balance scale-spring scale. 4- Iron copper. [B] Compare between each of the following: P.O.C. Medical thermometer Celsius thermometer (1) Range of scale 35 0 C-42 0 C 0 o C-100 o C (2) Usage it used to measure the human body temperature. 6 It used to measure the temperature of liquid. 2) [A] Give scientific term for the following : 1-Ozone gas. 2-Oxy-acetylene flame. 3-cartilage. 4-Spinal cord. [B] 1-a-Tasteless,colourless and odorless. b- It doesn t burn and doesn t help in burning. c- It scarcely dissolves in water. 1- a-manganese dioxide. b-hydrogen peroxide c-oxygen gas. 3)[A] 1-Nerve cell. 2-Dendrities 3-Axon terminal. [B] 1-weight 2-White 3-plastic or wood. 4)[A] 1-c 2-d 3-d 4-b [B] 1-Because the gravity of the Earth is more than the gravity of the moon. 2-Because it controls the involuntary movement of the body as heart beats and respiration.
7 Model exam 7-Monifinia Governorate El-Minia Educational Directorate Answer the following questions: 1) Complete the following statements: 1-balance scale-spring scale. 2-iron copper. 3-brain medulla oblongata. 4-the boiling point of water-melting point of ice or freezing point of water. 5-12pairs-33 vertebrae. 2)[A]1-Cerebellum. 2-Nitrogen. 3-Tendons. [B] 1-60Kg 2-60x10=600 Newton. 3)[A] 1-oxygen 2-1-manganese dioxide 2-water 3-oxygen 4-hydrogen peroxide. [B]1-It will produce carbon dioxide gas which expands by heating giving porous and tasty bread. 2-It will harm the locomotory system. 4) 1-Temperature 2-Neuron. 3-Mercury 4-carbondioxide 5-ozone. Model exam 8-Assiut Governorate Complete the following statements : 1-balance scale spring scale pairs -31pairs. 3-wood plastic C-420C 5-hydrogen peroxide manganese dioxide. 2)[A]1-c 2-d 3-d 4-a [B] Refer to the booklet. 3)[A] 1-Thermometer 2-Nitrogen 3-Oxy-acetylene 4-Joints. [B]1-To protect the brain. 2-To measure the temperature of the liquids. Assuat Educational Directorate 4)1-To produce carbon dioxide which expands by heating giving porous and tasty bread. 2-Because it is good conductor of heat. 3-Due to its constancy of the volume of the tires by changing temperature. Model exam 9-Sobag Governorate Educational Directorate Answer the following questions: A-Complete the following statements with suitable words: 1-planet where it exists-object s mass- distance away from the center of earth. 2-wood plastic C-420C. [B] 1-Because it is scarcely dissolves in water. 2-Because it doesn t burn and it doesn t help in burning. 7
8 3-Due to reflex action. 2)[A]1-Copper 2-mercury. 3-helps 4-slightly 5-cell body. [B] Weight = mass x gravity=6x10=60newton. Weight on moon=1/6 x weight on earth.=1/6x60=10 Newton. 3)a- 1-central nervous system. 2-Joints. 3-Heat conductors 4-Balance scale. 5-Ozone gas. b- 1-It will rust. 2-The nervous system will be harmed. 4)a- 1-c 2-a 3-a 4-b b- 1-The main component of the protein. 2-It used to measure the temperature of liquid. 3-To help them to make their food by photosynthesis process. 8
Hyndland Secondary School Biology Department
 Hyndland Secondary School Biology Department Body in Action Homework and Question Booklet 1 Body in Action (a) Movement... 2 Skeleton... 2 Joints... 3 Body in Action (b) The need for Energy... 5 Heart...
Hyndland Secondary School Biology Department Body in Action Homework and Question Booklet 1 Body in Action (a) Movement... 2 Skeleton... 2 Joints... 3 Body in Action (b) The need for Energy... 5 Heart...
Structures of the Respiratory System include:
 Respiratory System Structures of the Respiratory System include: ü Oral Cavity ü Nasal Cavity ü Pharynx ü Epiglottis ü Larynx ü Trachea ü Diaphragm ü Lung ü Bronchus ü Bronchioles ü Alveolus ü Pulmonary
Respiratory System Structures of the Respiratory System include: ü Oral Cavity ü Nasal Cavity ü Pharynx ü Epiglottis ü Larynx ü Trachea ü Diaphragm ü Lung ü Bronchus ü Bronchioles ü Alveolus ü Pulmonary
S.T.E.M Part 1: Prior Knowledge: Energy and Simple Machines
 S.T.E.M. 2016-7 Part 1: Prior Knowledge: Energy and Simple Machines Prior Knowledge: What is Energy? Redefining: energy, force, work and pressure Experiment 1: Floating Ping Pong Ball You need: - Ping
S.T.E.M. 2016-7 Part 1: Prior Knowledge: Energy and Simple Machines Prior Knowledge: What is Energy? Redefining: energy, force, work and pressure Experiment 1: Floating Ping Pong Ball You need: - Ping
Outline - Respiratory System. Function of the respiratory system Parts of the respiratory system Mechanics of breathing Regulation of breathing
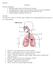
 Respiratory system Function Outline - Respiratory System I. II. III. IV. Respiratory System The function of the respiratory system is to bring in oxygen to the body and remove carbon dioxide. Function
Respiratory system Function Outline - Respiratory System I. II. III. IV. Respiratory System The function of the respiratory system is to bring in oxygen to the body and remove carbon dioxide. Function
Slide 1 of 64. End Show Copyright Pearson Prentice Hall. End Show Copyright Pearson Prentice Hall. Respiration. Slide 5 of 64
 33-3 Form and Function in Chordates Chordates Vertebrate organ systems exhibit a wide range of complexity. This is seen in the different ways that vertebrates feed, breathe, respond, move, and reproduce.
33-3 Form and Function in Chordates Chordates Vertebrate organ systems exhibit a wide range of complexity. This is seen in the different ways that vertebrates feed, breathe, respond, move, and reproduce.
Breathing: The normal rate is about 14 to 20 times a minute. Taking in of air is called Inspiration and the forcing out of air is called Expiration.
 Biology 12 Respiration Divisions of Respiration Breathing: entrance and exit of air into and out of the lungs External Respiration: exchange of gases(o2 and CO2) between air (in alveoli) and blood Internal
Biology 12 Respiration Divisions of Respiration Breathing: entrance and exit of air into and out of the lungs External Respiration: exchange of gases(o2 and CO2) between air (in alveoli) and blood Internal
practices makes perfect
 Human Respiratory system Chapter 1: Respiration PMR 03, 08 1. The human respiratory system consists of: a... b... c.. d... e. Our body has.. lungs (right & left). f. Each lung consists of the, and alveoli.
Human Respiratory system Chapter 1: Respiration PMR 03, 08 1. The human respiratory system consists of: a... b... c.. d... e. Our body has.. lungs (right & left). f. Each lung consists of the, and alveoli.
Human Biology Respiratory System
 Human Biology Respiratory System Respiratory System Responsible for process of breathing Works in cooperation with Circulatory system Three types: 1. Internal Respiration 2. External Respiration 3. Cellular
Human Biology Respiratory System Respiratory System Responsible for process of breathing Works in cooperation with Circulatory system Three types: 1. Internal Respiration 2. External Respiration 3. Cellular
GASEOUS EXCHANGE IN HUMANS 06 AUGUST 2014
 GASEOUS EXCHANGE IN HUMANS 06 AUGUST 2014 In this lesson we: Lesson Description Look at gaseous exchange in humans in terms of o Ventilation o Inspiration o Expiration o Transport of gases o Homeostatic
GASEOUS EXCHANGE IN HUMANS 06 AUGUST 2014 In this lesson we: Lesson Description Look at gaseous exchange in humans in terms of o Ventilation o Inspiration o Expiration o Transport of gases o Homeostatic
THE STUDY OF LUNG VELOCITY UNDER DIFFERENT CONDITIONS
 THE STUDY OF LUNG VELOCITY UNDER DIFFERENT CONDITIONS Natasha Chandra Cary Academy ABSTRACT The purpose of this study was to determine which activity made each student have the least lung velocity (L/sec).
THE STUDY OF LUNG VELOCITY UNDER DIFFERENT CONDITIONS Natasha Chandra Cary Academy ABSTRACT The purpose of this study was to determine which activity made each student have the least lung velocity (L/sec).
Glucose + Oxygen Carbon Dioxide + Water + Energy C6H12O CO2 + 6H20 + energy
 Cell Respiration - A food oxidization process in body cells to produce energy - Occurs (happens) in animal cells and plant cells Notes : Metabolic Reactions in Gaseous Exchange CELL RESPIRATION Photosynthesis
Cell Respiration - A food oxidization process in body cells to produce energy - Occurs (happens) in animal cells and plant cells Notes : Metabolic Reactions in Gaseous Exchange CELL RESPIRATION Photosynthesis
Respiration. The ins and outs
 Respiration The ins and outs Functions 1. To bring O 2 into the body and transfer it to the blood stream 2. To remove CO 2 Circulation and respiration work together to achieve these functions Why Do We
Respiration The ins and outs Functions 1. To bring O 2 into the body and transfer it to the blood stream 2. To remove CO 2 Circulation and respiration work together to achieve these functions Why Do We
Biology. Slide 1 of 53. End Show. Copyright Pearson Prentice Hall
 Biology 1 of 53 Chapter 33 Comparing Chordates 2 of 53 This chapter is a good revision of the material we saw during Unit III. 3 of 53 4 of 53 Controlling Body Temperature The control of body temperature
Biology 1 of 53 Chapter 33 Comparing Chordates 2 of 53 This chapter is a good revision of the material we saw during Unit III. 3 of 53 4 of 53 Controlling Body Temperature The control of body temperature
year 6 REVISION End of Year
 year 6 REVISION End of Year The Science Department This Revision Guide can be utilised at home or on your ipad. This electronic book has been made as an additional aid in the revision process. The material
year 6 REVISION End of Year The Science Department This Revision Guide can be utilised at home or on your ipad. This electronic book has been made as an additional aid in the revision process. The material
A breath of fresh air
 A breath of fresh air BIG IDEAS You are learning to: Describe the structure of the lungs Explain how the lungs clean themselves Interpret patterns in data Where are the lungs and what do they do? The lungs
A breath of fresh air BIG IDEAS You are learning to: Describe the structure of the lungs Explain how the lungs clean themselves Interpret patterns in data Where are the lungs and what do they do? The lungs
1. Harry investigated the effects of fizzy cola drink on his heart rate.
 1. Harry investigated the effects of fizzy cola drink on his heart rate. First he measured his heart rate every minute for 5 minutes when sitting down. Then he drank some cola. He continued to measure
1. Harry investigated the effects of fizzy cola drink on his heart rate. First he measured his heart rate every minute for 5 minutes when sitting down. Then he drank some cola. He continued to measure
Chapter 16 Respiratory System
 Introduction Chapter 16 Respiratory System The respiratory system consists of tubes that filter incoming air and transport it to alveoli where gases are exchanged. Think pair share: what organs are associated
Introduction Chapter 16 Respiratory System The respiratory system consists of tubes that filter incoming air and transport it to alveoli where gases are exchanged. Think pair share: what organs are associated
Respiration. The resspiratory system
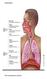 Respiration The resspiratory system The Alveoli The lungs have about 300 million alveoli, with a total crosssec onal area of 50 70 m2.. Each alveolar sac is surrounded by blood capillaries. The walls of
Respiration The resspiratory system The Alveoli The lungs have about 300 million alveoli, with a total crosssec onal area of 50 70 m2.. Each alveolar sac is surrounded by blood capillaries. The walls of
Welding. Module
 Welding Module 19.2.4 Module 19.2.4 Describe the oxy fuel gas welding process. Process: setting up the equipment, leak testing, oxy-fuel gas mixing, flame adjustment (including oxidizing, neutral and carburizing
Welding Module 19.2.4 Module 19.2.4 Describe the oxy fuel gas welding process. Process: setting up the equipment, leak testing, oxy-fuel gas mixing, flame adjustment (including oxidizing, neutral and carburizing
Gas Laws Chapter 14. Complete the following pressure conversion. Be sure to show how units cancel.
 Gas Laws Chapter 14 Complete the following pressure conversion. Be sure to show how units cancel. 1 atm = 760 mm Hg = 760 torr = 101.3 kpa = 14.7 psi = 1.013 bar 1. The air pressure for a certain tire
Gas Laws Chapter 14 Complete the following pressure conversion. Be sure to show how units cancel. 1 atm = 760 mm Hg = 760 torr = 101.3 kpa = 14.7 psi = 1.013 bar 1. The air pressure for a certain tire
(Slide 1) Lecture Notes: Respiratory System
 (Slide 1) Lecture Notes: Respiratory System I. (Slide 2) The Respiratory Tract A) Major structures and regions of the respiratory Tract/Route INTO body 1) nose 2) nasal cavity 3) pharynx 4) glottis 5)
(Slide 1) Lecture Notes: Respiratory System I. (Slide 2) The Respiratory Tract A) Major structures and regions of the respiratory Tract/Route INTO body 1) nose 2) nasal cavity 3) pharynx 4) glottis 5)
Unit 2 Kinetic Theory, Heat, and Thermodynamics: 2.A.1 Problems Temperature and Heat Sections of your book.
 Unit 2 Kinetic Theory, Heat, and Thermodynamics: 2.A.1 Problems Temperature and Heat Sections 10.1 10.2 of your book. Convert the following to Celsius and Kelvin temperatures: 1. 80.0 o F Early E. C.:
Unit 2 Kinetic Theory, Heat, and Thermodynamics: 2.A.1 Problems Temperature and Heat Sections 10.1 10.2 of your book. Convert the following to Celsius and Kelvin temperatures: 1. 80.0 o F Early E. C.:
1 MS Earth s Atmosphere
 CHAPTER 1 MS Earth s Atmosphere Chapter Outline 1.1 THE ATMOSPHERE 1.2 ENERGY IN THE ATMOSPHERE 1.3 LAYERS OF THE ATMOSPHERE 1.4 AIR MOVEMENT 1.5 REFERENCES Did you ever see such an awesome sight? This
CHAPTER 1 MS Earth s Atmosphere Chapter Outline 1.1 THE ATMOSPHERE 1.2 ENERGY IN THE ATMOSPHERE 1.3 LAYERS OF THE ATMOSPHERE 1.4 AIR MOVEMENT 1.5 REFERENCES Did you ever see such an awesome sight? This
2/28/18. Respiratory System. 1 Copyright 2016 by Elsevier Inc. All rights reserved. Introduction. Anatomy. Physiology. Respiratory System
 Introduction Respiratory System Chapter 28 Respiration: We inhale air, extract oxygen from it, exhale air Cardiovascular and respiratory systems work together Failure of either system: - Disruption of
Introduction Respiratory System Chapter 28 Respiration: We inhale air, extract oxygen from it, exhale air Cardiovascular and respiratory systems work together Failure of either system: - Disruption of
Nervous System: Reaction Time Student Version
 Nervous System: Reaction Time Student Version Key Concepts: Your nervous system allows your body to react to different stimuli (external events) Reactions can be voluntary (eg: swinging a bat at a ball
Nervous System: Reaction Time Student Version Key Concepts: Your nervous system allows your body to react to different stimuli (external events) Reactions can be voluntary (eg: swinging a bat at a ball
Lesson 9.1: The Importance of an Organ Delivery System
 Lesson 9.1: The Importance of an Organ Delivery System Animals require a continuous supply of oxygen (O 2 ) for cellular respiration, and they must expel carbon dioxide (CO 2 ), the waste product of this
Lesson 9.1: The Importance of an Organ Delivery System Animals require a continuous supply of oxygen (O 2 ) for cellular respiration, and they must expel carbon dioxide (CO 2 ), the waste product of this
REVISION: GASEOUS EXCHANGE 24 SEPTEMBER 2014 Lesson Description
 REVISION: GASEOUS EXCHANGE 24 SEPTEMBER 2014 Lesson Description In this lesson, we revise: Gaseous Exchange in Plants & Animals Gaseous Exchange in Humans Excretion in Humans Focus on the Kidney Gaseous
REVISION: GASEOUS EXCHANGE 24 SEPTEMBER 2014 Lesson Description In this lesson, we revise: Gaseous Exchange in Plants & Animals Gaseous Exchange in Humans Excretion in Humans Focus on the Kidney Gaseous
Mammalian systems. Chapter 3 Pages
 Mammalian systems Chapter 3 Pages 75-103 Learning intentions To know that multicellular organisms exist from specialized cells To know how the respiratory system is specialized and organized and how a
Mammalian systems Chapter 3 Pages 75-103 Learning intentions To know that multicellular organisms exist from specialized cells To know how the respiratory system is specialized and organized and how a
PREPARATION AND PROPERTIES OF ATMOSPHERIC GASES I: Carbon Dioxide and Oxygen
 PREPARATION AND PROPERTIES OF ATMOSPHERIC GASES I: Carbon Dioxide and Oxygen.... :O = C = O:.... :O = O: INTRODUCTION The atmosphere consists predominantly of three gases -- nitrogen (N 2 ) 78%, oxygen
PREPARATION AND PROPERTIES OF ATMOSPHERIC GASES I: Carbon Dioxide and Oxygen.... :O = C = O:.... :O = O: INTRODUCTION The atmosphere consists predominantly of three gases -- nitrogen (N 2 ) 78%, oxygen
Noxious Fumes and Gases
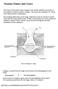 Noxious Fumes and Gases The tissues of the body require oxygen 0 2 for normal metabolic processes (ie. the oxidation of food to produce energy). They must also eliminate CO, which is the waste product
Noxious Fumes and Gases The tissues of the body require oxygen 0 2 for normal metabolic processes (ie. the oxidation of food to produce energy). They must also eliminate CO, which is the waste product
Internal Anatomy of Fish
 Internal Anatomy of Fish The Systems of a Fish Skeletal System Muscular System Respiratory System Digestive System Circulatory System Nervous System Reproductive System Special Organs Skeletal System
Internal Anatomy of Fish The Systems of a Fish Skeletal System Muscular System Respiratory System Digestive System Circulatory System Nervous System Reproductive System Special Organs Skeletal System
Honors Chemistry - Problem Set Chapter 13 Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT.
 Honors Chemistry - Problem Set Chapter 13 Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT. 1. Atmospheric pressure is 760 mm Hg. 2. The SI unit of pressure is
Honors Chemistry - Problem Set Chapter 13 Classify each of these statements as always true, AT; sometimes true, ST; or never true, NT. 1. Atmospheric pressure is 760 mm Hg. 2. The SI unit of pressure is
ExamLearn.ie. The Air & Oxygen
 ExamLearn.ie The Air & Oxygen The Air & Oxygen The air is a mixture of gases, which forms a blanket around the earth. Another name for the air is the atmosphere. *To investigate the percentage of oxygen
ExamLearn.ie The Air & Oxygen The Air & Oxygen The air is a mixture of gases, which forms a blanket around the earth. Another name for the air is the atmosphere. *To investigate the percentage of oxygen
Respiratory System 1
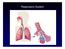 Respiratory System 1 Outline Respiratory structures Gills Air-Breathing Animals Amphibians and Reptiles Mammals Birds Structures and Mechanisms of Breathing 2 Copyright The McGraw-Hill Companies, Inc.
Respiratory System 1 Outline Respiratory structures Gills Air-Breathing Animals Amphibians and Reptiles Mammals Birds Structures and Mechanisms of Breathing 2 Copyright The McGraw-Hill Companies, Inc.
P215 Respiratory System, Part 2
 P15 Respiratory System, Part Gas Exchange Oxygen and Carbon Dioxide constant need for oxygen constant production of carbon dioxide exchange (and movement) lung alveoli pulmonary arteries pulmonary capillaries
P15 Respiratory System, Part Gas Exchange Oxygen and Carbon Dioxide constant need for oxygen constant production of carbon dioxide exchange (and movement) lung alveoli pulmonary arteries pulmonary capillaries
The equation describes anaerobic respiration in muscle cells. How can you tell from the equation that this process is anaerobic?
 Respiration. Name.. Q.Anaerobic respiration happens in muscle cells and yeast cells. The equation describes anaerobic respiration in muscle cells. glucose lactic acid (a) How can you tell from the equation
Respiration. Name.. Q.Anaerobic respiration happens in muscle cells and yeast cells. The equation describes anaerobic respiration in muscle cells. glucose lactic acid (a) How can you tell from the equation
Chapter 17 The Respiratory System: Gas Exchange and Regulation of Breathing
 Chapter 17 The Respiratory System: Gas Exchange and Regulation of Breathing Overview of Pulmonary Circulation o Diffusion of Gases o Exchange of Oxygen and Carbon Dioxide o Transport of Gases in the Blood
Chapter 17 The Respiratory System: Gas Exchange and Regulation of Breathing Overview of Pulmonary Circulation o Diffusion of Gases o Exchange of Oxygen and Carbon Dioxide o Transport of Gases in the Blood
Name Hour. The Behavior of Gases. Practice B
 Name Hour The Behavior of Gases Practice B B 1 Objective 1: Apply Boyle s Law, Charles s Law, and Gay-Lussac s Law to solve problems involving pressure and volume and temperature. 1. A high-altitude balloon
Name Hour The Behavior of Gases Practice B B 1 Objective 1: Apply Boyle s Law, Charles s Law, and Gay-Lussac s Law to solve problems involving pressure and volume and temperature. 1. A high-altitude balloon
Physiology of Respiration
 Physiology of Respiration External Respiration = pulmonary ventilation breathing involves 2 processes: inspiration expiration Inspiration an active process involves contraction of diaphragm innervated
Physiology of Respiration External Respiration = pulmonary ventilation breathing involves 2 processes: inspiration expiration Inspiration an active process involves contraction of diaphragm innervated
1. Label a diagram of the respiratory system. Objective sheet 3 Notes
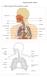 1. Label a diagram of the respiratory system Objective sheet 3 Notes 2. Functions of the respiratory structures Name Description Function Nasal Cavity Trachea Bronchi (Singular Bronchus) Bronchioles Lungs
1. Label a diagram of the respiratory system Objective sheet 3 Notes 2. Functions of the respiratory structures Name Description Function Nasal Cavity Trachea Bronchi (Singular Bronchus) Bronchioles Lungs
2. Pressure Conversions (Add to your Conversion Sheet
 The Gas Law Reference Sheet 1. The Kelvin Temperature Scale Degrees Kelvin = C + 273 Convert to K 1) 27 C 2) 0. C 3) 48 C 4) 16 C 5) 106 C 2. Pressure Conversions (Add to your Conversion Sheet 1 mm Hg
The Gas Law Reference Sheet 1. The Kelvin Temperature Scale Degrees Kelvin = C + 273 Convert to K 1) 27 C 2) 0. C 3) 48 C 4) 16 C 5) 106 C 2. Pressure Conversions (Add to your Conversion Sheet 1 mm Hg
14A. Neuromuscular Reflexes. Experiment
 Experiment 14A The automatic response of a muscle to a stimulus is called a reflex. The patellar reflex results from tapping the patellar tendon below the knee with a reflex hammer. This causes contraction
Experiment 14A The automatic response of a muscle to a stimulus is called a reflex. The patellar reflex results from tapping the patellar tendon below the knee with a reflex hammer. This causes contraction
FISH ANATOMY DIAGRAM AND QUESTIONS
 Name Block FISH ANATOMY DIAGRAM AND QUESTIONS External: 1. What percentage of fish are bony fish? 2. What is the operculum s function? 3. The nostrils are used for, not. 4. Which fins keeps the fish level
Name Block FISH ANATOMY DIAGRAM AND QUESTIONS External: 1. What percentage of fish are bony fish? 2. What is the operculum s function? 3. The nostrils are used for, not. 4. Which fins keeps the fish level
Respiratory Lecture Test Questions Set 3
 Respiratory Lecture Test Questions Set 3 1. The pressure of a gas: a. is inversely proportional to its volume b. is unaffected by temperature changes c. is directly proportional to its volume d. does not
Respiratory Lecture Test Questions Set 3 1. The pressure of a gas: a. is inversely proportional to its volume b. is unaffected by temperature changes c. is directly proportional to its volume d. does not
Respiratory System Lab
 Respiratory System Lab Note: Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin. Take time to organize the materials you will need and set
Respiratory System Lab Note: Review the safety materials and wear goggles when working with chemicals. Read the entire exercise before you begin. Take time to organize the materials you will need and set
Neuromuscular Reflexes
 Name: Neuromuscular Reflexes Experiment 14A The automatic response of a muscle to a stimulus is called a reflex. The patellar reflex results from tapping the patellar tendon below the knee with a reflex
Name: Neuromuscular Reflexes Experiment 14A The automatic response of a muscle to a stimulus is called a reflex. The patellar reflex results from tapping the patellar tendon below the knee with a reflex
T H E H U M A N B R A I N
 1 W O R D S allow = let area = place balance =to spread your weight so that you do not fall breathe = to take air into your lungs and send it out again cell = the smallest part of a living thing that can
1 W O R D S allow = let area = place balance =to spread your weight so that you do not fall breathe = to take air into your lungs and send it out again cell = the smallest part of a living thing that can
Respiration. Chapter 39
 Respiration Chapter 39 Impacts, Issues Up in Smoke Smoking immobilizes ciliated cells and kills white blood cells that defend the respiratory system; highly addictive nicotine discourages quitting 39.1
Respiration Chapter 39 Impacts, Issues Up in Smoke Smoking immobilizes ciliated cells and kills white blood cells that defend the respiratory system; highly addictive nicotine discourages quitting 39.1
Experiment 18 Properties of Gases
 Experiment 18 Properties of Gases E18-1 E18-2 The Task In this experiment you will investigate some of the properties of gases, i.e. how gases flow, their phase changes and chemical reactivity. Skills
Experiment 18 Properties of Gases E18-1 E18-2 The Task In this experiment you will investigate some of the properties of gases, i.e. how gases flow, their phase changes and chemical reactivity. Skills
The grade 6 English science unit, Gases, meets the academic content standards set in the Korean curriculum, which state students should:
 This area deals with the properties of gases as small collections of particles. Different kinds of intangible and invisible gases attract students curiosity and promote their use of reasoning skills. Students
This area deals with the properties of gases as small collections of particles. Different kinds of intangible and invisible gases attract students curiosity and promote their use of reasoning skills. Students
Chapter 1: Respiration
 Chapter 1: Respiration Respiration Human Breathing Mechanism Transport of oxygen Importance of a healthy respiratory system Respiratory system lungs inhalation exhalation Diffusion of oxygen by blood Transport
Chapter 1: Respiration Respiration Human Breathing Mechanism Transport of oxygen Importance of a healthy respiratory system Respiratory system lungs inhalation exhalation Diffusion of oxygen by blood Transport
4/26/16. Section 1 Understanding Solutions. Solutions and Suspensions. Solutions and Suspensions. Solutions and Suspensions. Solvents and Solutes
 Section 1 Understanding Solutions On a hot day, you may think that a cool glass of plain water would be refreshing. However, if the glass were actually filled with plain water, it would taste stale. This
Section 1 Understanding Solutions On a hot day, you may think that a cool glass of plain water would be refreshing. However, if the glass were actually filled with plain water, it would taste stale. This
Active Chemical Pty Ltd
 MATERIAL SAFETY DATA SHEET Product: WALLTECH - M Company Details Supplier: Active Chemicals Australia Address: 4/20 Powdrill Road Telephone No.: (02) 9826 0201 Facsimile No.: (02) 9826 0208 Prestons NSW
MATERIAL SAFETY DATA SHEET Product: WALLTECH - M Company Details Supplier: Active Chemicals Australia Address: 4/20 Powdrill Road Telephone No.: (02) 9826 0201 Facsimile No.: (02) 9826 0208 Prestons NSW
The table shows the effect of exercise on the action of one person s heart. Heart rate in beats per minute
 ## (a) The table shows the effect of exercise on the action of one person s heart. At rest During exercise Heart rate in beats per minute 7 165 Volume of blood leaving the heart in each beat in cm 3 Heart
## (a) The table shows the effect of exercise on the action of one person s heart. At rest During exercise Heart rate in beats per minute 7 165 Volume of blood leaving the heart in each beat in cm 3 Heart
Topic 13: Gas Exchange Ch. 42. Gas Exchange pp Gas Exchange. Gas Exchange in Fish pp Gas Exchange in Fish
 Topic 13: Gas Exchange Ch. 42 Fig. 42.24 Gas Exchange pp.979-989 Gas exchange involves the uptake of oxygen and the discharge of carbon dioxide (i.e. respiration or breathing). It is necessary for cellular
Topic 13: Gas Exchange Ch. 42 Fig. 42.24 Gas Exchange pp.979-989 Gas exchange involves the uptake of oxygen and the discharge of carbon dioxide (i.e. respiration or breathing). It is necessary for cellular
GASEOUS EXCHANGE 17 JULY 2013
 GASEOUS EXCHANGE 17 JULY 2013 Lesson Description In this lesson we: Discuss what is gaseous exchange? Consider requirements of an efficient gaseous exchange surface. Look at diversity in gas exchange systems.
GASEOUS EXCHANGE 17 JULY 2013 Lesson Description In this lesson we: Discuss what is gaseous exchange? Consider requirements of an efficient gaseous exchange surface. Look at diversity in gas exchange systems.
It is a product of proteins broken down in the mammal. It is exchanged for oxygen which is taken into the blood.
 5 The table shows the approximate composition of air breathed out by a mammal. Where does the nitrogen in the air breathed out come from? It is a product of proteins broken down in the mammal. It is a
5 The table shows the approximate composition of air breathed out by a mammal. Where does the nitrogen in the air breathed out come from? It is a product of proteins broken down in the mammal. It is a
Multicellular Organisms. Sub-Topic 2.7 Animal Transport & Exchange Systems
 Multicellular Organisms Sub-Topic 2.7 Animal Transport & Exchange Systems On completion of this sub-topic I will be able to state that: Rings of cartilage keep the main airways open Oxygen and carbon dioxide
Multicellular Organisms Sub-Topic 2.7 Animal Transport & Exchange Systems On completion of this sub-topic I will be able to state that: Rings of cartilage keep the main airways open Oxygen and carbon dioxide
Exercise 1/Microscopy Dr. Marvin E. Holtz Human Anatomy and Physiology Lab I
 Exercise 1/Microscopy Parts of the Microscope (page 1) Each student will locate, identify and familiarize themselves with all of the parts and associated functions for each microscope part listed in the
Exercise 1/Microscopy Parts of the Microscope (page 1) Each student will locate, identify and familiarize themselves with all of the parts and associated functions for each microscope part listed in the
(A) The partial pressure in the lungs is higher than in the blood, and oxygen diffuses out of the lungs passively.
 DAT Biology - Problem Drill 12: The Respiratory System Question No. 1 of 10 1. Which statement about the partial pressure of oxygen inside the lungs is correct? Question #01 (A) The partial pressure in
DAT Biology - Problem Drill 12: The Respiratory System Question No. 1 of 10 1. Which statement about the partial pressure of oxygen inside the lungs is correct? Question #01 (A) The partial pressure in
PARTS AND STRUCTURE OF THE RESPIRATORY SYSTEM
 PARTS AND STRUCTURE OF THE RESPIRATORY SYSTEM Parts of the Respiratory System The RS can be divided into two parts: 1. Respiratory Tract, (path that air follows). Nasal passage Pharynx Larynx Trachea Bronchi,
PARTS AND STRUCTURE OF THE RESPIRATORY SYSTEM Parts of the Respiratory System The RS can be divided into two parts: 1. Respiratory Tract, (path that air follows). Nasal passage Pharynx Larynx Trachea Bronchi,
Funsheet [WHAT IS PRESSURE AND TEMPERATURE] Gu 2015
![Funsheet [WHAT IS PRESSURE AND TEMPERATURE] Gu 2015 Funsheet [WHAT IS PRESSURE AND TEMPERATURE] Gu 2015](/thumbs/93/112589015.jpg) Funsheet 7.0 7.1 [WHAT IS PRESSURE AND TEMPERATURE] Gu 2015 1. Convert the following pressures. a) 101 kpa =? atm b) 55 Torr =? psi c) 60. mmhg =? bar d) 45 Torr =? kpa e) 5 psi =? atm f) 0.0056 atm =?
Funsheet 7.0 7.1 [WHAT IS PRESSURE AND TEMPERATURE] Gu 2015 1. Convert the following pressures. a) 101 kpa =? atm b) 55 Torr =? psi c) 60. mmhg =? bar d) 45 Torr =? kpa e) 5 psi =? atm f) 0.0056 atm =?
The Muscular System. Biology 105 Lecture 12 Chapter 6
 The Muscular System Biology 105 Lecture 12 Chapter 6 Outline I. Characteristics of muscles II. Three types of muscles III. Functions of muscles IV. Structure of skeletal muscles V. Mechanics of muscle
The Muscular System Biology 105 Lecture 12 Chapter 6 Outline I. Characteristics of muscles II. Three types of muscles III. Functions of muscles IV. Structure of skeletal muscles V. Mechanics of muscle
Chapter 1, Lesson 5: Air, It s Really There
 Chapter 1, Lesson 5: Air, It s Really There Key Concepts In a gas, the particles (atoms and molecules) have weak attractions for one another. They are able to move freely past each other with little interaction
Chapter 1, Lesson 5: Air, It s Really There Key Concepts In a gas, the particles (atoms and molecules) have weak attractions for one another. They are able to move freely past each other with little interaction
Directions: The following two questions refer to the diagram below, which shows a group of cells from the respiratory tract.
 Topic 7 Gas exchange in humans Directions: The following two questions refer to the diagram below, which shows a group of cells from the respiratory tract. X Y 1. What is the function of structure X? A.
Topic 7 Gas exchange in humans Directions: The following two questions refer to the diagram below, which shows a group of cells from the respiratory tract. X Y 1. What is the function of structure X? A.
Chapter 1 The Atmosphere
 Chapter 1 The Atmosphere Section 1 - Guiding Questions How is the atmosphere important to living things? What gases are present in Earth s atmosphere? Section 1 - The Air Around You Importance of the Atmosphere
Chapter 1 The Atmosphere Section 1 - Guiding Questions How is the atmosphere important to living things? What gases are present in Earth s atmosphere? Section 1 - The Air Around You Importance of the Atmosphere
Exam Review Mass, Weight, Density, Buoyancy, States of Matter
 Exam Review Mass, Weight, Density, Buoyancy, States of Matter Volume An object s volume is the amount of space it takes up. The volume of a cup of water can change if you freeze it in to a solid or boil
Exam Review Mass, Weight, Density, Buoyancy, States of Matter Volume An object s volume is the amount of space it takes up. The volume of a cup of water can change if you freeze it in to a solid or boil
Then the partial pressure of oxygen is x 760 = 160 mm Hg
 1 AP Biology March 2008 Respiration Chapter 42 Gas exchange occurs across specialized respiratory surfaces. 1) Gas exchange: the uptake of molecular oxygen (O2) from the environment and the discharge of
1 AP Biology March 2008 Respiration Chapter 42 Gas exchange occurs across specialized respiratory surfaces. 1) Gas exchange: the uptake of molecular oxygen (O2) from the environment and the discharge of
The content of your gas cylinders. Pocket Safety Guide No. 1
 The content of your gas cylinders Pocket Safety Guide No. 1 Dear Gas User, Messer produces and supplies a broad portfolio of gases. Gases are safe to handle so long as you respect their particular properties.
The content of your gas cylinders Pocket Safety Guide No. 1 Dear Gas User, Messer produces and supplies a broad portfolio of gases. Gases are safe to handle so long as you respect their particular properties.
1. Safety glasses are to be worn at all times in the laboratory except in the study area adjacent to the lab.
 SAFETY RULES page 1 General Precautions 1. Safety glasses are to be worn at all times in the laboratory except in the study area adjacent to the lab. 2. No horseplay. This includes running, throwing of
SAFETY RULES page 1 General Precautions 1. Safety glasses are to be worn at all times in the laboratory except in the study area adjacent to the lab. 2. No horseplay. This includes running, throwing of
Respiratory Pulmonary Ventilation
 Respiratory Pulmonary Ventilation Pulmonary Ventilation Pulmonary ventilation is the act of breathing and the first step in the respiratory process. Pulmonary ventilation brings in air with a new supply
Respiratory Pulmonary Ventilation Pulmonary Ventilation Pulmonary ventilation is the act of breathing and the first step in the respiratory process. Pulmonary ventilation brings in air with a new supply
Chapter 22 Gas Exchange
 Chapter 22 Gas Exchange PowerPoint Lectures for Biology: Concepts & Connections, Sixth Edition Campbell, Reece, Taylor, Simon, and Dickey Lecture by Edward J. Zalisko Introduction: Surviving in Thin Air
Chapter 22 Gas Exchange PowerPoint Lectures for Biology: Concepts & Connections, Sixth Edition Campbell, Reece, Taylor, Simon, and Dickey Lecture by Edward J. Zalisko Introduction: Surviving in Thin Air
Date: Period: Gas Laws Worksheet #1 - Boyle s, Charles, Gay-Lussac s, and Combined Gas Law
 Name: Date: Period: Gas Laws Worksheet #1 - Boyle s, Charles, Gay-Lussac s, and Combined Gas Law Boyle s Law: V1P1 = V2P2 1. A gas sample contained in a cylinder equipped with a moveable piston occupied
Name: Date: Period: Gas Laws Worksheet #1 - Boyle s, Charles, Gay-Lussac s, and Combined Gas Law Boyle s Law: V1P1 = V2P2 1. A gas sample contained in a cylinder equipped with a moveable piston occupied
 Question 1: Define vital capacity. What is its significance? Vital capacity is the maximum volume of air that can be exhaled after a maximum inspiration. It is about 3.5 4.5 litres in the human body. It
Question 1: Define vital capacity. What is its significance? Vital capacity is the maximum volume of air that can be exhaled after a maximum inspiration. It is about 3.5 4.5 litres in the human body. It
Respiratory System Homework
 Respiratory System Homework The R S is the body s breathing equipment. Similar to the D system, it takes S from outside the body (G, particularly O ), circulates them through the body to C and T, then
Respiratory System Homework The R S is the body s breathing equipment. Similar to the D system, it takes S from outside the body (G, particularly O ), circulates them through the body to C and T, then
A. What are the three states of matter chemists work with?
 Chapter 10 and 12 The Behavior of Gases Chapter 10 The States of Matter A. What are the three states of matter chemists work with? Section 10.1 Pg 267 B. We will explain the behavior of gases using the
Chapter 10 and 12 The Behavior of Gases Chapter 10 The States of Matter A. What are the three states of matter chemists work with? Section 10.1 Pg 267 B. We will explain the behavior of gases using the
SCIENCE 8 RESPIRATION
 SCIENCE 8 RESPIRATION WHEN WE BREATHE, WHAT DO WE BREATHE IN? O2, N2, CO2, O3, NO2 (gas fumes), CO, pollutant, fragrants, toxins, etc. WHAT IS THE PURPOSE OF BREATHING IN? WHAT DOES OUR BODY WANT? O2 WHY?
SCIENCE 8 RESPIRATION WHEN WE BREATHE, WHAT DO WE BREATHE IN? O2, N2, CO2, O3, NO2 (gas fumes), CO, pollutant, fragrants, toxins, etc. WHAT IS THE PURPOSE OF BREATHING IN? WHAT DOES OUR BODY WANT? O2 WHY?
Temperature Temperature
 Temperature Temperature is a measure of how hot or cold an object is compared to another object. indicates that heat flows from the object with a higher temperature to the object with a lower temperature.
Temperature Temperature is a measure of how hot or cold an object is compared to another object. indicates that heat flows from the object with a higher temperature to the object with a lower temperature.
1. [Chang7 5.P.013.] Convert 295 mmhg to kpa. kpa Convert 2.0 kpa to mmhg. mmhg
![1. [Chang7 5.P.013.] Convert 295 mmhg to kpa. kpa Convert 2.0 kpa to mmhg. mmhg 1. [Chang7 5.P.013.] Convert 295 mmhg to kpa. kpa Convert 2.0 kpa to mmhg. mmhg](/thumbs/76/74310398.jpg) Score 1. [Chang7 5.P.013.] Convert 295 mmhg to kpa. kpa Convert 2.0 kpa to mmhg. mmhg 2. [Chang7 5.P.019.] The volume of a gas is 5.80 L, measured at 1.00 atm. What is the pressure of the gas in mmhg if
Score 1. [Chang7 5.P.013.] Convert 295 mmhg to kpa. kpa Convert 2.0 kpa to mmhg. mmhg 2. [Chang7 5.P.019.] The volume of a gas is 5.80 L, measured at 1.00 atm. What is the pressure of the gas in mmhg if
BIOLOGY 12: UNIT J - CHAPTER 15 - REVIEW WORKSHEET RESPIRATORY SYSTEM
 BIOLOGY 12: UNIT J - CHAPTER 15 - REVIEW WORKSHEET RESPIRATORY SYSTEM A. CHAPTER REVIEW 1. Define the four components of respiration. 2. What happens to the air as it moves along the air passages? What
BIOLOGY 12: UNIT J - CHAPTER 15 - REVIEW WORKSHEET RESPIRATORY SYSTEM A. CHAPTER REVIEW 1. Define the four components of respiration. 2. What happens to the air as it moves along the air passages? What
OB11 Carry out qualitative tests to compare the carbon dioxide levels of inhaled and exhaled air
 Biology: 5. Respiration and Breathing Please remember to photocopy 4 pages onto one sheet by going A3 A4 and using back to back on the photocopier OB9 Syllabus Describe the process of aerobic respiration
Biology: 5. Respiration and Breathing Please remember to photocopy 4 pages onto one sheet by going A3 A4 and using back to back on the photocopier OB9 Syllabus Describe the process of aerobic respiration
Unit 11 Gas Laws Chapters 13 of your textbook
 Unit 11 Gas Laws Chapters 13 of your textbook Early Booklet E.C.: + 2 Unit 11 Hwk. Pts.: / 19 Unit 11 Lab Pts.: / 20 Late, Incomplete, No Work, No Units Fees? Y / N Learning Targets for Unit 11 1.1 I can
Unit 11 Gas Laws Chapters 13 of your textbook Early Booklet E.C.: + 2 Unit 11 Hwk. Pts.: / 19 Unit 11 Lab Pts.: / 20 Late, Incomplete, No Work, No Units Fees? Y / N Learning Targets for Unit 11 1.1 I can
Kinetic Molecular Theory imaginary Assumptions of Kinetic Molecular Theory: Problems with KMT:
 AP Chemistry Ms. Ye Name Date Block Kinetic Molecular Theory Explains properties of gases, liquids, and solids in terms of energy using an ideal gas, an imaginary which fits all the assumptions of kinetic
AP Chemistry Ms. Ye Name Date Block Kinetic Molecular Theory Explains properties of gases, liquids, and solids in terms of energy using an ideal gas, an imaginary which fits all the assumptions of kinetic
The diagram shows an alveolus next to a blood capillary in a lung. (a) (i) Draw a ring around the correct answer to complete the sentence. diffusion.
 BREATHING / GAS EXCHANGE. NAME. Q.Gas exchange takes place in the lungs. The diagram shows an alveolus next to a blood capillary in a lung. The arrows show the movement of two gases, A and B. (a) (i) Draw
BREATHING / GAS EXCHANGE. NAME. Q.Gas exchange takes place in the lungs. The diagram shows an alveolus next to a blood capillary in a lung. The arrows show the movement of two gases, A and B. (a) (i) Draw
Boyle s Law Practice Problems Name:
 Boyle s Law Practice Problems Name: 1. If a gas at 25.0 C occupies 3.60 L at a pressure of 1.00 atm, what will be its volume at a pressure of 2.50 atm? 2. 500.0 ml of a gas is collected at 745.0 mm Hg.
Boyle s Law Practice Problems Name: 1. If a gas at 25.0 C occupies 3.60 L at a pressure of 1.00 atm, what will be its volume at a pressure of 2.50 atm? 2. 500.0 ml of a gas is collected at 745.0 mm Hg.
Hazardous Communication. 60 Minutes
 Hazardous Communication 60 Minutes OBJECTIVE By the end of this session, you will know and understand the most current OSHA requirements of a good Hazardous Communication program Label Components with
Hazardous Communication 60 Minutes OBJECTIVE By the end of this session, you will know and understand the most current OSHA requirements of a good Hazardous Communication program Label Components with
Temp in Kelvin = (Temp in O C) Temp in o C = (Temp in Kelvin) Perform the following conversions:
 Study: *Questions About Gases WS and More Questions About Gases WS *Go back over your notes taken from the book and in class. *Use your book: Chapter 3 PG 66-97 The Kelvin temperature of a gas (or any
Study: *Questions About Gases WS and More Questions About Gases WS *Go back over your notes taken from the book and in class. *Use your book: Chapter 3 PG 66-97 The Kelvin temperature of a gas (or any
GAS EXCHANGE & PHYSIOLOGY
 GAS EXCHANGE & PHYSIOLOGY Atmospheric Pressure Intra-Alveolar Pressure Inspiration 760 mm HG at Sea Level (= 1 atm) Pressure due to gases (N2, O2, CO2, Misc.) Pressure inside the alveolus (air sac) Phrenic
GAS EXCHANGE & PHYSIOLOGY Atmospheric Pressure Intra-Alveolar Pressure Inspiration 760 mm HG at Sea Level (= 1 atm) Pressure due to gases (N2, O2, CO2, Misc.) Pressure inside the alveolus (air sac) Phrenic
Gaseous exchange. Grade 11
 z Gaseous exchange Grade 11 z Terminology 1. Breathing 2. Gaseous exchange 3. Diffusion 4. Spongy mesophyll cells 5. Tracheae 6. Gills 7. Alveoli 8. Larynx 9. Diaphragm 10. Endothelium 1. Pleura 2. Squamous
z Gaseous exchange Grade 11 z Terminology 1. Breathing 2. Gaseous exchange 3. Diffusion 4. Spongy mesophyll cells 5. Tracheae 6. Gills 7. Alveoli 8. Larynx 9. Diaphragm 10. Endothelium 1. Pleura 2. Squamous
Supporting inquiry based teaching and learning. Respiration. Student Name: Class:
 Supporting inquiry based teaching and learning Respiration Student Name: Class: Teacher Name: School: Question 1 a) Respiration is the process that the swimmer uses to release energy from the digested
Supporting inquiry based teaching and learning Respiration Student Name: Class: Teacher Name: School: Question 1 a) Respiration is the process that the swimmer uses to release energy from the digested
Key Terms Chapter 7. boiling boiling point change of state concentration condensation deposition evaporation flow rate fluid freezing point
 Foldable Activity Using the instructions on page 267 in your textbook on how to make foldables, write a key term on each front tab, and the definition on the inside (see example that I made up). You will
Foldable Activity Using the instructions on page 267 in your textbook on how to make foldables, write a key term on each front tab, and the definition on the inside (see example that I made up). You will
Unit 9 Packet: Gas Laws Introduction to Gas Laws Notes:
 Name: Unit 9 Packet: Gas Laws Introduction to Gas Laws Notes: Block: In chemistry, the relationships between gas physical properties are described as gas laws. Some of these properties are pressure, volume,
Name: Unit 9 Packet: Gas Laws Introduction to Gas Laws Notes: Block: In chemistry, the relationships between gas physical properties are described as gas laws. Some of these properties are pressure, volume,
alveoli Chapter 42. Gas Exchange elephant seals gills AP Biology
 alveoli Chapter 42. Gas Exchange gills elephant seals Gas exchange O 2 & CO 2 exchange exchange between environment & cells provides O 2 for aerobic cellular respiration need moist membrane need high
alveoli Chapter 42. Gas Exchange gills elephant seals Gas exchange O 2 & CO 2 exchange exchange between environment & cells provides O 2 for aerobic cellular respiration need moist membrane need high
Unit 14 Gas Laws Funsheets
 Name: Period: Unit 14 Gas Laws Funsheets Part A: Vocabulary and Concepts- Answer the following questions. Refer to your notes and the PowerPoint for help. 1. List 5 different common uses for gases: a.
Name: Period: Unit 14 Gas Laws Funsheets Part A: Vocabulary and Concepts- Answer the following questions. Refer to your notes and the PowerPoint for help. 1. List 5 different common uses for gases: a.
Human gas exchange. Question Paper. Save My Exams! The Home of Revision. Cambridge International Examinations. 56 minutes. Time Allowed: Score: /46
 Human gas exchange Question Paper Level Subject Exam oard Topic Sub Topic ooklet O Level iology ambridge International Examinations Respiration Human gas exchange Question Paper Time llowed: 56 minutes
Human gas exchange Question Paper Level Subject Exam oard Topic Sub Topic ooklet O Level iology ambridge International Examinations Respiration Human gas exchange Question Paper Time llowed: 56 minutes
Dead Perch Parts. ACADEMIC STANDARDS: 4 th Grade B. Know that living things are made up of parts that have specific functions.
 Dead Perch Parts Fish Anatomy Adapted from: An original Creek Connections activity created from the Fish Anatomy model. Grade Level: Intermediate or advanced Duration: 30 minutes Setting: classroom Summary:
Dead Perch Parts Fish Anatomy Adapted from: An original Creek Connections activity created from the Fish Anatomy model. Grade Level: Intermediate or advanced Duration: 30 minutes Setting: classroom Summary:
To derive from experiment the relationships between Pressure (P), Volume (V), Temperature (T), and Water Solubility of gases.
 PROPERTIES OF GASES: PRESSURE, VOLUME, TEMPERATURE, & SOLUBILITY RELATIONSHIPS PURPOSE: To derive from experiment the relationships between Pressure (P), Volume (V), Temperature (T), and Water Solubility
PROPERTIES OF GASES: PRESSURE, VOLUME, TEMPERATURE, & SOLUBILITY RELATIONSHIPS PURPOSE: To derive from experiment the relationships between Pressure (P), Volume (V), Temperature (T), and Water Solubility
CONCENTRATION CARBON DIOXIDE
 MONITORING THE CONCENTRATION OF CARBON DIOXIDE In a New York City Classroom BY: Ameena Peters Director: Philip Orton Advisor: Mauricio Gonzalez New York Harbor School Marine Research, 2011-2013 Contents
MONITORING THE CONCENTRATION OF CARBON DIOXIDE In a New York City Classroom BY: Ameena Peters Director: Philip Orton Advisor: Mauricio Gonzalez New York Harbor School Marine Research, 2011-2013 Contents
The Human Body. Everyone Needs Healthy Systems. Blood Vessels
 The Human Body Everyone Needs Healthy Systems There are several systems that make up the human body. Although their functions differ, they all work together to keep your body running smoothly. Some of
The Human Body Everyone Needs Healthy Systems There are several systems that make up the human body. Although their functions differ, they all work together to keep your body running smoothly. Some of
Chapter 13 Temperature, Kinetic Theory, and the Gas Laws 497
 Chapter 13 Temperature, Kinetic Theory, and the Gas Laws 497 Figure 13.25 This photograph of Apollo 17 Commander Eugene Cernan driving the lunar rover on the Moon in 1972 looks as though it was taken at
Chapter 13 Temperature, Kinetic Theory, and the Gas Laws 497 Figure 13.25 This photograph of Apollo 17 Commander Eugene Cernan driving the lunar rover on the Moon in 1972 looks as though it was taken at
